US Pharm. 2016;41(3):24-26.
ABSTRACT: In July 2015, the FDA updated the label warnings on nonaspirin, nonsteroidal anti-inflammatory drugs (NSAIDs) as a result of findings presented at the joint meeting of the Arthritis Advisory Committee and Drug Safety and Risk Management Advisory Committee in February 2014. These warnings reflect new data that show NSAIDs have a higher risk of cardiovascular toxicity than previously suspected. Pharmacists need to weigh the risks and benefits of NSAID use with regard to the safety issues and the clinical implications of the newest FDA label changes to optimize patient care.
Nonsteroidal anti-inflammatory drugs (NSAIDs) are one of the most commonly used drug classes in the world.1 It is estimated that more than 30 million people use these medications on a daily basis, and they account for 60% of the analgesic market in the United States.2 There are approximately 20 different OTC and prescription NSAIDs available in the U.S. (TABLE 1).1,3 NSAIDs are used for the treatment of fever, acute or chronic pain, and inflammation caused by a variety of conditions.
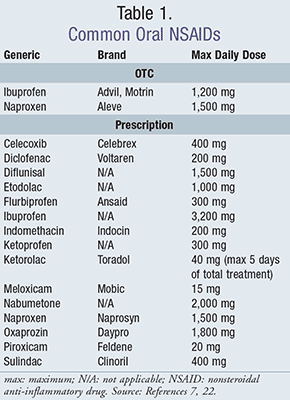
The mechanism of action of NSAIDs involves inhibition of cyclooxygenase-1 (COX-1) and/or COX-2 enzymes. COX-1 catalyzes the production of prostaglandins involved in various phys-iological functions (i.e., maintenance of renal function, mucosal protection in the gastrointestinal [GI] tract, platelet activation). COX-2 is expressed as part of the inflammatory response, resulting in vasodilation, platelet inhibition, and inhibition of smooth cell proliferation. The inhibition of COX-2 by NSAIDs plays a role in mediating pain, fever, and inflammation.2,4 Nonselective NSAIDs inhibit both COX-1 and COX-2 enzymes. Inhibition of COX-1 results in an increased risk of GI bleeding.
COX-2 selective NSAIDs were developed to maintain analgesia efficacy while minimizing the GI effects associated with COX-1 inhibition.4 Despite the potential GI benefit, COX-2 selective NSAIDs are presumed to have a higher risk of cardiovascular (CV) events.5 In 2004, a prospective analysis was performed to evaluate adverse drug reactions as a cause of hospital admission in patients >16 years of age.6 NSAIDs accounted for 29% of the cases leading to hospital admission. The adverse events observed included GI bleeds, peptic ulceration, hemorrhagic cerebrovascular accidents, and renal impairment.6 Although NSAIDs are effective, widespread use presents risks that can increase with long-term use and higher doses. In response to these risks, the FDA recently updated previous warnings regarding NSAID use.
FDA Warnings and Safety Concerns
In 2005, the FDA mandated that all prescription NSAIDs include a boxed warning and Medication Guide to inform patients of an increased risk of CV events and GI bleeding.7 In addition, manufacturers of OTC NSAIDs were asked to revise their labels to include more specific information about the potential CV and GI risks and strengthen reminders about dose limits and duration of NSAID treatment. This decision came after the joint meeting of the FDA’s Arthritis Advisory Committee (AAC) and Drug Safety and Risk Management Advisory Committee (DSaRM). At this meeting, the risk-benefit profile for NSAIDs was evaluated and it was concluded that Pfizer’s COX-2 selective NSAID, Bextra (valdecoxib), had an unfavorable overall risk compared to its benefit profile. Due to lack of data on its CV safety after long-term use, the FDA requested that Pfizer withdraw Bextra from the market.7 As a result, there was also a voluntary withdrawal of Vioxx (rofecoxib) and the addition of a boxed warning for Celebrex (celecoxib), now the only FDA-approved COX-2 inhibitor available.7
In 2015, the FDA strengthened the warnings regarding CV risk with nonaspirin NSAID use based on data collected over the last 10 years.8-10 The increased risk of thrombotic events, including stroke and myocardial infarction, in some studies was estimated to be 10% to >50%, depending on the specific NSAID and dose used. After evaluation of observational studies and a combined analysis of clinical trials, the FDA has determined that CV risk is more serious than initially determined in 2005. Mandatory label changes to both prescription and OTC NSAIDs will reflect the most recent safety information discussed at the AAC and DSaRM meeting in 2014.8-10
According to the FDA, NSAID labels will now be required to state that the risk of heart attack or stroke can occur as early as the first weeks of using an NSAID, and that the risk may increase with higher doses and longer use of the NSAID. Additionally, the label must address that, although patients with heart disease are at an increased risk of heart attack or stroke with NSAID use, NSAIDs can increase the risk of these events in patients without heart disease or risk factors for heart disease.9 Lastly, the label is also required to inform patients that there is an increased risk of heart failure with NSAID use.10,11
An estimated 10% to 40% of people aged ≥65 years use prescription or OTC NSAIDs daily to treat pain and inflammation.2 Patients in this population are at an increased risk of adverse events associated with NSAID use (e.g., CV, GI, renal, and hepatic).11 Because of the physiological changes that occur with age (e.g., renal elimination, cardiac output, protein binding, drug distribution/clearance), elderly patients are more susceptible to NSAID-associated adverse effects.12 Additionally, multiple comorbid conditions and polypharmacy pose a concern in the elderly. Dangerous drug interactions with NSAIDs may occur when used concomitantly with anticoagulants, diuretics, antihypertensives, and hypoglycemic drugs.12
CV Risk
In the past decade, CV risk associated with NSAID use has been extensively reviewed.13 A large-scale meta-analysis by Bhala et al found that compared to placebo, the risk of major vascular events was increased by 33% in patients taking a COX-2 selective agent or diclofenac.10 Additionally, the risk of heart failure was doubled by both nonselective and COX-2 selective NSAIDs.10
The mechanism of CV toxicity associated with NSAIDs is due to the inhibition of the cardioprotective prostaglandin PGI2, which is generated by COX-2.14 COX-2 selective agents have a higher risk of CV toxicity as compared to nonselective agents. Naproxen, a nonselective NSAID, differs slightly from other NSAIDs because of its potent COX-1 inhibition and long half-life. Therefore, it is reasonable to believe that naproxen has a better CV safety profile.14
GI Risk
The use of NSAIDs increases the risk of GI toxicity, such as the development of peptic ulcer disease (PUD), upper GI hemorrhage, or GI perforation. The risk of GI complications may vary among NSAIDs. About 25% of patients who use NSAIDs chronically will develop PUD.15 A meta-analysis by Castellsague et al pooled relative risks (RRs) of upper GI complications associated with individual NSAID use.16 Agents such as celecoxib and ibuprofen were shown to have a low RR (1.5 and 1.8, respectively), while piroxicam and ketorolac have a higher RR (7.4 and 11.5, respectively).16
All nonselective NSAIDs inhibit platelet aggregation through the inhibition of COX-1 and the thromboxane A2 (TXA2) pathway. COX-2 selective inhibitors do not affect the TXA2 pathway and therefore have minimal antiplatelet effects, minimizing the risk for GI bleed. Because prostaglandin-mediated GI protection occurs through the COX-1 enzyme, inhibiting the COX-2 enzyme alone provides anti-inflammatory properties without losing the GI protective properties of COX-1.17
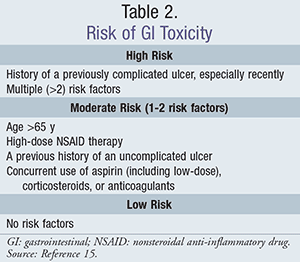
While the NSAID itself plays a role in the risk that a patient will develop a GI complication, there are other patient-determined factors that can increase the risk. According to guidelines from the American Journal of Gastroenterology, patients requiring NSAID therapy who are at high risk (TABLE 2) should receive alternative therapy.15 However, if anti-inflammatory treatment is absolutely necessary, a COX-2 inhibitor combined with misoprostol, a prostaglandin analogue, or a high-dose proton pump inhibitor (PPI) is recommended. Patients at moderate GI risk may be treated with a COX-2 inhibitor alone or with a traditional nonselective NSAID plus misoprostol or a PPI.15 It is important to also consider the risk of CV side effects of COX-2 inhibitors when initiating or evaluating a patient on an NSAID who is at risk for GI toxicity as previously stated.
Renal Risk
Chronic NSAID use can lead to severe kidney impairment due to its direct and indirect effects on the organ. The use of NSAIDs can increase blood pressure (thus making antihypertensive drugs less effective), cause fluid retention, and decrease kidney function in patients with kidney disease. Other agents such as acetaminophen, tramadol, or opioids used short-term may be safer alternatives and as effective as NSAIDs in the management of pain.18
Hsu et al determined that patients with hypertension who used NSAIDS for ≥90 days (n = 10,589) had a 32% increased risk of chronic kidney disease (CKD).19 The study also found that hypertensive patients taking higher doses of NSAIDs have a greater risk of developing CKD than patients on lower doses.19
NSAIDs may attenuate the effect of other antihypertensive agents and increase the risk of elevated serum creatinine and potassium levels when combined with other agents, such as aldosterone antagonists. Additionally, through the inhibition of COX-1 and COX-2, NSAIDs have an effect on kidney function by decreasing renal perfusion, which can lead to changes in renal blood flow. This can, in turn, increase blood pressure and worsen edema in patients who are being treated for hypertension.19 Patients who are on these therapies or have CKD and are taking NSAIDs should have their serum creatinine and potassium checked routinely to avoid further damage.20 Physicians may not be aware that patients are using OTC NSAIDs; therefore, they may not be monitoring patients routinely.
Hepatotoxicity Risk
About 10% of total drug-induced hepatotoxicity is NSAID-related.21 A review of NSAID-induced hepatotoxicity by Bessone concluded that ibuprofen has the best liver safety profile out of all prescription and OTC NSAIDs, while sulindac has shown the highest risk of severe liver damage.21 Although limited data exist, evidence does indicate that most NSAIDS have low hepatotoxic risks.21
Role of the Pharmacist
NSAIDs, both prescription and OTC, are widely used by a variety of patient populations. As one of the most trusted and accessible healthcare professionals, pharmacists are in a unique position to both identify and prevent adverse events associated with NSAID use.
Every patient using either OTC or prescription NSAIDS should be screened for all of the potential risks previously mentioned. Patients should be cautioned to check with the pharmacy before purchasing OTC products, especially combination products that may contain NSAIDs. After conducting a thorough patient interview, pharmacist should be prepared to offer alternative, evidence-based therapies to patients at high risk of adverse effects associated with NSAIDs. NSAIDs should be avoided in patients with a high risk for both CV and GI events. Patients should be educated that NSAIDs must be used at the lowest possible dose for the shortest duration possible to avoid adverse effects.
Pharmacists should encourage patients to use one pharmacy to fill all prescriptions and have a comprehensive medication card whenever starting a new prescription or OTC drug, so careful monitoring can take place for drug-drug and drug-disease interactions. Open communication between patients, prescribers, and pharmacists is key for prevention and early detection of NSAID-associated adverse events.
REFERENCES
1. Holubek WJ. Chapter 37: Nonsteroidal anti-inflammatory drugs. In: Hoffman RS, Howland M, Lewin NA, et al, eds. Goldfrank’s Toxicologic Emergencies. 10th ed. New York, NY: McGraw-Hill; 2015.
2. Conaghan PG. A turbulent decade for NSAIDs: update on current concepts of classification, epidemiology, comparative efficacy, and toxicity. Rheumatol Int. 2011;32(6):1491-1502.
3. Soloman DH. NSAIDs: mechanism of action. UptoDate. 2015. www.uptodate.com/contents/nsaids-mechanism-of-action. Accessed October 23, 2015.
4. Zhou Y, Boudreau DM, Freedman AN. Trends in the use of aspirin and nonsteroidal anti-inflammatory drugs in the general U.S. population. Pharmacoepidemiol Drug Saf. 2013;23(1):43-50.
5. Fosbol EL, Folke F, Jacobsen S, et al. Cause-specific cardiovascular risk associated with nonsteroidal anti-inflammatory drugs among healthy individuals. Circ Cardiovasc Qual Outcomes. 2010;3(4):395-405.
6. Pirmohamed M, James S, Meakin S, et al. Adverse drug reactions as cause of admission to hospital: prospective analysis of 18,820 patients. BMJ. 2004;329(7456):15-19.
7. FDA Public Health Advisory—FDA announces important changes and additional warnings for COX-2 selective and non-selective non-steroidal anti-inflammatory drugs (NSAIDs). April 7, 2005. www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm150314.htm. Accessed December 2, 2015.
8. FDA Drug Safety Communication: FDA strengthens warning that non-aspirin nonsteroidal anti-inflammatory drugs (NSAIDs) can cause heart attacks or strokes. July 9, 2015. www.fda.gov/Drugs/DrugSafety/ucm451800.htm. Accessed December 2, 2015.
9. FDA Briefing Document. Joint meeting of the Arthritis Advisory Committee and Drug Safety and Risk Management Advisory Committee. February 10-11, 2014. Nonsteroidal anti-inflammatory drugs and cardiovascular thrombotic risk. www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/ArthritisAdvisoryCommittee/UCM383180.pdf. Accessed October 28, 2015.
10. Bhala N, Emberson J, Merhi A, et al; Coxib and traditional NSAID Trialists’ (CNT) Collaboration. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: meta-analyses of individual participant data from randomised trials. Lancet. 2013;382:769-779.
11. Smith S.G. Dangers of nonsteroidal anti-inflammatory drugs in the elderly. Can Fam Physician. 1989;35:653-654.
12. Durrance SA. Older adults and NSAIDS. Geriatr Nurs. 2003;24:6. Medscape. www.medscape.com/viewarticle/466796_3. Accessed November 22, 2015.
13. Bello AE, Holt RJ. Cardiovascular risk with nonsteroidal anti-inflammatory drugs: clinical implications. Drug Saf. 2014; 37(11):897-902.
14. Patrignani P, Tacconelli S, Bruno A, et al. Managing the adverse effects of nonsteroidal anti-inflammatory drugs. Expert Rev Clin Pharmocol. 2011;4(5):605-621.
15. Lanza FL, Chan FK, Quigley EM; Practice Parameters Committee of the American College of Gastroenterology. Prevention of NSAID-related ulcer complications. Am J Gastroenterol. 2009;104:728-738.
16. Castellsague J, Riera-Guardia N, Calingaert B, et al. Individual NSAIDs and upper gastrointestinal complications: a systematic review and meta-analysis of observational studies (the SOS Project). Drug Saf. 2012;35(12):1127-1146.
17. Risser A, Donovan D, Heintzman J, Page T. NSAID prescribing precautions. Am Fam Physician. 2009;80(12):1371-1378.
18. American Society of Nephrology. Five things physicians and patients should question: 3. Avoid nonsteroidal anti-inflammatory drugs (NSAIDS) in individuals with hypertension or heart failure or CKD of all causes, including diabetes. Choosing Wisely. April 4, 2012. www.choosingwisely.org/societies/american-society-of-nephrology. Accessed October 17, 2015.
19. Hsu CC, Wang H, Hsu YH, et al. Use of nonsteroidal anti-inflammatory drugs and risk of chronic kidney disease in subjects with hypertension: Nationwide Longitudinal Cohort Study. Hypertension. 2015;66:524-533.
20. Fournier JP, Lapeyre-Mestre M, Sommet A, et al. Laboratory monitoring of patients treated with antihypertensive drugs and newly exposed to nonsteroidal anti-inflammatory drugs: a cohort study. PLoS One. 2012;7(3):e34187.
21. Bessone F. Non-steroidal anti-inflammatory drugs: what is the actual risk of liver damage? World J Gastroenterol. 2010;16(45):5651-5661.
22. NSAIDs. Lexi-Comp Online. http://online.lexi.com. Hudson, Ohio: Lexi-Comp, Inc. Accessed December 2, 2015.
To comment on this article, contact rdavidson@uspharmacist.com.





