US Pharm. 2015;40(1):24-28.
ABSTRACT: Cannabis, the substance more commonly known as marijuana, has gained interest in recent years for its potential use as an antiepileptic agent. The two main components of marijuana are delta-9-tetrahydrocannabinol, which has had mixed effects in epilepsy, and cannabidiol, which has shown more consistent anticonvulsant effects. Data supporting the use of marijuana for this purpose are limited, as the majority of clinical trials were conducted before 1990. There are case reports showing promising results; however, the data are inconsistent and cannot be generalized. The legal issues surrounding marijuana in the United States also may limit the use of this substance as an antiepileptic.
Epilepsy is defined as recurrent and ongoing seizures caused by changes in neuronal firing in the brain. Whereas nonepileptic seizures are not associated with neurophysiological changes, 3% of the population is predisposed to otherwise unprovoked, recurrent epileptic seizures.1 Current pharmacotherapy for epilepsy aims to restore normal neuronal function and decrease seizure frequency. Prospective, randomized trials estimate that individuals experiencing a first, unprovoked seizure have a 40% to 50% chance of seizure recurrence at 2 years. The risk of recurrence, which diminishes with time, is highest immediately following the first seizure, with 80% to 90% of patients experiencing recurrent seizures within the first 2 years.2,3 Despite available treatments, about 30% of patients remain resistant to therapy (fail two or more antiepileptics), resulting in poorly controlled and recurring seizures.4,5 This review discusses the current research on, rationale for, and limitations to the use of marijuana for the treatment of seizure disorders.
Historical Medical Use of Cannabis
The earliest documented use of cannabis (marijuana) occurred in about 2,700 bc in China, where it was used for a variety of medical ailments, including gout, malaria, constipation, menstrual disorders, and absentmindedness. Western medicine adopted the use of cannabis as a common analgesic in the 19th century.6
Cannabis was available in U.S. pharmacies as an OTC product until the 1937 Marihuana Tax Act limited its accessibility. Subsequently, the passing of the Controlled Substances Act in 1970 gave cannabis a Schedule I classification, making its use illegal.7 Since 1970, there has been increasing interest in the use of marijuana for its possible antiepileptic properties.4
Pharmacology
Cannabis sativa and Cannabis indica are two species of the Cannabis genus of flowering plants. Both of these species have a long history of use as an antiepileptic, with sativa strains causing more psychotropic and stimulating effects and indica strains causing more sedation.6 Compounds contained in the cannabis plant are known as cannabinoids.
Cannabis contains two main components: the psychoactive portion of marijuana known as delta-9-tetrahydrocannabinol (THC) and the nonpsychoactive portion known as cannabidiol (CBD). What makes cannabis an attractive agent for epilepsy is the presence of cannabinoid type 1 receptors in the hippocampus and amygdala, both of which are associated with partial seizures.8 The THC component of cannabis is a partial agonist at these receptors.9 Conversely, CBD interacts with other nonendocannabinoid signaling systems, reducing the psychotropic activity of THC while increasing tolerance.6 Recent trials of CBD have shown more consistent anticonvulsant properties, and this cannabinoid has gained interest as a possible agent for epilepsy.9,10
There are many potential routes of administration for synthetic CBD, the only non–delta-9-THC phytocannabinoid assessed for its anticonvulsant effects in clinical trials.6 The most common delivery route is by inhalation, either recreationally or for medicinal purposes. Because of the highly lipophilic nature of CBD and its high volume of distribution, the lungs are an effective route of medication delivery, with rapid distribution into the brain, adipose tissue, and organs. Cannabinoids are extensively metabolized by the liver, predominantly by CYP3A2, CYP3A4, CYP2C8, CYP2C9, and CYP2C19. Owing to significant first-pass metabolism through the liver, CBD is only about 6% bioavailable, thus rendering oral, oral-mucosal, and sublingual routes of delivery less desirable. The transdermal route of administration has also been considered; however, this route may be economically impractical, since special delivery systems are needed to prevent excessive accumulation of CBD in the skin.11,12
Marijuana as Treatment
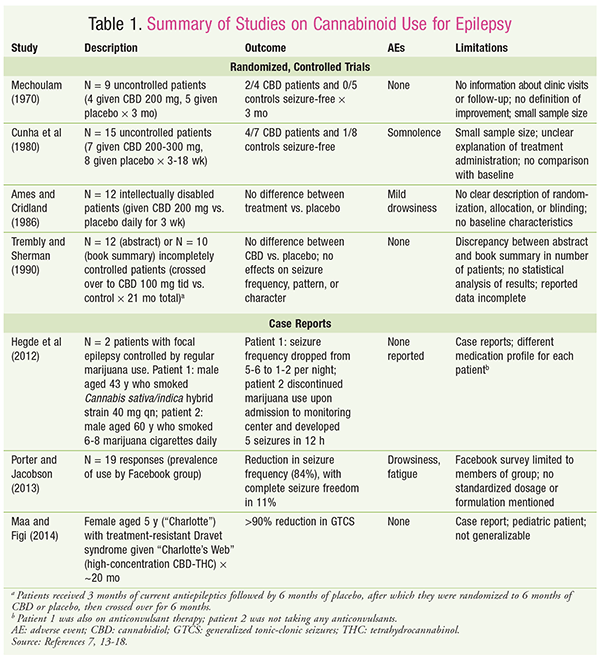
Clinical trials examining the efficacy of marijuana for treating epilepsy are limited. One of the earliest was a small randomized, controlled study conducted by Mechoulam in 1970.13 In this trial, nine patients with treatment-resistant temporal lobe epilepsy received either CBD or placebo for 5 weeks in addition to their current antiepileptic therapy. Two of the four CBD patients were seizure-free at 3-month follow-up, whereas none of the five placebo patients showed improvement. However, this trial was limited by its small sample size and lack of statistical analysis or power calculation.13
Cunha and colleagues conducted a small randomized, controlled trial involving 15 patients with generalized epilepsy.14 Seven patients received CBD and eight received matching placebo for 3 to 18 weeks. There were no reported toxicities, and four CBD patients compared with just one placebo patient were seizure-free. This study was limited in that it had no performed power calculation, no statistical analysis, and a small sample size.14
Although a number of early small, controlled studies demonstrated some efficacy of CBD for epilepsy, more recent studies indicate that CBD has limited or no effect on epilepsy. In one trial, Trembly and Sherman examined the effect of marijuana on uncontrolled epilepsy.15 No discernible effect was found overall, and there was no statistical analysis of trial outcomes or main effects. A study conducted by Ames and Cridland showed no difference between CBD and placebo.16 As was the case in previous trials, the study population was small and there were no power calculations.16
Various case reports have identified adult patients favoring marijuana as antiepileptic treatment, as well as parents who have sought marijuana with CBD content to treat resistant epilepsy in their children.7,17,18 Some of these reports yield promising information; however, there is no consensus on dosage, formulation, route of administration, or duration of marijuana therapy. Placebo effect and recall bias may be confounding variables. These limited case reports highlight that there is a paucity of safety and efficacy data from randomized, controlled trials to establish the use of marijuana for the treatment of epilepsy. See TABLE 1 for a summary of studies on cannabinoid use as an antiepileptic.
Adverse Effects
Adverse effects of chronic marijuana use include addiction risk, negative effects on brain development, increased risk of certain mental illnesses, motor-vehicle accidents, and various effects on health. According to the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, 9% of individuals who use marijuana may become addicted. In addition, discontinuation of habitual cannabis use may cause withdrawal symptoms such as irritability, insomnia, dysphoria, enhanced cravings, and increased anxiety.19
Marijuana initiation at a young age may impact brain development, as it can impair neural connectivity in specific areas of the brain, resulting in altered learning, memory, and function. Marijuana use in adolescence could lead to a heightened response to other drugs, paving the way for addictions to other drugs in adulthood.
Consistent marijuana use has been correlated with anxiety and depression; however, a causal relationship has not been established. There may also be a positive association between chronic marijuana smoking and an increased incidence of lung cancer, as well as other pulmonary diseases, through lung airway inflammation, airway resistance, and hyperinflation.8,19
Heavy and chronic marijuana use may be correlated with the majority of the adverse effects, mostly attributable to the THC component. However, there may also be confounding variables; therefore, causality cannot be established. Future studies involving CBD may result in a different side-effect profile, since multiple small studies of CBD safety in humans have shown adequate tolerance with no significant side effects at dosages up to 1,500 mg per day by mouth or 30 mg IV when used acutely and chronically.6,20
Current Medical Use
Currently, the FDA has not approved marijuana for any indication. However, 23 states and Washington, DC, have legalized the use of medical marijuana (containing delta-9-THC), and 22 states have approved it for seizure use (TABLE 2). In addition, Massachusetts allows marijuana for “conditions as determined in writing by a qualifying patient’s physician.”21 Medical marijuana use is licensed in Canada, the Netherlands, and Israel.9

Future research involving drug products derived from botanical marijuana or synthetic versions and substances that act similarly to marijuana must be approved via an Investigational New Drug Application before human trials can be conducted. In addition, the Drug Enforcement Administration reviews the researcher registration application, and the National Institute of Drug Abuse is responsible for supplying research marijuana for trials. Ultimately, for a drug product to be approved for the U.S. market, the FDA must ensure that it meets necessary quality standards and is safe and efficacious.22
Conclusion
There is insufficient evidence to form a reliable conclusion regarding the efficacy of marijuana as an antiepileptic agent. Despite case reports demonstrating efficacy in reducing seizure frequency and severity, limited clinical studies have been published on its use for this indication. Additionally, the studies conducted were inadequately powered, lacked complete information, and used small sample sizes. There are few studies of long-term administration of cannabis and its safety profile. Currently, legal restrictions on cannabis make it difficult to conduct large-scale clinical trials, as the FDA has classified marijuana as a Schedule I controlled substance. The utility of marijuana for the therapeutic treatment of epilepsy cannot be determined at this time; more large-scale studies are needed that assess the efficacy and safety of treatment with either high CBD-THC ratio marijuana or isolated CBD compounds.
REFERENCES
1. Chang BS, Lowenstein DH. Epilepsy. N Engl J Med. 2003;349:1257-1266.
2. Marson A, Jacoby A, Johnson A, et al. Immediate versus deferred antiepileptic drug treatment for early epilepsy and single seizures: a randomised controlled trial. Lancet. 2005;365:2007-2013.
3. Berg AT. Risk of recurrence after a first unprovoked seizure. Epilepsia. 2008;49(suppl 1):13-18.
4. Gloss D, Vickrey B. Cannabinoids for epilepsy. Cochrane Database Syst Rev. 2014;(3):CD009270.
5. Hill TD, Cascio MG, Romano B, et al. Cannabidivarin-rich cannabis extracts are anticonvulsant in mouse and rat via a CB1 receptor-independent mechanism. Br J Pharmacol. 2013;170:679-692.
6. Devinsky O, Cilio MR, Cross H, et al. Cannabidiol: pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. Epilepsia. 2014;55:791-802.
7. Maa E, Figi P. The case for medical marijuana in epilepsy. Epilepsia. 2014;55:783-786.
8. Watson SJ, Benson JA Jr, Joy JE. Marijuana and medicine: assessing the science base: a summary of the 1999 Institute of Medicine report. Arch Gen Psychiatry. 2000;57:547-552.
9. Cilio MR, Thiele EA, Devinsky O. The case for assessing cannabidiol in epilepsy. Epilepsia. 2014;55:787-790.
10. Hill AJ, Williams CM, Whalley BJ, Stephens GJ. Phytocannabinoids as novel therapeutic agents in CNS disorders. Pharmacol Ther. 2012;133:79-97.
11. Hawksworth G, McArdle K. Metabolism and pharmacokinetics of cannabinoids. In: Whittle BA, Guy GW, Robson PJ, eds. Medicinal Uses of Cannabis and Cannabinoids. London, England: Pharmaceutical Press; 2004:205-228.
12. Lodzki M, Godin B, Rakou L, et al. Cannabidiol-transdermal delivery and anti-inflammatory effect in a murine model. J Control Release. 2003;93:377-387.
13. Mechoulam R. Marihuana chemistry. Science. 1970;168:1159-1166.
14. Cunha JM, Carlini EA, Pereira AE, et al. Chronic administration of cannabidiol to healthy volunteers and epileptic patients. Pharmacology. 1980;21:175-185.
15. Trembly B, Sherman M. Double-blind clinical study of cannabidiol as a secondary anticonvulsant. Paper presented at: Marijuana ’90 International Conference on Cannabis and Cannabinoids; July 8-11, 1990; Kolympari, Crete.
16. Ames FR, Cridland S. Anticonvulsant effect of cannabidiol. S Afr Med J. 1986;69:14.
17. Porter BE, Jacobson C. Report of a parent survey of cannabidiol-enriched cannabis use in pediatric treatment-resistant epilepsy. Epilepsy Behav. 2013;29:574-577.
18. Hegde M, Santos-Sanchez C, Hess CP, et al. Seizure exacerbation in two patients with focal epilepsy following marijuana cessation. Epilepsy Behav. 2012;25:563-566.
19. Volkow ND, Baler RD, Compton WM, Weiss SR. Adverse health effects of marijuana use. N Engl J Med. 2014;370:2219-2227.
20. Bergamaschi MM, Queiroz RH, Zuardi AW, Crippa JA. Safety and side effects of cannabidiol, a Cannabis sativa constituent. Curr Drug Saf. 2011;6:237-249.
21. ProCon.org. Legal medical marijuana states and DC. http://medicalmarijuana.procon.org/view.resource.php?resourceID=000881. Accessed September 9, 2014.
22. FDA. FDA and marijuana. www.fda.gov/NewsEvents/PublicHealthFocus/ucm421163.htm. Accessed December 3, 2014.
To comment on this article, contact rdavidson@uspharmacist.com.





