US Pharm. 2016;41(2):17-20.
ABSTRACT: Cholesterol concentrations in many patients with familial hypercholesterolemia are poorly controlled despite dietary changes and maximum pharmacologic therapy. Increased concentrations of proprotein convertase subtilisin/kexin type 9 (PCSK9) contribute to elevated concentrations of LDL cholesterol (LDL-C) in these patients. Monoclonal antibodies, which inhibit PCSK9, have been shown to significantly reduce LDL-C in those with familial hypercholesterolemia. Two recently approved monoclonal antibodies—evolocumab and alirocumab—are injected subcutaneously either biweekly or monthly and have demonstrated favorable safety profiles, in addition to efficacy in lowering LDL-C. A third monoclonal antibody is currently being investigated. Long-term studies are necessary to determine the effectiveness of these new agents in preventing cardiovascular disease.
Cardiovascular disease (CVD) is the leading cause of death globally and accounts for approximately one-third of all deaths in the United States.1,2 High concentrations of LDL-cholesterol (LDL-C) are a well-known risk factor for the development of CVD. Statins, the current therapy of choice for lowering LDL-C, are indicated for primary and secondary prevention of cardiovascular events.3 Statins have a favorable safety profile, a relatively low cost, and the ability to lower LDL-C and CVD risk.3
Background
The American College of Cardiology/American Heart Association (ACC/AHA) and the National Lipid Association (NLA) have each published guidelines for the treatment of dyslipidemia.3,4 Both guidelines recommend that therapy initiation be based on the presence of risk factors for atherosclerotic CVD (ASCVD); however, they differ on treatment goals for LDL-C. The ACC/AHA guidelines do not recommend LDL-C treatment goals, whereas the NLA recommends an LDL-C goal of <100 mg/dL for patients with risk factors for ASCVD and an LDL-C goal of <70 mg/dL for patients with ASCVD or type 1 or 2 diabetes. More intensive lipid-lowering correlates with a proportional decrease in the risk of cardiovascular events without a significant increase in adverse events.5
Despite maximum lipid-lowering therapy with statins, cholesterol absorption inhibitors, niacin, and bile acid sequestrants, many patients still have elevated LDL-C and remain at risk for cardiovascular disease progression.6 Some of these patients may have an inherited condition, familial hypercholesterolemia, involving mutations in a variety of genes controlling concentrations of the enzyme proprotein convertase subtilisin/kexin type 9 (PCSK9). Individuals with higher PCSK9 concentrations have higher LDL-C concentrations.7 Patients with heterozygous familial hypercholesterolemia (HeFH) have a baseline LDL-C range of 200 to 400 mg/dL, whereas those with homozygous familial hypercholesterolemia (HoFH) can have LDL-C concentrations >600 mg/dL.8 With LDL-C concentrations this high, even a 50% reduction from baseline provided by a high-intensity statin may still leave patients with cardiovascular risks.
Concentrations of circulating LDL-C are partly affected by the number of LDL receptors (LDLRs) on the cell surface, as well as by PCSK9 concentrations. The greater the number of LDLRs on the cell surface, the more LDL-C is removed from circulation. As LDL-C binds the receptor, the complex is internalized, allowing the cell to use the newly obtained cholesterol. The LDLR is then recycled back to the cell surface. In contrast, increased plasma concentrations of PCSK9 lead to higher concentrations of LDL-C by interfering with LDLR recycling, which facilitates the degradation of LDLRs.9 Once PCSK9 binds the extracellular portion of the LDLR, the LDLR-PCSK9 complex is internalized and targeted for lysosomal degradation.10 As LDL-C receptors are cleared from the surface, fewer receptors are available for removing circulating LDL-C from the plasma.
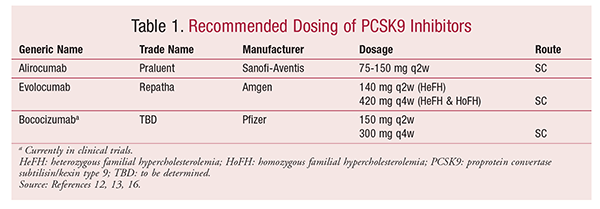
Two PCSK9 inhibitors were recently approved, and a third agent is currently being investigated. These agents are monoclonal antibodies, which target circulating PCSK9 by recognizing the binding portion of PCSK9 that normally interacts with the LDLR.9 Inhibition of PCSK9 prevents degradation of LDLRs, permitting more LDL-C to be removed from circulation.7 These monoclonal antibodies (evolocumab, alirocumab, and bococizumab) inhibit the interaction between PCSK9 and the LDLRs. The recommended dosing for all three agents is listed in TABLE 1.
Alirocumab Dosing and Adverse Effects
Praluent (alirocumab) is a fully human monoclonal antibody manufactured by Sanofi-Aventis U.S. and Regeneron Pharmaceuticals, Inc. In July 2015, the FDA granted approval for Praluent as an adjunct to diet and maximum tolerated statin therapy in patients with HeFH or those with clinical ASCVD who require additional lowering of LDL-C. The recommended starting dosage is 75 mg injected SC every 2 weeks, with the potential to increase to 150 mg every 2 weeks. Praluent is available as 75 mg/mL or 150 mg/mL solution for injection in a single-use prefilled pen or a single-use prefilled syringe.11 The reported wholesale acquisition cost (WAC) for each dosage strength is $1,120 every 28 days, or $14,600 annually.12 The most common adverse effect is injection-site reactions. Other adverse effects occurring more frequently than with placebo include coldlike and flulike symptoms. No dosage adjustments are required for patients with mild-to-moderate renal or hepatic impairment or for the geriatric population.12
Evolocumab Dosing and Adverse Effects
Repatha (evolocumab) is a fully human monoclonal antibody manufactured by Amgen Inc. In August 2015, the FDA approved Repatha for use in addition to diet and maximally tolerated statin therapy in adults with HeFH or patients with clinical ASCVD requiring additional lowering of LDL-C. This agent was also approved as an adjunct to diet and other lipid-lowering therapies in patients with HoFH. Recommended dosing for HeFH or clinical ASCVD is 140 mg injected SC every 2 weeks or 420 mg injected SC every month; for HoFH, 420 mg injected SC every month is recommended. A dosage of 420 mg every 2 weeks for HoFH is being considered by the FDA.13 Evolocumab is available as 140 mg/mL disposable prefilled syringes for single use and as a SureClick autoinjector pen, which is a single-use, disposable mechanical spring-injection device. Evolocumab is packaged with a needle cover made of natural rubber, which may be a concern in patients with latex allergies. To achieve a 420-mg dose, patients must inject three prefilled 140-mg syringes consecutively within 30 minutes. (Amgen anticipates releasing a single-dose 420 mg syringe in 2017.) The reported WAC is $542.31 for one 140-mg syringe or $14,100 per year for the 140-mg biweekly dose. The most common adverse effects occurring more frequently than with placebo were nasopharyngitis, upper respiratory tract infection, influenza, back pain, and injection-site reactions. No dosage adjustments are required in patients with mild-to-moderate renal or hepatic impairment or in the geriatric population.14
Bococizumab Dosing and Adverse Effects
The third PCSK9 inhibitor, bococizumab, is a humanized monoclonal antibody manufactured by Pfizer that is currently being investigated. Early phase I and IIa clinical trials showed LDL-C reductions of up to 75%.15 A 24-week double-blind, placebo-controlled, phase II, dosing-range study examined the effectiveness of varying dosages of bococizumab versus placebo in patients already receiving statin therapy. Bococizumab dosages of 150 mg every 2 weeks or 300 mg every 4 weeks administered SC were found to be the most effective, reducing LDL-C by 54.2 mg/dL (53.4%) and 38.3 mg/dL (44.9%), respectively, compared with 2.8 mg/dL for placebo.16 Approximately 33% of patients receiving 150 mg every 2 weeks and 34% of those receiving 300 mg every 4 weeks required dosage reductions because LDL-C concentrations fell to <25 mg/dL. Mild injection-site reactions were reported in the bococizumab group; otherwise, adverse effects were similar across control and treatment groups. In patients who experienced LDL-C concentrations <25 mg/dL, there was no increase in reported adverse effects.
Efficacy and Safety Studies
A meta-analysis by Zhang and colleagues examined rates of common adverse events and reductions in LDL-C with evolocumab and alirocumab compared with placebo or ezetimibe.17 Twenty-five studies (12 on evolocumab and 13 on alirocumab) with a total of 12,200 patients were included in this meta-analysis. Bococizumab was not included since too few studies were available.
Significantly greater reductions in LDL-C were seen in the evolocumab and alirocumab groups compared with the placebo and ezetimibe groups. At 12 weeks of follow-up, the evolocumab 140 mg biweekly dosage reduced LDL-C by 60.4% compared with placebo, and the evolocumab 420 mg monthly dosage reduced LDL-C by 54.6%. Compared with ezetimibe, the 140-mg biweekly dosage reduced LDL-C by 38.2%, and the 420-mg monthly dosage reduced LDL-C by 36.3%. Biweekly dosing of alirocumab, which varied from 50 to 150 mg, showed a mean reduction in LDL-C of 52.6% compared with placebo and 29.9% compared with ezetimibe. Monthly dosing of alirocumab, which varied from 150 to 300 mg, reduced LDL-C by 32.2% compared with placebo. Compared with placebo patients, fewer patients receiving alirocumab died (P = .04).
Safety outcomes showed that treatment-emergent adverse events did not differ significantly among evolocumab treatment groups compared with placebo or ezetimibe groups.17 Of the patients receiving evolocumab, 1.9% experienced a serious adverse event, and 1.6% of patients discontinued therapy at 12 weeks. Rates of death were similar between the evolocumab groups and the control groups. No significant differences in adverse events emerged in a comparison of the monthly 420-mg and biweekly 140-mg dosages at 12 weeks of follow-up. Pooled estimates for overall incidence of any treatment-emergent adverse events at 12 weeks’ follow-up were 52.2% for the evolocumab groups, 45.2% for the placebo groups, and 54.7% for the ezetimibe groups.
Safety outcomes in the alirocumab trials were similar in that there were no significant differences in rates of adverse events in the alirocumab, placebo, and ezetimibe groups.17 Pooled estimates for any treatment-emergent adverse events at 12, 24, or 52 weeks of follow-up were 71% for the alirocumab groups, 68.4% for the placebo groups and 70.1% for the ezetimibe groups. Of alirocumab-treated patients, 8.6% reported serious adverse events and 4.8% experienced an event that led to discontinuation of therapy.
There are reports of neurocognitive events, including confusion, amnesia, and attention disorders in individuals receiving PCSK9 inhibitors, but in two long-term studies, no significant differences were observed between treatment and placebo. In a study of evolocumab, Sabatine and colleagues followed patients for a mean of 11.1 months.18 Neurocognitive events were reported by 0.9% of patients receiving evolocumab and 0.3% of those receiving placebo.18 In another study, Robinson and colleagues followed patients who received alirocumab for 78 weeks.19 Neurocognitive events were reported by 1.2% of the treatment group and 0.5% of the placebo group.19
Conclusion
Statin therapy is the most effective option currently available for lowering LDL-C and reducing cardiovascular risk. Despite maximum tolerated statin therapy, many patients cannot obtain suitable LDL-C reduction and require further lipid-lowering pharmacologic interventions. Clinical trials have shown that, when added to statin therapy, PCSK9 inhibitors can help patients achieve an LDL-C reduction >50%, with no significant differences in reported adverse events compared with placebo. The SC route may affect compliance, and patients will need training on proper drug delivery. Some patients may not favor SC delivery, but an injection frequency of only once or twice a month and significant LDL-C reduction may be sufficient incentive to overcome the fear or discomfort of injections. The biggest disadvantage of PCSK9 inhibitors is the cost, which patients and insurers will be reluctant to pay. However, significant reductions in LDL-C are necessary in some patients with familial hypercholesterolemia, those already on maximum tolerated lipid-lowering therapy, and those who cannot tolerate statin therapy. PCSK9 inhibitors may prove to be the best option for reducing LDL-C and ASCVD risk in these patients.
REFERENCES
1. World Health Organization. Global Status Report on Noncommunicable Diseases 2014. Geneva, Switzerland: World Health Organization; 2014.
2. Mozaffarian D, Benjamin EJ, Go AS, et al. Heart disease and stroke statistics—2015 update: a report from the American Heart Association. Circulation. 2015;131:e29-e322.
3. Stone NJ, Robinson JG, Lichtenstein AH, et al. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(25 suppl 2):S1-S45.
4. Jacobson TA, Ito MK, Maki KC, et al. National Lipid Association recommendations for patient-centered management of dyslipidemia: part 1—executive summary. J Clin Lipidol. 2014;8:473-488.
5. Cholesterol Treatment Trialists’ (CTT) Collaboration; Baigent C, Blackwell L, Emberson J, et al. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. 2010;376:1670-1681.
6. Hovingh GK, Davidson MH, Kastelein JJ, O’Connor AM. Diagnosis and treatment of familial hypercholesterolaemia. Eur Heart J. 2013;34:962-971.
7. Desai NR, Sabatine MS. PCSK9 inhibition in patients with hypercholesterolemia. Trends Cardiovasc Med. 2015;25:567-574.
8. Abifadel M, Varret M, Rabès JP, et al. Mutations in PCSK9 cause autosomal dominant hypercholesterolemia. Nat Genet. 2003;34:154-156.
9. Lambert G, Sjouke B, Choque B, et al. The PCSK9 decade. J Lipid Res. 2012;53:2515-2524.
10. Stein EA, Mellis S, Yancopoulos GD, et al. Effect of a monoclonal antibody to PCSK9 on LDL cholesterol. N Engl J Med. 2012;366:1108-1118.
11. Sanofi and Regeneron announce FDA approval of Praluent® (alirocumab) injection, the first PCSK9 inhibitor in the U.S., for the treatment of high LDL cholesterol in adult patients. www.news.sanofi.us/2015-07-24-Sanofi-and-Regeneron-Announce-FDA-Approval-of-Praluent-alirocumab-Injection-the-First-PCSK9-Inhibitor-in-the-U-S-for-the-Treatment-of-High-LDL-Cholesterol-in-Adult-Patients. Accessed August 2, 2015.
12. Praluent (alirocumab) product information. Bridgewater, NJ: Sanofi-Aventis U.S. LLC; October 2015.
13. FDA approves Amgen’s new cholesterol-lowering medication Repatha™ (evolocumab). wwwext.amgen.com/media/media_pr_detail.jsp?year=2015&releaseID=2082837. Accessed September 9, 2015.
14. FDA Briefing Document. Endocrinologic and Metabolic Drugs Advisory Committee (EMDAC) Meeting: June 10, 2015. Memorandum. www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/EndocrinologicandMetabolicDrugsAdvisoryCommittee/UCM450072.pdf. Accessed June 21, 2015.
15. Dadu RT, Ballantyne CM. Lipid lowering with PCSK9 inhibitors. Nat Rev Cardiol. 2014;11:563-575.
16. Ballantyne CM, Neutel J, Cropp A, et al. Results of bococizumab, a monoclonal antibody against proprotein convertase subtilisin/kexin type 9, from a randomized, placebo-controlled, dose-ranging study in statin-treated subjects with hypercholesterolemia. Am J Cardiol. 2015;115:1212-1221.
17. Zhang XL, Zhu QQ, Zhu L, et al. Safety and efficacy of anti-PCSK9 antibodies: a meta-analysis of 25 randomized, controlled trials. BMC Med. 2015;13:123.
18. Sabatine MS, Giugliano RP, Wiviott SD, et al. Efficacy and safety of evolocumab in reducing lipids and cardiovascular events. N Engl J Med. 2015;372:1500-1509.
19. Robinson JG, Farnier M, Krempf M, et al. Efficacy and safety of alirocumab in reducing lipids and cardiovascular events. N Engl J Med. 2015;372:1489-1499.
To comment on this article, contact rdavidson@uspharmacist.com.





