US Pharm. 2009;34(11):40-53.
For the community pharmacist, the ability to aid in the management of opioid dependence is vital to closing the gap between treated and untreated opioid-dependent individuals. In the United States, approximately 2 million adults are dependent on heroin or other nonmedically prescribed opioids, yet only about 14% receive treatment.1,2 In an effort to provide care to a greater number of patients with dependence or addiction treatment needs, the Drug Addiction Treatment Act of 2000 (DATA 2000) was passed. DATA 2000 states that physicians who qualify can treat opioid dependence in the office setting with a schedule III, IV, or V drug that is FDA-approved for this indication.3 In 2002, buprenorphine (Subutex) and buprenorphine/naloxone (Suboxone) sublingual tablets were approved for the management of opioid dependence. As the practice of treating opioid dependence expands, pharmacists must become familiar with this chronic condition and stay up-to-date with current treatment options for patients on maintenance therapy.
NEUROBIOLOGY OF OPIOID DEPENDENCE AND WITHDRAWAL
Over the past 30 years, much has been discovered about opioid dependence that has improved our understanding of addiction as a chronic disease. Opioids activate specific opioid receptors (mu, delta, and kappa). Initially, when heroin or other short-acting opioids are taken, receptor agonists induce euphoria. Subsequent doses will quickly produce tolerance--the need for increasingly higher doses to induce the same effect--and physical dependence. Currently, it is believed that tolerance is a result of a reduction in either the number or the responsivity of opioid receptors.4,5
Chronic exposure to opioids causes upregulation of the cyclic adenosine monophosphate signaling pathways in neurons that are responsible for the release of noradrenaline. When the inhibitory opioid is no longer present, the firing rates of these neurons are unopposed and result in adrenergic overactivation, which manifests as the constellation of withdrawal symptoms.4,5
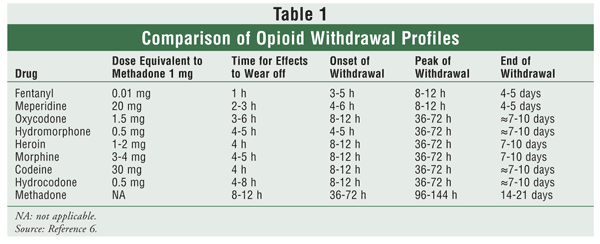
Many characteristics of opioids, especially onset of action and half-life, contribute to the potential for dependence and to the onset of withdrawal. Withdrawal may present as anxiety, bone pain, chills, piloerection, sweating, nervousness, nausea, diarrhea, rhinorrhea, or constant yawning. More severe symptoms include hot and cold flashes, increased blood pressure and pulse, mydriasis, abdominal and muscle cramps, and vomiting. Symptoms continue for 48 to 96 hours after the last dose, but may persist for weeks to months in some individuals.6 See TABLE 1 for withdrawal profiles of various opioids.
PHARMACOLOGIC TREATMENT
There are three major approaches to opioid dependence: opioid detoxification, agonist maintenance, and antagonist maintenance. Opioid detoxification, also known as medically supervised withdrawal, is utilized mainly to transition into or out of a maintenance program, over a very short period of time. In antagonist maintenance, naltrexone--an opioid antagonist like naloxone--is used. Unlike naloxone, it can be used orally because of its superior bioavailability. Unfortunately, neither opioid detoxification nor antagonist maintenance has proved to be as efficacious as agonist maintenance for producing long-term abstinence.7 However, research is being conducted using novel approaches like rapid detoxification under general anesthesia and subcutaneous naltrexone implantation.8,9
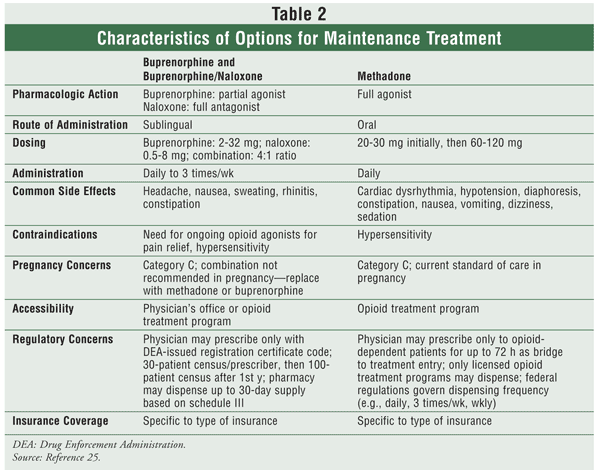
To be an effective option for maintenance treatment, an agent must be able to do the following: block the euphoric and sedating effects of dependent opioids; relieve the cravings (the major cause of relapse); relieve and prevent withdrawal symptoms; permit the patient to participate in society; and allow for at least once-daily dosing.10 The pharmacologic agents that embody these characteristics and have proven their efficacy are buprenorphine, buprenorphine/naloxone, and methadone (see TABLE 2).
Methadone and Levo-Alpha Acetyl Methadol (LAAM)
Methadone, a schedule II controlled substance, has been the most frequently used medication in opioid treatment programs. Access to methadone for the treatment of opioid dependence is available only through DEA-licensed methadone clinics. LAAM is no longer being produced or marketed because of the increased risk of cardiac death.
Clinical Pharmacology: Both methadone and LAAM are synthetic mu-opioid receptor agonists, like heroin, and serve to replace heroin or other opioids' occupation of mu-opioid receptors. Methadone also is an N-methyl-D-aspartate antagonist.
Methadone is a highly fat-soluble drug that is rapidly and extensively absorbed. The oral suspension has variable bioavailability (range 36% to 100%) and reaches its peak effects 1 to 7.5 hours after intake.11 Methadone is a 50:50 racemic mixture. The R-enantiomer has a significantly higher affinity to mu and kappa receptors. Methadone has an average half-life of 24 hours (range 13 to 50 hours). Metabolism of the drug occurs through the CYP450 system--mainly CYP3A4, but also CYP2B6, CYP2C9, CYP2C19, and CYP2D6. Methadone is an inhibitor of CYP2D6.12,13
Dosing: In an opioid-naïve patient beginning methadone maintenance, the first dose should not exceed 40 mg owing to the risk of death from respiratory depression. A common starting dose is 20 mg to 30 mg. The patient usually is monitored for 2 to 4 hours to allow the methadone to peak. After the first day, additional doses of 5 mg to 10 mg may be given if withdrawal symptoms persist. The dose may be slowly titrated over the next couple of weeks to achieve a dose that prevents withdrawal and drug cravings without oversedating or causing other side effects. Most patients can be maintained on a dose of 60 mg to 120 mg, although some patients will need doses outside this range.6
For maintenance, methadone should be dispensed to the patient as a 1-mg/mL solution. Doses of less than 50 mg/day have been associated with increased relapse rates and less retention in programs.14,15
Side Effects: Because methadone acts upon central opioid receptors, its side-effect profile mirrors that of other opioids. These effects include sweating, somnolence, dizziness, mild nausea, anorexia, constipation, and pruritus. The most serious adverse effects are respiratory depression and cardiac arrhythmias. There also have been reports of increased risk of cardiac death. Patients should be counseled regarding possible weight gain, sexual dysfunction, and oligomenorrhea or amenorrhea.10,14
Interactions: There are numerous potential drug interactions with methadone (see TABLE 3). Methadone's drug interactions occur primarily through inhibition or induction of liver enzymes and changes in protein binding.12,13 No clinically significant protein-binding drug interaction has been reported.
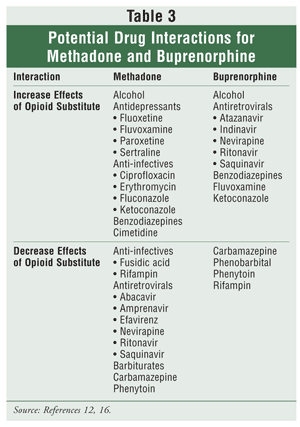
Giving methadone with opioid antagonists, mixed agonist/antagonists, and partial agonists (i.e., naloxone, naltrexone, pentazocine, nalbuphine, butorphanol, and buprenorphine) may precipitate withdrawal. Somnolence and respiratory depression may be potentiated if methadone is taken with other opioids.
Buprenorphine and Naloxone
Some of the drawbacks of methadone treatment are that the drug has increased risks of respiratory depression, death from overdose, QT prolongation, divergence, and difficult withdrawal. Buprenorphine is a partial agonist that carries fewer risks than methadone.16 Naloxone is formulated in combination with buprenorphine to decrease the abuse potential: When taken correctly sublingually, naloxone has no clinical effect because of poor bioavailability; if injected, however, naloxone precipitates withdrawal.
Pharmacology: Buprenorphine is a synthetic opioid with partial mu-opioid receptor agonism and kappa-receptor antagonism. It has a higher affinity for mu-opioid receptors; therefore, it displaces morphine, methadone, and other full agonists from the receptor site. This partial agonism allows buprenorphine to have a ceiling effect on the opioid effects at higher doses, making it safer in the event of overdose.
Naloxone is a mu-opioid receptor antagonist with poor oral bioavailability owing to limited absorption and extensive first-pass metabolism. It has a rapid onset of action when given intravenously. Because it has a higher affinity for mu-opioid receptors than heroin, morphine, or methadone, naloxone displaces these drugs from receptors and blocks their effects.17
Buprenorphine/naloxone (Suboxone) is available as a 4:1 fixed combination. Subutex (buprenorphine), a white tablet, is available in 2-mg and 8-mg strengths; Suboxone is an orange tablet and is available in 2/0.5-mg and 8/2-mg formulations. Buprenorphine is rapidly absorbed sublingually, and peak effects are reached 90 minutes after administration. Naloxone does not affect the pharmacokinetics of buprenorphine. The mean half-life of buprenorphine is 37 hours; that of naloxone is 1.1 hours.18
Dosing: Because buprenorphine displaces the other opioid from mu-receptor sites and induces withdrawal, it should be initiated only when the patient already has evident signs of withdrawal; otherwise, the patient might associate the buprenorphine with withdrawal, thereby reducing adherence. The buprenorphine/naloxone combination is the drug of choice for initiating therapy in both U.S. and European guidelines. Buprenorphine may be started alone at doses of 4 mg to 8 mg or in combination in a 4:1 ratio to naloxone. A second dose of 4 mg may be given in 2 to 4 hours, and the patient may be given an additional dose of 2 mg to 4 mg to take home in case of withdrawal within the next 24 hours. The physician should monitor for buprenorphine-precipitated withdrawal while the patient is in the office. This is not to be confused with withdrawal from underdosing of buprenorphine, which usually occurs in the second half of the 24-hour dosing interval.16
The maintenance dose may be achieved by doubling the dose each day, to a maximum of 24 mg to 32 mg. If withdrawal symptoms arise at any time during the 24-hour dosing interval, the dose is too low and needs to be increased. If induction occurs too slowly, the patient may prematurely terminate treatment. Therefore, it is important for the practitioner to be diligent in monitoring the patient. When converting to or from the naltrexone combination, a 1:1 ratio of the buprenorphine dose may be used.16
When the maintenance dose is achieved, buprenorphine may be administered from every other day to 3 times weekly (e.g., Monday, Wednesday, and Friday) in order to increase compliance and patient satisfaction. Every-4-day regimens have been associated with increased withdrawal symptoms. The daily dose may be doubled for every-other-day dosing and also for thrice-weekly dosing, but Friday's dose would be 2.5 times the daily dose.16
New patients should be advised that sublingual tablets must be dissolved under the tongue, as the medication is much less effective if swallowed.18 No more than two tablets should be taken at one time, to avoid swallowing them by mistake. Wetting the mouth before placing the tablets under the tongue may help them dissolve faster. Full absorption may take up to 10 minutes. Patients should refrain from smoking for 10 to 15 minutes before taking the medication, as this seems to help the tablets dissolve faster.18
Side Effects: Buprenorphine/naloxone is generally well tolerated. Side effects are associated mainly with buprenorphine, since naloxone is not readily absorbed. In clinical trials, the most common adverse effects were headache, withdrawal syndrome, pain, nausea, insomnia, sweating, rhinitis, constipation, abdominal pain, flulike syndrome, and flushing.17
Interactions: Buprenorphine goes through hepatic metabolism via CYP3A4. The drug has the potential for many of the same interactions as methadone (see TABLE 3). One dangerous interaction to monitor for is the potentially fatal interaction with benzodiazepines.19 Concomitant administration should be avoided. Compared with methadone, buprenorphine may be a safer choice in patients receiving antiretrovirals.20
CLINICAL EFFICACY OF METHADONE VERSUS BUPRENORPHINE
It is well established that both methadone and buprenorphine are effective for decreasing illicit drug use. It is worthwhile to consider the results of studies examining the efficacy of methadone versus buprenorphine.7
The 2008 Cochrane review determined that methadone dosed at 60 mg/day to 120 mg/day has superior efficacy compared with buprenorphine.21 The specific studies yield varied results. One study found less illicit heroin use with buprenorphine than with methadone, but the methadone arm had higher retention rates.22 Another study concluded that high-dose methadone had a higher retention rate and less illicit opioid use compared with buprenorphine 8 mg.23 A double-blind, randomized trial comparing an average dose of buprenorphine (10 mg/day) versus methadone (70 mg/day) showed a higher retention rate with methadone, but found that the drugs had equal efficacy in reducing illicit heroin use.24 However, a study from 1992 concluded that buprenorphine had better retention rates than methadone at 25 weeks.26 Overall, it is accepted that buprenorphine and methadone have comparable efficacy and that treatment should be individualized.
CONCLUSION
Because of DATA 2000 and ongoing research on opioid dependence, pharmacists must be prepared to face an increase in the number of prescriptions being written for opioid maintenance treatment. When presented with a new prescription, a pharmacist may visit the site www.buprenorphine.samhsa.gov to confirm physician eligibility. Pharmacists must monitor and counsel patients about withdrawal symptoms and overdose possibilities. Because buprenorphine is a partial agonist, the risk of overdose is smaller, and its use in combination with naloxone further reduces the risk of intravenous abuse. Historically, daily visits to methadone clinics have been the most frequently utilized method of treating opioid dependence, but with the current availability of sublingual buprenorphine products, more patients will be able to receive treatment in a convenient office-based setting.
REFERENCES
1. Substance Abuse and Mental Health Services Administration (SAMHSA). Results from the 2006 National Survey on Drug Use and Health: national findings. Figure 7.1. www.oas.samhsa.gov/NSDUH/
2. Rounsaville BJ, Kosten TR. Treatment for opioid dependence: quality and access. JAMA. 2000;283:1337-1339.
3. SAMHSA. The Drug Addiction Treatment Act of 2000 (DATA 2000). Physician waiver qualifications. http://buprenorphine.samhsa.
4. Kosten TR, George TP. The neurobiology of opioid dependence: implications for treatment. Sci Pract Perspect. 2002;16:13-20.
5. Cami J, Farré M. Drug addiction. N Engl J Med. 2003;349:975-986.
6. Galanter M, Kleber HD, eds. The American Psychiatric Publishing Textbook of Substance Abuse Treatment. 4th ed. Arlington, VA: American Psychiatric Publishing; 2008.
7. Krantz MJ, Mehler PS. Treating opioid dependence. Growing implications for primary care. Arch Intern Med. 2004;164:277-288.
8. Albanese AP, Gevirtz C, Oppenheim B, et al. Outcome and six month follow up of patients after Ultra Rapid Opiate Detoxification (UROD). J Addict Dis. 2000;19:11-28.
9. Kunoe N, Lobmaier P, Vederhus JK, et al. Naltrexone implants after in-patient treatment for opioid dependence: randomised controlled trial. Br J Psychiatry. 2009;194:541-546.
10. Centers for Disease Control and Prevention. Methadone maintenance treatment. http://cdc.gov/IDU/facts/
11. Dolophine (methadone HCl) product information. Columbus, OH: Roxane Laboratories, Inc; October 2006.
12. Brown R, Kraus C, Fleming M, Reddy S. Methadone: applied pharmacology and use as adjunctive treatment in chronic pain. Postgrad Med J. 2004;80:654-659.
13. Eap CB, Buclin T, Baumann P. Interindividual variability of the clinical pharmacokinetics of methadone: implications for the treatment of opioid dependence. Clin Pharmacokinet.
14. Strain EC, Stitzer ML, Liebson IA, Bigelow GE. Dose-response effects of methadone in the treatment of opioid dependence. Ann Intern Med. 1993;119:23-27.
15. Preston KL, Umbricht A, Epstein DH. Methadone dose increase and abstinence reinforcement for treatment of continued heroin use during methadone maintenance. Arch Gen Psychiatry. 2000;57:395-404.
16. Jones HE. Practical considerations for the clinical use of buprenorphine. Sci Pract Perspect. 2004;2:4-20.
17. Orman JS, Keating GM. Buprenorphine/naloxone: a review of its use in the treatment of opioid dependence. Drugs. 2009;69:577-607.
18. Suboxone (buprenorphine HCl and naloxone HCl)/Subutex (buprenorphine HCl) product information. Richmond, VA: Reckitt Benckiser Pharmaceuticals, Inc; September 2006.
19. Fischer G, Gombas W, Eder H, et al. Buprenorphine versus methadone maintenance for the treatment of opioid dependence. Addiction. 1999;94:1337-1347.
20. Fiellin DA, Friedland GH, Gourevitch MN. Opioid dependence: rationale for and efficacy of existing and new treatments. Clin Infect Dis. 2006;43:S173-S177.
21. Mattick RP, Kimber J, Breen C, Davoli M. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database Syst Rev. 2003;(2):CD002207.
22. Fudala PJ, Bridge TP, Herbert S, et al. Office-based treatment of opiate addiction with a sublingual-tablet formulation of buprenorphine and naloxone. N Engl J Med. 2003;349:949-958.
23. Ling W, Wesson DR, Charuvastra C, Klett CJ. A controlled trial comparing buprenorphine and methadone maintenance in opioid dependence. Arch Gen Psychiatry. 1996;53:401-407.
24. Petitjean S, Stohler R, Déglon JJ, et al. Double-blind randomized trial of buprenorphine and methadone in opiate dependence. Drug Alcohol Depend. 2001;62:97-104.
25. Sullivan LE, Fiellin DA. Narrative review: buprenorphine for opioid-dependent patients in office practice. Ann Intern Med. 2008;148:662-670.
26. Johnson RE, Jaffe JH, Fudala PJ. A controlled trial of buprenorphine treatment for opioid dependence. JAMA. 1992;267:2750-2755. 2002;41:1153-1193.






