US Pharm. 2015;40(11):53-57.
ABSTRACT: Posttraumatic stress disorder (PTSD) is a mental health condition that is triggered by experiencing or witnessing a traumatic event involving the possibility of serious injury or death. Research demonstrates that cognitive-behavioral therapy (CBT) is the most effective treatment for PTSD. The two types of CBT that are effective for PTSD are cognitive processing therapy and prolonged exposure therapy. If nonpharmacologic treatments fail to control symptoms of PTSD, the Veterans Affairs/Department of Defense clinical practice guideline and the American Psychiatric Association guideline recommend using medications in the treatment of PTSD. Both guidelines recommend selective serotonin reuptake inhibitors or serotonin-norepinephrine reuptake inhibitors as first-line treatment.
Posttraumatic stress disorder (PTSD) is a mental health condition that is triggered by experiencing or witnessing a traumatic event (e.g., sexual assault, terrorism, exposure to war as a combatant or civilian, threatened or actual physical assault) involving the possibility of serious injury or death.1 According to the National Comorbidity Survey, the estimated lifetime prevalence of PTSD among adult Americans is 7.8%.2 Women are twice as likely as men to have PTSD at some point in their lives.2
PTSD is associated with a high risk of suicidal ideation and suicide attempts, comorbid mental health disorders (i.e., depression, anxiety, panic disorder, and bipolar disorder), and substance abuse.1,3 Patients with PTSD also are more likely to engage in high-risk behavior (i.e., smoking, alcohol/drug abuse, unsafe weapon storage, reckless driving, sexual promiscuity).3 Because of the high prevalence of substance abuse and psychiatric comorbidities, patients being managed for PTSD should receive a comprehensive assessment for substance-use disorder and should be screened for depression, bipolar disorder, and other psychiatric disorders.3,4 Patients should also be assessed for safety and risk to self or others.3,4
Pathophysiology
Several neurobiologic systems have been implicated in the pathophysiology of PTSD, including the noradrenergic, serotonergic, endogenous cannabinoid (CB), and opioid systems and the hypothalamic-pituitary-adrenal (HPA) axis. The noradrenergic system includes a group of G protein–coupled receptors (adrenoreceptors) consisting of three major classifications: alpha1-, alpha2-, and beta-receptors with associated subtypes.5 The norepinephrine transporter (NET) is a potential noradrenaline target for the treatment of PTSD.5 The serotonergic (5-HT) system is involved in cognition, emotional processing, and behavioral regulation. 5-HT1A and 5-HT1B have been implicated in stress disorders and are targets for treatment. The CBs exert much of their action through the two known CB receptors (CB1, CB2), which play an important role in stress responses.5 Opioid receptors are generally classified into at least three subtypes—delta (encephalin-preferring), kappa (dynorphin-preferring), and mu (morphine-preferring)—and have been implicated in PTSD through the signaling response during acute stress.1 Finally, the HPA axis promotes adaptation to stress and is dysregulated in PTSD through increased levels of corticotropin-releasing factor and increased release of cortisol.5
Other neurotransmitters have been implicated in the pathophysiology of PTSD, including the major excitatory neurotransmitter in the brain (glutamate) and the major inhibitory neurotransmitter (gamma-aminobutyric acid).5
Diagnostic Features
Diagnostic criteria for PTSD in adults, adolescents, and children aged >6 years are based on specific criteria from The Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). Diagnostic criteria for PTSD include exposure to a traumatic event that results in specific symptoms: intrusion, avoidance, negative alterations in cognition and mood, and alterations in arousal and reactivity.1 These symptoms must persist for >1 month and cause clinically significant impairment in functioning. Lastly, the disturbance should not be associated with the physiological effects of a substance (e.g., alcohol) or another medical condition.1
Nonpharmacologic Treatment Options
It is strongly recommended that patients diagnosed with PTSD be offered evidence-based, trauma-focused psychotherapeutic interventions (TABLE 1).3 According to the Veterans Affairs/Department of Defense (VA/DoD) 2010 Clinical Practice Guideline for Management of Post-Traumatic Stress, psychotherapy interventions aim to reduce symptom severity, improve global functioning, and improve quality of life and functioning in social and occupational areas.3 Cognitive-behavioral therapy (CBT) is the most effective treatment for PTSD.6 The two types of CBT that have proven most effective for PTSD are cognitive processing therapy (CPT) and prolonged exposure therapy (PE).7,8 CPT helps patients challenge and change negative thoughts about trauma, and PE helps patients confront memories and thoughts that they have been avoiding. Another effective therapy is eye movement desensitization and reprocessing (EMDR). EMDR encourages patients to think of images and feelings that are distressing while simultaneously focusing on hand movements or tapping.7 These evidence-based psychotherapy treatments are time-limited, typically weekly for 3 to 4 months. Patients who participate in these therapies are able to positively manage symptoms for years.7

Pharmacologic Treatment Options
If nonpharmacologic treatment options fail to control PTSD symptoms, the VA/DoD clinical practice guideline and the American Psychiatric Association (APA) guideline recommend using medications to minimize and manage symptoms. Medication may be administered (TABLE 2) in conjunction with CBT.1,2 The VA/DoD and APA guidelines endorse the use of selective serotonin reuptake inhibitors (SSRIs; fluoxetine, paroxetine, sertraline) or serotonin-norepinephrine reuptake inhibitors (SNRIs; venlafaxine) as first-line treatment.3,4 If first-line agents fail to control symptoms, both guidelines recommend the use of mirtazapine, nefazodone, tricyclic antidepressants (TCAs; amitriptyline and imipramine), or monoamine oxidase (MAO) inhibitors (phenelzine) as second-line treatment.3 In terms of residual symptoms, prazosin may be added to manage nightmares.
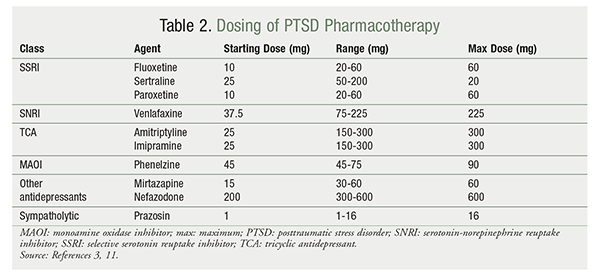
TABLE 3 provides a listing of which core PTSD symptoms (reexperiencing, avoidance/numbing, and hyperarousal) respond to the various pharmacologic agents used to treat PTSD.3
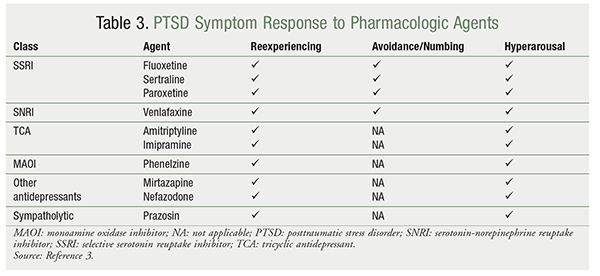
SSRIs: SSRIs reduce the three core symptoms of PTSD.3,4 Sertraline and paroxetine are currently the only pharmacotherapy agents FDA-approved for the management of PTSD.9,10 As a class, SSRIs work by increasing the level of serotonin in the peripheral and central nervous systems.9,11,12 Different SSRIs have slightly different mechanisms that may contribute to their efficacy.
Sertraline displays mild antagonist actions at sigma receptors; it also blocks the dopamine reuptake pump.11,12 This dopamine reuptake–blocking property may contribute to sertraline’s ability to improve symptoms of hypersomnia, low energy, and mood reactivity.11,12 The antagonist action at sigma receptors is not well understood, but it may contribute to sertraline’s anxiolytic effects and help relieve symptoms of psychotic and delusional depression.11,12
Paroxetine has muscarinic anticholinergic and weak NET inhibitory actions.11,12 Paroxetine’s anticholinergic properties may cause the calming and sedating effects of the medication, and its NET inhibitory properties may contribute to efficacy in depression.12
Fluoxetine also has antagonist properties at 5-HT2C receptors, which may lead to the release of norepinephrine and dopamine.11,12 This unique property may be useful in patients with daytime fatigue and help improve concentration and attention.12
Other SSRIs may be beneficial in the management of symptoms of PTSD; however, there is currently insufficient evidence to recommend them as first-line agents.
SNRIs: The guidelines also recommend using an SNRI (venlafaxine) as first-line treatment.3,4 Venlafaxine reduces the three core symptoms of PTSD. This agent works by affecting the balance of serotonergic and noradrenergic neurotransmission.9,11,12 At lower dosages, venlafaxine inhibits the reuptake of serotonin; at higher dosages, it inhibits the reuptake of norepinephrine.11,12 Other SNRIs may be beneficial for managing PTSD core symptoms; however, there is currently insufficient evidence to recommend them as first-line agents.
TCAs: The guidelines recommend using TCAs as second-line agents based on their tolerability and side-effect profile. Amitriptyline and imipramine are the only TCAs that have demonstrated positive outcomes in PTSD management. TCAs reduce the core PTSD symptoms of reexperiencing and hyperarousal, but they have no effect on avoidance/numbing.3 As a class, TCAs work by blocking norepinephrine and serotonin reuptake pumps.11,12 TCAs also have unwanted pharmacologic actions; they block the muscarinic cholinergic receptors, H1 (histamine) receptors, alpha1-adrenergic receptors, and voltage-sensitive sodium channels.11,12 Blockage of M1-muscarinic cholinergic receptors causes dry mouth, blurred vision, urinary retention, and constipation; blockage of histaminic receptors causes sedation and weight gain. Blockage of alpha1-adrenergic receptors causes orthostatic hypotension and dizziness, and blockage of voltage-sensitive sodium channels in the heart and brain can cause seizures, cardiac arrhythmia, cardiac arrest, and death, especially in overdose cases.9,11,12
Other Antidepressants: Phenelzine, mirtazapine, and nefazodone reduce the core PTSD symptoms of reexperiencing and hyperarousal, but they have no effect on avoidance/numbing.3
Mirtazapine works by blocking the presynaptic alpha2-adrenergic inhibitory autoreceptor, resulting in an increased release of norepinephrine and serotonin.11,12 It also blocks postsynaptic 5-HT2 and 5-HT3 receptors and H1 receptors.11,12 One advantage of this agent is that it causes less sexual dysfunction than do SSRIs.9 Mirtazapine may also help manage insomnia in patients with PTSD.
Nefazodone blocks 5-HT2 receptors potently and blocks reuptake of serotonin and norepinephrine pumps less potently.9,11,12 It is no longer commonly used, owing to its black box warning regarding liver failure.11,12
Phenelzine inhibits MAO, an enzyme that degrades neurotransmitters such as serotonin, norepinephrine, and dopamine.11,12 Phenelzine is not commonly used owing to its potential for serious drug interactions, as well as dietary restrictions.11,12 Patients taking phenelzine must limit tyramine-rich foods (e.g., aged cheeses and meats, sauerkraut) owing to the risk of hypertensive crisis.11,12
Adrenergic Inhibitors: Prazosin may be used adjunctively for the management of nightmares associated with PTSD. This agent reduces the core PTSD symptoms of reexperiencing and hyperarousal, but has no effect on avoidance/numbing.3 Prazosin works by blocking alpha1-adrenergic receptors to reduce noradrenergic hyperactivation.11,12 Stimulation of central noradrenergic receptors during sleep is thought to activate traumatic memories; therefore, blocking these receptors may reduce nightmares.12
Medications With Limited or Minimal Proven Efficacy
Some medications are currently being prescribed for management of PTSD without supporting evidence from studies. Many small, open-label trials have shown promise for certain drug classes, whereas randomized, controlled trials have displayed conflicting results.13 These medications include benzodiazepines, mood stabilizers/anticonvulsants, and antipsychotics. These classes have shown poor effectiveness in managing PTSD, and their side-effect profiles and monitoring parameters outweigh their benefits.3,13
Benzodiazepines are commonly prescribed for PTSD without supporting evidence of improvements in core PTSD symptoms.3 They have been shown effective for quick symptomatic control of insomnia, anxiety/panic, and irritability, but they worsen PTSD by potentiating the acquisition of fear responses in military veterans and impede recovery from trauma.3,13,14 In a small study, eight veterans with combat-induced PTSD exhibited severe withdrawal reactions while taking a benzodiazepine.3 Withdrawal symptoms of benzodiazepines included anxiety, sleep disturbances, rage, hyperalertness, increased nightmares, and intrusive thoughts.3 Another problem associated with benzodiazepine use is the development of tolerance and dependence.
Anticonvulsants (carbamazepine, oxcarbazepine, valproate, lamotrigine, gabapentin, topiramate, phenytoin) are prescribed for PTSD based on their mood-stabilizing characteristics.13,14 There is little evidence supporting their use in open-label trials, and randomized, controlled trials have not had promising results.14 Because of conflicting evidence of efficacy, monitoring parameters (e.g., serum trough levels), and side effects (sedation, rash, poor concentration, sexual dysfunction, memory problems), guidelines do not support the use of anticonvulsants in PTSD.3,14,15
Atypical antipsychotics are another drug class used for PTSD. As with previously mentioned classes, evidence supporting their use in PTSD is conflicting and their use is not recommended. The largest clinical trial examining use of an atypical antipsychotic (risperidone) for PTSD was conducted by the VA Cooperative Study Group in 2010.3 After 6 months of treatment, there were no clinically significant differences in symptoms of anxiety, depression, sleep, and quality of life compared with placebo.3 The risk of metabolic side effects (weight gain, dyslipidemia, elevated blood glucose) with atypical antipsychotics outweighs the benefits.3,14
Conclusion
The VA/DoD and APA guidelines recommend using CBT as first-line treatment for PTSD. Medication may be added to CBT to control symptoms. Currently, the guidelines recommend using SSRIs or SNRIs as first-line agents. Sertraline and paroxetine are the only two medications with an FDA indication for PTSD. Further research is needed to determine the place of medications such as anticonvulsants and antipsychotics in PTSD therapy.
REFERENCES
1. American Psychiatric Association. The Diagnostic and Statistical Manual of Mental Health Disorders, Fifth Edition. Arlington, VA: American Psychiatric Association; 2013.
2. Gradus JL. Epidemiology of PTSD. Washington, DC: U.S. Department of Veterans Affairs; 2007.
3. VA/DoD Clinical Practice Guideline for Management of Post-Traumatic Stress. Washington, DC: U.S. Department of Veterans Affairs and Department of Defense; 2010.
4. Benedek DM, Friedman MJ, Zatzick D, Ursano RJ. Guideline Watch (March 2009): Practice Guideline for the Treatment of Patients With Acute Stress Disorder and Posttraumatic Stress Disorder. Arlington, VA: American Psychiatric Association; 2009.
5. Bailey CR, Cordell E, Sobin S, Neumeister A. Recent progress in understanding the pathophysiology of post-traumatic stress disorder: implications for targeted pharmacological treatment. CNS Drugs. 2013;27:221-232.
6. National Center for PTSD. Understanding PTSD treatment [brochure]; August 2013. www.ptsd.va.gov/public/understanding_TX/booklet.pdf. Accessed October 13, 2015.
7. National Center for PTSD. Cognitive processing therapy. www.ptsd.va.gov/public/treatment/therapy-med/cognitive_processing_therapy.asp. Accessed July 20, 2015.
8. National Center for PTSD. Prolonged exposure therapy. www.ptsd.va.gov/public/treatment/therapy-med/prolonged-exposure-therapy.asp. Accessed July 20, 2015.
9. Jeffreys M. Clinician’s guide to medications for PTSD. National Center for PTSD. www.ptsd.va.gov/professional/treatment/overview/clinicians-guide-to-medications-for-ptsd.asp Accessed October 13, 2015.
10. Sertraline and paroxetine. Lexi-Drugs [subscription database]. Hudson, OH: Lexi-Comp, Inc; January 29, 2015.
11. Stahl SM. Stahl’s Essential Psychopharmacology: Prescriber’s Guide. 5th ed. New York, NY: Cambridge University Press; 2014.
12. Stahl SM. Stahl’s Essential Psychopharmacology: Neuroscientific Basis and Practical Applications. 4th ed. New York, NY: Cambridge University Press; 2013.
13. Warner CH, Warner CM, Appenzeller GN, Hoge CW. Identifying and managing posttraumatic stress disorder. Am Fam Physician. 2013;88:827-834.
14. Kapfhammer HP. Patient-reported outcomes in post-traumatic stress disorder. Part II: focus on pharmacological treatment. Dialogues Clin Neurosci. 2014;16:227-237.
15. Van Ameringen M, Mancini C, Pipe B, Bennett M. Antiepileptic drugs in the treatment of anxiety disorders: role in therapy. Drugs. 2004;64:2190-2220.
To comment on this article, contact rdavidson@uspharmacist.com.





