US Pharm. 2016;41(6):HS2-HS8.
ABSTRACT: The number of medications used to manage autoimmune disorders has increased dramatically. These agents—the immunomodulators—have resulted in improvements in patient outcomes and, in some cases, slowing of disease progression. Despite these advances, immunomodulators are also associated with substantial medication-safety concerns. The potential for adverse drug reactions, serious drug-related complications, and medication errors is significant. Pharmacists must be acutely aware of immunomodulators’ complexity and associated potential for adverse events. Attention to all aspects of the medication-use process—procurement and storage, prescribing, dispensing, administration, and monitoring—is required to assure that these agents are used in the safest and most efficacious manner.
Autoimmune disorders comprise a broad spectrum of disease states and conditions most simply described as the immune system’s reaction to components of its own body. Accordingly, the vast number of medications used to manage these disorders either suppress or stimulate an element of the immune system, overriding the devastating effects of immune-mediated disease. These medications, referred to as immunomodulating agents or immunomodulators, have resulted in improvements in patient outcomes and, in some cases, slowing of disease progression. Despite their contribution to disease management, these powerful agents often augment complications, and their complexity leads to medication safety concerns. It is beyond the scope of this review to discuss all immunomodulators; rather, categories of associated adverse events (AEs) and necessary safeguards when using this drug class will be identified.
Classification of Agents
In general, immunomodulators may be divided into two categories: immunosuppressants and immunostimulants. Within each category, there are several types of medications, each with unique mechanisms of action, immune targets, and indications (both labeled and unlabeled).
Medication Safety, AEs, and the Medication-Use Process
In the realm of medication safety, pharmacists must focus on adverse drug reactions as well as medication errors. Adverse reactions are unexpected in nature and imply an inability to predict which patients will experience an event. For example, in the absence of history, it is difficult to know which patient may have a hypersensitivity reaction to a monoclonal antibody (Mab). However, if a given agent has a high likelihood of infusion reactions, these reactions can be minimized with pretreatment and initiation at slower infusion rates. Medication errors are considered preventable (e.g., wrong dose or drug), and vigilance in improving the medication-use process to avoid common medication errors is of the utmost importance. This review will identify steps in the medication-use process for immunomodulators and safeguards that may be implemented in order to minimize adverse drug reactions and avoid medication errors.
Drug Procurement and Storage
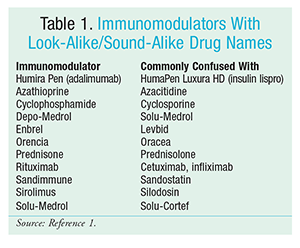
The potential for medication errors begins with the purchasing and storage of this complex group of drugs. Look-alike/sound-alike (LASA) nomenclature and packaging create the potential for confusion between products. It is beneficial to recognize known LASA drugs and use strategies such as tall-man lettering and caution labels in storage areas. The Institute for Safe Medication Practices (ISMP) regularly updates its list of commonly confused drug names and is an excellent source for preventive strategies at the start of the medication-use process.1 See TABLE 1.
It is also important to regularly review storage requirements. Although most immunomodulators are stable at room temperature, a review of the package labeling for unique requirements and avoidance of extreme temperature excursions will preserve the product’s integrity and protect the financial investment.
Prescribing
One of the greatest concerns for patients receiving immunomodulators is the risk of infection, both primary infection and reactivation of latent infections. Problems include reactivation of hepatitis B and hepatitis C viruses, latent tuberculosis (TB), invasive fungal infections, Pneumocystis jiroveci pneumonia (PJP), herpesviruses, HIV, and progressive multifocal leukoencephalopathy (PML). Prior to initiation, patients must be screened for risk factors or historical exposure to the pathogens associated with these infections. In some circumstances, prophylaxis (e.g., trimethoprim-sulfamethoxazole for PJP), concurrent therapy (e.g., treatment of latent TB), or immunization prior to therapy (e.g., zoster vaccine live 2-4 weeks before therapy in patients aged 60 years) may be indicated. In many cases, evidence-based guidelines are lacking and the decision to initiate or continue immunomodulator therapy is one of risk versus benefit.2,3
PML, a serious demyelinating brain disease caused by the JC polyomavirus (JCV), is a significant concern that has been reported in patients receiving selected agents,4,5 prompting intensive patient education and monitoring. JCV antibody testing is recommended in patients who are candidates for natalizumab. If the result is antibody-negative, the patient has not been exposed and is deemed to be at negligible risk for PML. Despite this recent advance, vigilant monitoring for PML signs and symptoms remains critical.6
Although it is not practical for the health-system pharmacist to be acutely aware of the emerging data on infection risk for the multitude of immunomodulators currently available, the pharmacist should work with health-system rheumatologists, neurologists, and others to assure that a strategy is in place to screen and monitor patients for potential infection risk.
With respect to infection prevention, the pharmacist should take an active role in ensuring that patients are current with preventive inactivated vaccines (e.g., influenza, pneumococcal). The use of live vaccines is not an absolute contraindication; however, as a general rule, immunomodulator therapy should be withheld for 3 months. The exception is patients receiving glucocorticoids, in which case a 1-month washout period is recommended.7
Given the expansive AE profile associated with the immunomodulator class (TABLE 2), it is also important to measure pertinent baseline laboratory parameters and follow them prior to drug initiation. Some reactions are not preventable (e.g., transaminase elevation, blood pressure changes, lipid abnormalities), but if the patient is rigorously monitored, decisions can be made regarding potential alternatives, risk versus benefit, or additional medications to manage side effects.
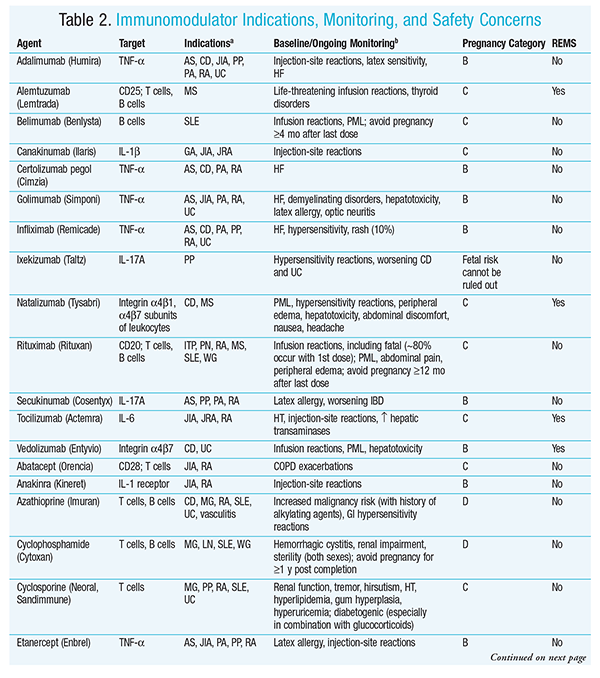
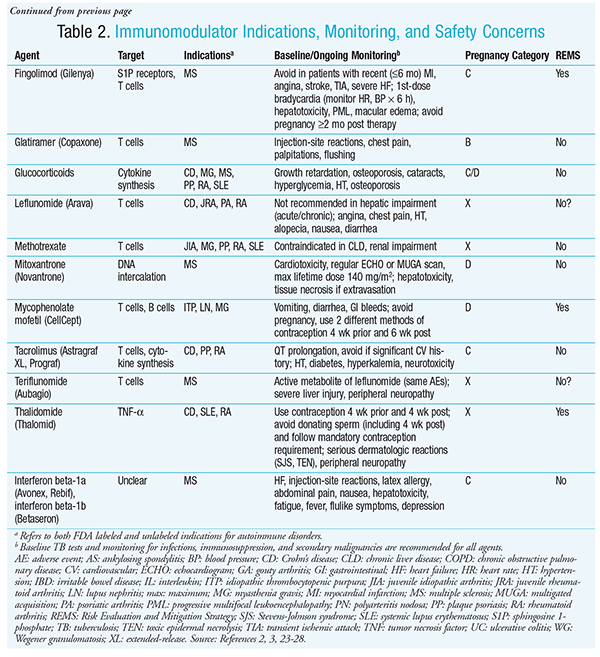
Several immunomodulators (e.g., mitoxantrone, teriflunomide) are known to be teratogenic, with pregnancy categories of D and X (TABLE 2). Pregnancy tests and counseling on contraceptive options are of the utmost importance prior to drug initiation.
One simple safeguard for prescribing is to discourage the use of abbreviations. The complicated nature of many drug names and administration schedules increases the likelihood of abbreviated instructions; history, however, has taught that this practice can lead to medication errors. For example, ISMP reported that one patient prescribed adalimumab “qoweek” (whereas “every other week” was intended) received a doubled dose of adalimumab every week for >9 months.8
Finally, drug-interaction screening and appropriate safety alerts within electronic systems are very important. Concomitant administration of multiple immunomodulators is generally not recommended (with the exception of glucocorticoids), and patients should not receive live vaccines. Other drug-drug interactions that are specific to individual drugs should be avoided as well. A key medication-safety tool for pharmacists when immunomodulators are being used is a reliable drug-interaction reference.
Dispensing
First, pharmacists should commit to a careful review of the aforementioned prescribing-safety issues prior to verification or dispensing of immunomodulators. Another consideration is careful handling to avoid occupational risk to healthcare professionals and to maintain product integrity. The National Institute for Occupational Safety and Health regularly publishes guidance documents and procedures for safe handling of hazardous drugs to avoid occupational risk.9 Also, several medications within the Mab class require gentle handling or no shaking to avoid foaming and other issues related to product integrity. For these reasons, such medications should routinely be placed on the “Do Not Tube” list in facilities that use pneumatic tube systems for drug delivery.
Administration
There are multiple considerations for medication safety in the administration portion of the medication-use process. Fingolimod, for example, is associated with first-dose bradycardia and requires close observation for >6 hours upon initial administration.10
Drugs administered SC are frequently associated with injection-site reactions and pain. The tumor necrosis factor-alpha blockers adalimumab and etanercept accounted for >10,000 injection-site reactions in a recent quarterly summary of injury reports received by the FDA.11 The occurrence of immediate postinjection reactions with SC glatiramer administration is well documented.12
Patients and nursing staff should be educated on methods to minimize injection-site reactions—for example, site rotation, warm or cold compresses, and smaller volumes of drug administration. There are many websites and blogs, particularly in the multiple sclerosis (MS) community, that address injection-site pain or reactions and offer remedies for improvement, although not all of these resources are scientifically based. It is also critically important to enhance patient adherence. Patients receiving interferon therapy for MS often report injection-site reactions and side effects such as fatigue, flulike symptoms, and muscle rigidity as reasons for drug discontinuation.13 Injection-site reactions and flulike symptoms tend to diminish after 1 year; if the patient is responding clinically, it is beneficial to encourage him or her to continue therapy, if possible.14 Also, a lower incidence of injection-site reactions has been reported with weekly IM dosing versus three times weekly SC dosing of interferon.15
Another safety concern is infusion-associated reactions (IARs) to immunomodulators administered IV. Reactions include headache, urticaria, fever, chills, hypotension, bradycardia, tachycardia, and anaphylaxis. Medications to manage reactions should be readily available, along with guidelines or algorithms for management. Premedication with antihistamines, antipyretics, antiemetics, and/or analgesics may be indicated depending on the medication and the patient’s history. Slowing the infusion rate may minimize minor reactions; however, infusions should be discontinued in severe circumstances. Pharmacists should take an active role in developing appropriate protocols and optimizing procedures to manage IARs.16,17
Lastly, latex allergy or sensitivity may be an issue with selected products. Adalimumab, etanercept, golimumab, and secukinumab are packaged in prefilled syringes with needle caps that contain latex.18-21 Some injection-site reactions may be the result of latex sensitivity, in which case patient screening and education may assist in minimizing side effects.
Monitoring
The primary safety issues concerning patient monitoring include those addressed in the Prescribing section, with attention centered on decision making regarding alternative therapy, polypharmacy to manage AEs, or methods for minimizing side effects. Lifelong users of immunomodulator therapy are at particular risk for infections, secondary malignancies, nephrotoxicity, hepatotoxicity, and impaired glycemic control. Recent preliminary data suggest that adults hospitalized with selected autoimmune diseases have a 20% increased risk of venous thromboembolism (VTE). Although more research is needed, close attention to VTE risk assessment and prophylaxis is especially important in this patient population.22
Conclusion
The immunomodulator class of medications is constantly evolving, presenting both great risks and great rewards to patients with devastating chronic illness. The pharmacist plays a critical role in ensuring that these medications are used safely and effectively. Systems to assure safe and appropriate drug selection, as well as dispensing and administration, should be in place and optimized where possible. Finally, consistent monitoring and ongoing patient counseling that includes reminders about appropriate contraception, immunizations (where appropriate), and vigilance regarding laboratory assessments are essential.
REFERENCES
1. Institute for Safe Medication Practices (ISMP). ISMP’s list of confused drug names. www.ismp.org/tools/confuseddrugnames.pdf. Accessed February 29, 2016.
2. Vodopivec I, Miloslavsky EM, Kotton CN, Cho TA. A neurologist’s guide to safe use of immunomodulatory therapies. Semin Neurol. 2014;34:467-478.
3. Wingerchuk DM, Carter JL. Multiple sclerosis: current and emerging disease-modifying therapies and treatment strategies. Mayo Clin Proc. 2014;89:225-240.
4. FDA. Tysabri (natalizumab): drug safety communication—new risk factor for progressive multifocal leukoencephalopathy (PML). www.fda.gov/safety/medwatch/safetyinformation/safetyalertsforhumanmedicalproducts/ucm288602.htm. Accessed March 20, 2016.
5. FDA. FDA drug safety communication: FDA warns about cases of rare brain infection with MS drug Gilenya (fingolimod) in two patients with no prior exposure to immunosuppressant drugs. www.fda.gov/Drugs/DrugSafety/ucm456919.htm. Accessed March 20, 2016.
6. Jeffery DR. Recent advances in treating multiple sclerosis: efficacy, risks and place in therapy. Ther Adv Chronic Dis. 2013;4:45-51.
7. Orlicka K, Barnes E, Culver EL. Prevention of infection caused by immunosuppressive drugs in gastroenterology. Ther Adv Chronic Dis. 2013;4:167-185.
8. ISMP. QOWEEK. www.ismp.org/newsletters/ambulatory/archives/200612_2.asp. Accessed March 5, 2016.
9. Connor TH, MacKenzie BA, DeBord DG, et al. NIOSH list of antineoplastic and other hazardous drugs in health care settings, 2014. Cincinnati, OH: National Institute for Occupa-tional Safety and Health; 2014. Publication Number 2014-138.
10. Galenya (fingolimod) product information. East Hanover, NJ: Novartis Pharmaceuticals Corp; February 2016.
11. ISMP. QuarterWatch 2014 annual report: signals for TNF blocking agents, anticoagulants, Lipitor, growth hormone. www.ismp.org/newsletters/acutecare/showarticle.aspx?id=119. Accessed March 5, 2016.
12. Anderson G, Meyer D, Herrman CE, et al. Tolerability and safety of half milliliter formulation of glatiramer for subcutaneous injection: an open-label, multicenter, randomized comparative study. J Neurol. 2010;257:1917-1923.
13. Daugherty KK, Butler JS, Mattingly M, Ryan M. Factors leading patients to discontinue multiple sclerosis therapies. J Am Pharm Assoc (2003). 2005;45:371-375.
14. Kappos L, Polman CH, Freedman MS, et al. Treatment with interferon beta-1b delays conversion to clinically definite and McDonald MS in patients with clinically isolated syndromes. Neurology. 2006;67:1242-1249.
15. Panitch H, Goodin DS, Francis G, et al. Randomized, comparative study of interferon beta-1a treatment regimens in MS: The EVIDENCE trial. Neurology. 2002;59:1496-1506.
16. McEwan L, Caon C, Chieffe C, et al. Best practices in alemtuzumab administration: practical recommendations for infusion in patients with multiple sclerosis. J Infus Nurs. 2016;39:93-104.
17. Namey M, Halper J, O’Leary S, et al. Best practices in multiple sclerosis: infusion reactions versus hypersensitivity associated with biologic therapies. J Infus Nurs. 2010;33:98-111.
18. Humira (adalimumab) product information. North Chicago, IL: AbbVie Inc; March 2016.
19. Enbrel (etanercept) product information. Thousand Oaks, CA: Amgen, Inc; March 2015.
20. Simponi (golimumab) product information. Horsham, PA: Centocor Ortho Biotech; 2009.
21. Cosentyx (secukinumab) product information. East Hanover, NJ: Novartis Pharmaceuticals Corp; January 2016.
22. Yusuf HR, Hooper WC, Beckman MG, et al. Risk of venous thromboembolism among hospitalizations of adults with selected autoimmune diseases. J Thromb Thrombolysis. 2014;38:306-313.
23. Ryan M, Deno S, Zwibel HL. Review of the clinical debate regarding interventions for multiple sclerosis. J Manag Care Pharm. 2009;15(suppl S-b):S1-S17.
24. Hart IK, Sathasivam S, Sharshar T. Immunosuppressive agents for myasthenia gravis. Cochrane Database Syst Rev. 2007;(4):CD005224.
25. Agarwal SK, Farheen K, Oderda GM, et al. Managed care strategies for improving patient outcomes in rheumatoid arthritis. J Manag Care Pharm. 2011;17(suppl 9-b):S3-S8.
26. Cheng FK, McLean LP, Cross RK. What is the role of vedolizumab in the era of anti-TNF agents? Ann Transl Med. 2014;2:1-4.
27. Roubille C, Haraoui B. Interstitial lung diseases induced or exacerbated by DMARDS and biologic agents in rheumatoid arthritis: a systematic literature review. Semin Arthritis Rheum. 2014;43:613-626.
28. Micromedex Healthcare Series [Internet database]. Version 5.1. Greenwood Village, CO: Thomson Micromedex; 2016.
To comment on this article, contact rdavidson@uspharmacist.com.






