US Pharm. 2022;47(7):6-12.
ABSTRACT: Since the start of the coronavirus disease 2019 (COVID-19) pandemic caused by severe acute respiratory syndrome-associated coronavirus-2, concerns about immunity and boosting the immune system have increased the demand for nutritional supplementation. Supplementation with vitamins C and D, zinc, quercetin, and melatonin has been found to be beneficial against infectious diseases by regulating the immune system. Adequate intake of these nutrients is particularly important for elderly patients, who are more vulnerable to the deleterious effects of COVID-19. Increased supplementation with these nutrients during COVID-19 infection may be advantageous, but toxicity should be addressed in order to ensure patient safety. Additional clinical trials should be conducted to further assess the potential of nutritional supplementation as a readily accessible and affordable treatment specifically for COVID-19.
Since the beginning of the coronavirus disease 2019 (COVID-19) pandemic, the public’s concerns about immunity and consequently heightened interest in boosting the immune system have led to an increased demand for nutritional supplementation.1 Nutritional status and supplementation are crucial for optimal prognosis in patients with SARS-associated coronavirus-2 (SARS-CoV-2), which is the cause of COVID-19 infection; these factors can also influence the clinical severity of COVID-19. Adequate intake of these nutrients is particularly important for elderly patients, who are more vulnerable to the illness and mortality associated with COVID-19.2
Various studies have examined the utility of vitamins A, B, C, D, E, and K, as well as zinc, echinacea, elderberry, melatonin, potassium, quercetin, and calcium, for managing COVID-19. Supplementation with vitamins C and D, quercetin, zinc, and melatonin has been reported to be potentially beneficial for infectious diseases, as these nutrients help regulate the immune system and may be useful in COVID-19 management as preventive therapy against lung infection.3 Proper nutrition is critically important for a strong immune system. Supplementation should be investigated further as the search for options for COVID-19 prevention and treatment continues. This review discusses current research on nutritional supplementation and the role of vitamins C and D, quercetin, zinc, and melatonin in treating patients with COVID-19.
PATHOPHYSIOLOGY
Nutrition is a determining factor for maintaining homeostasis within the body. The properties of vitamins D and C, quercetin, zinc, and melatonin with regard to COVID-19 are discussed in the following sections.
Vitamins
Vitamin D: Occurring in foods such as fish, fortified milk, red meat, and egg yolks, vitamin D is involved in a variety of bodily functions, including innate and adaptive immunologic responses; it also helps regulate blood sugar, the heart and blood vessels, and the lungs and airways.4 More specifically, vitamin D protects the lungs and airways via the antimicrobial peptide cathelicidin, which has antibacterial and antiviral activity. Cathelicidin aids in the recruitment of monocytes, neutrophils, and T cells, promoting clearance of respiratory pathogens.5 Because COVID-19 affects this pathway, vitamin D administration in patients with COVID-19 may promote recovery or result in less severe disease.6
Vitamin C: Vitamin C is found in a variety of foods, including citrus fruits, tomatoes, potatoes, and bell peppers. It protects epithelial-barrier function against infections and enhances the skin’s oxidant-scavenging capacity, potentially defending against environment-related oxidative stress. Vitamin C exerts its antiviral properties by supporting lymphocytic activity, enhancing interferon-alpha production, regulating cytokines, lowering inflammation, improving endothelial dysfunction, and restoring mitochondrial function.7,8 It has been suggested that vitamin C can counteract the cytokine storm, a life-threatening systemic inflammatory syndrome of various infectious and noninfectious diseases that can occur in patients with advanced COVID-19 infection.8
Other Nutrients
Quercetin: Quercetin is a plant flavonoid found in various vegetables (e.g., kale and other leafy greens, cherry tomatoes, broccoli, and onions), seeds, grains, tea, and wine. Its properties of interest include antioxidant and anti-inflammatory activity, immune-system regulation, and thrombin inhibition.9 Quercetin may serve as a potent nuclear factor erythroid 2–related factor 2 (NRF2) agonist to inhibit SARS-CoV-2 entry into and replication in lung cells. NRF2 helps regulate antioxidant proteins that protect against oxidative damage triggered by inflammation. Quercetin also inhibits proinflammatory pathways activated by nuclear factor kappa light chain enhancer of activated B cells, inflammasome, and interleukin-6 signals. These pathways elicit cytokine release syndrome, which promotes acute respiratory distress syndrome (ARDS), an often-fatal condition involving fluid buildup in the lungs and low blood oxygen that can occur in COVID-19. Finally, quercetin regulates thrombus-formation abnormalities by inhibiting plasma protein disulfide isomerase, which is secreted by platelets and endothelial cells on vascular injury.9 These properties make quercetin an excellent candidate for prevention and management of COVID-19, given the involvement of oxidative stress, inflammation, and the immune system.
Zinc: Zinc is an essential trace mineral involved in both innate and acquired responses to viral infection. This nutrient is found in meat, shellfish, legumes, and nuts. Zinc is important for neutrophil granulocyte recruitment and chemotactic activity, as well as for natural killer cells, phagocytosis, the formation of oxidative burst, and CD4+ and CD8+ T cells. Zinc has been shown to inhibit the synthesis, replication, and transcription complex of coronaviruses. A deficiency results in an increase in proinflammatory cytokines and lung-tissue remodeling, which can be somewhat offset if zinc supplements are taken.10 Zinc alters cell-barrier function in lung epithelial tissues via upregulation of interferon gamma, tumor necrosis factor alpha, and Fas receptor signaling as well as in vitro apoptosis. It can also interact significantly with viral replication and protein synthesis, resulting in antiviral benefits and therapeutic effects.11
Melatonin: This naturally occurring tryptophan derivative, which is generated in the pineal gland and immune cells, is recognized for its ability to boost sleep and lessen anxiety, benefiting the immune system.12 Foods containing melatonin include tart cherries, goji berries, warm milk, eggs, fish, and nuts. Melatonin is a potent antioxidant and immune regulator. It suppresses oxidative stress as a powerful hydroxyl radical scavenger and stimulator of antioxidative enzymes such as glutathione peroxidase and superoxide dismutase, protecting against cellular oxidative damage. It has been proposed as a treatment option for COVID-19 symptoms.12 Melatonin also inhibits proinflammatory activity, thereby reducing lung inflammation, and lowers the risk of pulmonary fibrosis, which is a major concern for COVID-19 patients.
ETIOLOGY AND EPIDEMIOLOGY
Over the past two decades, various viral epidemics have occurred, including SARS-CoV-1 in 2003, H1N1 influenza (swine flu) in 2009, and the Middle East respiratory syndrome coronavirus in 2012. In late 2019, a new coronavirus caused by SARS-CoV-2 was identified as the source of a cluster of pneumonia cases in Wuhan, China. This virus, later named COVID-19 by the World Health Organization (WHO), spread quickly throughout China and snowballed into a global pandemic with rapidly rising numbers of cases. As of June 19, 2022, more than 536 million confirmed cases of COVID-19 and more than 6.3 million deaths worldwide have been documented by the WHO.13
CLINICAL PRESENTATION
Clinical manifestations in persons infected with SARS-CoV-2 range from asymptomatic to severe illness. The incubation period of COVID-19, known as the presymptomatic stage, occurs 5 to 6 days after exposure to the virus.14 After a person contracts SARS-CoV-2, viral replication occurs within the host cells. As the viral load increases, symptoms develop. During this stage, the individual is contagious and can transmit the virus. The chief symptoms at COVID-19 onset, including fever, dry cough, dyspnea, headaches, new loss of taste or smell, sore throat, congestion/runny nose, nausea/vomiting, and diarrhea, range from mild to severe and are progressive. Patients with COVID-19 are considered to have severe illness if they have an oxygen saturation <94% on room air at sea level, partial pressure of oxygen/fraction of inspired oxygen <300 mmHg, a respiratory rate >30 breaths/minute, or lung infiltrates >50%. These patients may experience rapid clinical deterioration and develop pneumonia, dyspnea, myalgia, and fatigue after 7 days. These clinical conditions may cause septic shock, ARDS, hypoxemia, metabolic acidosis, and coagulation dysfunction, which can lead to death.14
DIAGNOSIS
Various tests are available to check for an active or previous COVID infection. A viral test determines whether an individual is currently infected. Antigen and nucleic acid amplification tests are two forms of viral tests that may be employed.15 A negative result indicates that SARS-CoV-2 was not detected; a positive result indicates the detection of SARS-CoV-2 and likely COVID-19. However, it is possible for a test to yield an inaccurate result. Antigen tests have lower sensitivity compared with reverse transcription-polymerase chain reaction (RT-PCR) testing. Negative antigen test results in a symptomatic patient should be followed with an RT-PCR test, as a false-negative result could lead to delays in diagnosis, isolation, and treatment.16 Although antibodies remain in the body approximately 3 to 6 months after COVID-19 infection, serologic test results should not be considered a determinant of immunity. Also, serologic test findings should not be used to avoid prophylactic actions such as social distancing or mask wearing.
PREVENTION AND MANAGEMENT
The search for preventive and therapeutic antiviral therapies is especially relevant and urgent with regard to SARS-CoV-2 and COVID-19. Current pharmacotherapy for COVID-19 includes the FDA-approved drug remdesivir (Veklury), dexamethasone (Decadron), and SARS-CoV-2–targeting monoclonal antibodies (e.g., bebtelovimab, molnupiravir, and sotrovimab) under Emergency Use Authorization (EUA) in the United States.17
Three types of COVID-19 vaccines are available in the U.S.: protein-based, viral vector, and messenger RNA (mRNA). Protein-based vaccines use harmless components of a virus that are most effective at stimulating the immune system; the immune system identifies the spike protein (S protein) and produces antibodies to combat COVID-19 after infection. In viral vector vaccines, genetic material from a virus is inserted into a modified version of a separate virus or vector; the viral vector enters the cell and delivers genetic information instructing cells to create copies of the S protein, creating an immune response by producing antibodies. The mRNA vaccine consists of an mRNA-encoding targeting gene in which mRNA is translated into a target protein and triggers an immunologic response. Pfizer-BioNTech’s two-dose COVID-19 mRNA vaccine and Moderna’s two-dose COVID-19 mRNA vaccine are FDA approved for persons aged 16 years and older and were granted EUA for those aged 5 years and older; Janssen’s one-dose adenoviral vector vaccine was granted EUA for those aged 18 years and older.17 The CDC has recommended immunization with any of these vaccines based on their effectiveness and the resultant substantially reduced risk of COVID-19, especially severe and critical cases.
In addition to these medications, research has shown that supplementation with vitamins C and D, quercetin, zinc, and melatonin may be beneficial as preventive and adjunctive therapy. As noted in TABLE 1, the recommended daily intake of these nutrients can be obtained from a broad variety of foods.
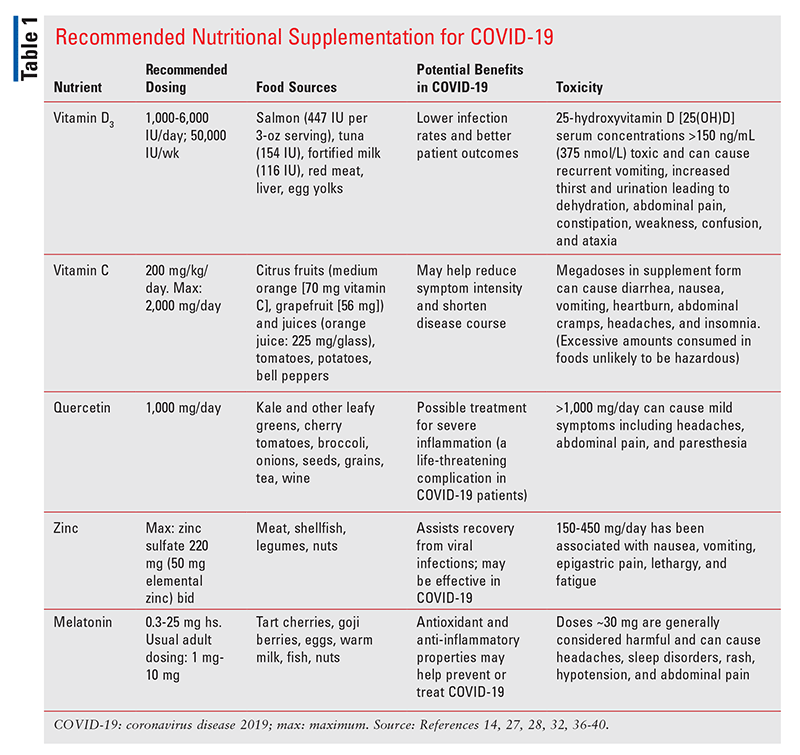
NUTRITIONAL SUPPLEMENTATION
Vitamin D3
Vitamin D (cholecalciferol) supplementation has been shown to reduce the incidence and severity of viral illness, suggesting an inverse connection between serum 25-hydroxyvitamin D levels and upper respiratory tract infection.18 In some clinical trials, vitamin D insufficiency or low levels of vitamin D were frequently observed in patients hospitalized with severe COVID-19 who had an increased mortality risk.19 Vitamin D insufficiency has been linked to poorer results in patients with the following characteristics: advanced age, male sex, obesity, hypertension, coagulopathy, and living in a northern climate.
Clinical trials have been conducted to investigate the effectiveness of vitamin D for improving immunity against COVID-19 in order to minimize patient mortality. A randomized, open-label clinical trial in Spain assessed 76 patients aged 18 years and older who were hospitalized with COVID-19 and receiving standard care (hydroxychloroquine and azithromycin); of these, 50 patients were given calcifediol (vitamin D3) treatment and the remaining 26 patients received placebo.20 Only 2% of calcifediol patients required ICU admission, compared with 50% of placebo patients (P <.001). This study highlights the potential for high-dose vitamin D supplementation to reduce disease severity in hospitalized patients with clinical acute respiratory infection.
Vitamin D deficiency may increase the risk of community-acquired pneumonia (CAP). In a meta-analysis involving more than 20,000 patients from eight observational studies, CAP patients with vitamin D deficiency—serum 25(OH)D levels <20 ng/mL—had an increased risk of CAP (odds ratio [OR], 1.64; 95% CI, 1.00-2.67).21 This result points to an association between vitamin D deficiency and increased risk of CAP.21 Although this meta-analysis was conducted before the advent of COVID-19, the findings may apply to patients with COVID-19 pneumonia, given that similarities include dyspnea, fever, cough, fatigue, and chills.
A recent retrospective study assessed 1,176 patients with a baseline 25(OH)D level collected from medical records within the previous 2 years who had two positive PCR tests for COVID-19.22 The cosinor model was used to normalize vitamin D levels for all patients to account for seasonal variations. Patients were then grouped based on sex, ethnicity, BMI, and COVID-19 severity (mild, moderate, severe, or critical). Patients with severe or critical disease (<20 ng/mL [87.4%]) had significantly lower vitamin D levels (P <.001) than those with mild or moderate disease (<20 ng/mL [34.3%]). Patients with vitamin D deficiency (<20 ng/mL) were 14 times more likely than those with vitamin D levels 40 ng/mL or higher to develop severe or critical disease (OR, 14; 95% CI, 4-51; P <.001). The study demonstrated an association between preinfection vitamin D deficiency and poorer COVID-19 outcomes, particularly in patients older than 50 years.22
The COvid-19 and high-dose VITamin D trial (COVIT-TRIAL), conducted from April 2020 to early 2021, was the first randomized, controlled study to investigate the effect of vitamin D supplementation on COVID-19 prognosis in high-risk elderly adults.23 High-dose vitamin D cholecalciferol supplementation (two 200,000-IU drinking vials once, or one 50,000-IU drinking vial) was examined in 260 participants aged 65 years and older who had COVID-19. The researchers concluded that high-dose vitamin D could be a readily accessible and well-tolerated therapy for COVID-19.23 Although the effectiveness of vitamin D against SARS-CoV-2 infection has not been demonstrated conclusively, vitamin D supplementation could potentially reduce proinflammatory cytokines and subsequently limit ARDS-associated mortality in patients with COVID-19 infection.
Vitamin C
The immunomodulatory properties of vitamin C (ascorbic acid) make it a potential therapeutic option for COVID-19 infection, prevention, treatment, and adjuvant therapy in critical care.24 Plasma vitamin C levels are reduced during acute respiratory infections such as pneumonia or tuberculosis. Supplemental vitamin C could help reduce the severity and duration of pneumonia, rendering this nutrient an area of interest for COVID-19. As noted earlier, it is believed that vitamin C can counteract the cytokine storm in COVID-19 patients.8
A 2019 meta-analysis of 12 controlled trials examined vitamin C supplementation in 1,766 patients with systematic inflammatory response including sepsis and critically ill patients requiring ventilation.25 Supplemental vitamin C reduced ICU stay by a mean of 8% (P ≤.003) and duration of mechanical ventilation by 18.2% (P = .001). In six trials, patients were administered oral vitamin C in dosages of 1 g to 3 g/day (mean 2 g/day), and other trials used IV infusions of 0.025 g/kg to 0.2 g/kg for 4 to 5 days.25 The researchers found that vitamin C significantly reduced the length of ICU stay and concluded that further research should be conducted. In a systematic review in 2021, 1 g to 2 g/day of vitamin C demonstrated efficacy both in C-reactive protein (CRP) and endothelial function, which are inflammatory markers in infectious diseases such as COVID-19.26 Although there is no clear evidence that vitamin C is effective against COVID-19, the reported benefits of vitamin C in the ICU suggest the need for further investigation into its role in boosting innate immune response.
Quercetin
Quercetin may be therapeutic for COVID-19 when it is used early in the course of infection. A randomized, controlled, open-label trial conducted in 2021 investigated the adjuvant effect of quercetin in the treatment of early symptoms and prevention of severe outcomes in 152 outpatients with COVID-19.27 All patients received standard care (analgesics/antipyretics, antibiotics, and steroids), and 76 of them also took quercetin 1,000 mg daily for 30 days. Quercetin treatment resulted in reductions in the frequency and length of hospitalization, need for noninvasive oxygen therapy, progression to ICU, and number of deaths. Hospitalization occurred in 22 (28.9%) standard-care patients (mean length of stay, 6.77 ± 3.08 days) and 7 (9.2%) quercetin patients (average length of stay, 1.57 ± 0.53 days). Fifteen standard-care patients (19.7%) and one quercetin patient (1.3%) needed noninvasive oxygen therapy. All of these results demonstrated highly significant between-group differences. Eight standard-care patients (10.5%) progressed to the ICU and three of them died, whereas no patients in the quercetin group progressed to the ICU, and none died.27 The ICU-progression rates and mortality rates in the standard-care group indicate significant detrimental outcomes compared with quercetin supplementation.
Quercetin also inhibits reverse transcriptase enzymes, which may disrupt viral replication. In a randomized, open-label, clinical trial published in 2021, the therapeutic efficacy of quercetin in combination with remdesivir and favipiravir was assessed in 60 hospitalized patients with severe COVID-19.28 The intervention group (n = 30) was treated with antiviral therapy plus quercetin 1,000 mg daily for 7 days; the control group received antiviral therapy plus placebo for 7 days. Compared with controls, intervention patients demonstrated reductions in alkaline phosphatase, CRP, and lactate dehydrogenase as well as significant increases in hemoglobin and respiratory rate; they also had an earlier hospital discharge.28 Given its anti-inflammatory and thrombin-inhibiting properties, quercetin should be considered for COVID-19 treatment based on the patient’s clinical symptoms.
Zinc
Zinc may provide a benefit against COVID-19 infection.29 A systematic review from 2020 concluded that adequate zinc status appeared to have a protective effect as supportive therapy for COVID-19 by reducing lung inflammation, improving mucociliary clearance, preventing ventilator-induced lung injury, and modulating antibacterial and antiviral immunity, particularly in elderly patients.29,30
In a randomized, double-blind, placebo-controlled trial, administration of either zinc sulfate 220 mg or placebo for 42 days in 55 zinc-deficient patients resulted in increased serum zinc concentrations.31 The control group’s serum CRP concentrations increased over time but did not achieve statistical significance. The zinc-supplemented group had a gradual but nonsignificant drop in serum CRP concentrations from the beginning to the end of the study period (13.5 ± 3.8 mg/L standard error of mean [SEM] vs. 10.5 ± 3.5 mg/L SEM, respectively).31 Zinc has antioxidant, anti-inflammatory, immunomodulatory, and antiviral activities, most of which are considered to be mediated by its capacity to suppress RNA viruses, thereby protecting cells and tissues against viral infections, oxidative damage, and malfunction.32 Based on current knowledge of this mineral’s beneficial and well-tolerated properties, its benefit-to-risk ratio supports zinc supplementation for COVID-19.
Melatonin
Melatonin may be useful for lessening the likelihood of SARS-CoV-2 entry into cells. Benefits of its use include the reduction of uncontrolled hyperinflammation and immune cell activation, decreased tissue damage and multiorgan failure caused by free radicals, and diminished ventilator-induced lung injury and disability caused by fibrotic changes in the lungs.33
Melatonin’s antioxidant and anti-inflammatory properties may help prevent or aid in the treatment of COVID-19. A randomized, double-blind clinical trial examined the efficacy of a 3-mg dose of melatonin as adjunctive therapy in 74 hospitalized patients with mild-to-moderate COVID-19.34 Patients received either standard care (determined by the treating physicians) or standard care plus melatonin 3 mg three times daily for 14 days, with 24 intervention patients and 20 controls completing treatment. Patients in the intervention group demonstrated a significant improvement (P <.05) in CRP levels as well as improvements in clinical symptoms including cough, dyspnea, and fatigue. The mean time to hospital discharge was 4.65 ± 3.37 days in the intervention group versus 8.15 ± 5.97 days in the control group, resulting in a significantly shorter stay in intervention patients compared with controls (P <.021).34 In a matching observational study of 26,779 subjects from a COVID-19 registry, those who took melatonin had a 28% lower risk of contracting COVID-19, and the infection risk was lowered by 52% in African American patients, a population that is disproportionately impacted by the disease.33
Although melatonin may have anti-inflammatory benefits for the treatment of COVID-19, more research on its effects is needed. However, based on its strong safety profile and potential anti–SARS-CoV-2 properties, it is preferred for treating sleep disorders in COVID-19 patients, and clinical trials currently support its use as a preventive and adjunctive therapy.35
CONCLUSION
Supplementation with vitamins D and C, zinc, quercetin, and melatonin has antiviral effects, may boost immune response, and may reduce viral replication. These nutrients are necessary for a normally functioning immune system, and most can be obtained through adequate dietary intake. That said, the potential of nutritional supplementation as a readily accessible and affordable treatment specifically for COVID-19 needs to be further evaluated in clinical trials. A patient’s levels of vitamins, minerals, and other nutrients must be sufficient during the course of COVID-19 infection to lower the symptom load and shorten the duration of respiratory infection.
Increased dietary intake of vitamins D and C, quercetin, and zinc, as well as melatonin for sleep, may help boost immunologic function during COVID-19 infection. Much of the research on the use of these nutrients in COVID-19 patients used levels that are too high to be obtained exclusively from the diet. Supplementation with higher doses of these nutrients during COVID-19 infection has yielded favorable results, and given their low-risk profile, they are a sensible addition to patient care. However, further research is needed to determine optimal dosing for an individual patient’s protection from and alleviation of symptoms of COVID-19. Clinical trials evaluating different combinations of nutritional supplementation should be conducted to determine if particular combinations offer better protection against COVID-19 compared with a single agent. Additionally, to identify under what conditions treatment yields the best results, patients in such trials should be grouped into subsets based on comorbidities, baseline nutrient levels, and level of severity of COVID-19. Overall, given the low-risk profile of supervised nutritional supplementation, it is important to ensure adequate, if not enhanced, intake of these critical nutrients in at-risk patients diagnosed with COVID-19.
REFERENCES
1. World Health Organization. WHO coronavirus (COVID-19) dashboard. https://covid19.who.int/. Accessed March 8, 2022.
2. Laviano A, Koverech A, Zanetti M. Nutrition support in the time of SARS-CoV-2 (COVID-19). Nutrition. 2020;74:110834.
3. de Faria Coelho-Ravagnani C, Corgosinho FC, Sanches FFZ, et al. Dietary recommendations during the COVID-19 pandemic. Nutr Rev. 2021;79(4):382-393.
4. Stroehlein JK, Wallqvist J, Iannizzi C, et al. Vitamin D supplementation for the treatment of COVID-19: a living systematic review. Cochrane Database Syst Rev. 2021;5(5):CD015043.
5. Xu Y, Baylink DJ, Chen CS, et al. The importance of vitamin d metabolism as a potential prophylactic, immunoregulatory and neuroprotective treatment for COVID-19. J Transl Med. 2020;18(1):322.
6. Leaf DE, Ginde AA. Vitamin D3 to treat COVID-19: different disease, same answer. JAMA. 2021;325(11):1047-1048.
7. Carr AC, Maggini S. Vitamin C and immune function. Nutrients. 2017;9(11):1211.
8. Holford P, Carr AC, Jovic TH, et al. Vitamin C—an adjunctive therapy for respiratory infection, sepsis and COVID-19. Nutrients. 2020;12(12):3760.
9. Manjunath SH, Thimmulappa RK. Antiviral, immunomodulatory, and anticoagulant effects of quercetin and its derivatives: potential role in prevention and management of COVID-19. J Pharm Anal. 2021;12(1):29-34.
10. Pal A, Squitti R, Picozza M, et al. Zinc and COVID-19: basis of current clinical trials. Biol Trace Elem Res. 2021;199(8):2882-2892.
11. Kleszczynski K, Slominski AT, Steinbrink K, Reiter RJ. Clinical trials for use of melatonin to fight against COVID-19 are urgently needed. Nutrients. 2020;12(9):2561.
12. Cross KM, Landis DM, Sehgal L, Payne JD. Melatonin for the early treatment of COVID-19: a narrative review of current evidence and possible efficacy. Endocr Pract. 2021;27(8):850-855.
13. World Health Organization. Weekly epidemiological update on COVID-19–22 June 2022. www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19---22-june-2022. Accessed June 23, 2022.
14. National Institutes of Health. Coronavirus disease 2019 (COVID-19) treatment guidelines. www.covid19treatmentguidelines.nih.gov/. Accessed March 9, 2022.
15. CDC. COVID-19 testing: what you need to know. www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/testing.html. Accessed June 9, 2022.
16. Brihn A, Chang J, Oyong K, et al. Diagnostic performance of an antigen test with RT-PCR for the detection of SARS-CoV-2 in a hospital setting—Los Angeles County, California, June–August 2020. MMWR Morb Mortal Wkly Rep. 2021;70(19):702-706.
17. FDA. Emergency use authorization. www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/emergency-use-authorization#coviddrugs. Accessed March 10, 2022.
18. Weir EK, Thenappan T, Bhargava M, Chen Y. Does vitamin D deficiency increase the severity of COVID-19? Clin Med (Lond). 2020;20(4):e107-e108.
19. Chen J, Mei K, Xie L, et al. Low vitamin D levels do not aggravate COVID-19 risk or death, and vitamin D supplementation does not improve outcomes in hospitalized patients with COVID-19: a meta-analysis and GRADE assessment of cohort studies and RCTs. Nutr J. 2021;20(1):89.
20. Entrenas Castillo M, Entrenas Costa LM, Vaquero Barrios JM, et al. “Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: a pilot randomized clinical study.” J Steroid Biochem Mol Biol. 2020;203:105751.
21. Zhou YF, Luo BA, Qin LL. The association between vitamin D deficiency and community-acquired pneumonia: a meta-analysis of observational studies. Medicine (Baltimore). 2019;98(38):e17252.
22. Dror AA, Morozov N, Daoud A, et al. Pre-infection 25-hydroxyvitamin D3 levels and association with severity of COVID-19 illness. PLos One. 2022;17(2):e0263069.
23. Annweiler C, Beaudenon M, Gautier J, et al; COVIT-TRIAL study group. COvid-19 and high-dose VITamin D supplementation TRIAL in high-risk older patients (COVIT-TRIAL): study protocol for a randomized controlled trial. Trials. 2020;21(1):1031.
24. Carr AC, Maggini S. Vitamin C and immune function. Nutrients. 2017;9(11):1211.
25. Hemila H, Chalker E. Vitamin C can shorten the length of stay in the ICU: a meta-analysis. Nutrients. 2019;11(4):708.
26. Corrao S, Mallaci Bocchio R, Lo Monaco M, et al. Does evidence exist to blunt inflammatory response by nutraceutical supplementation during COVID-19 pandemic? An overview of systematic reviews of vitamin D, vitamin C, melatonin, and zinc. Nutrients. 2021;13(4):1261.
27. Di Pierro F, Derosa G, Maffioli P, et al. Possible therapeutic effects of adjuvant quercetin supplementation against early-stage COVID-19 infection: a prospective, randomized, controlled, and open-label study. Int J Gen Med. 2021;14:2359-2366.
28. Shohan M, Nashibi R, Mahmoudian-Sani MR, et al. The therapeutic efficacy of quercetin in combination with antiviral drugs in hospitalized COVID-19 patients: a randomized controlled trial. Eur J Pharmacol. 2022;914:174615.
29. Skalny AV, Rink L, Ajsuvakova OP, et al. Zinc and respiratory tract infections: perspectives for COVID 19 (review). Int J Mol Med. 2020;46(1):17-26.
30. Wessels I, Pupke JT, von Trotha KT, et al. Zinc supplementation ameliorates lung injury by reducing neutrophil recruitment and activity. Thorax. 2020;75(3):253-261.
31. Rashidi AA, Salehi M, Piroozmand A, Sagheb MM. Effects of zinc supplementation on serum zinc and C-reactive protein concentrations in hemodialysis patients. J Ren Nutr. 2009;19(6):475-478.
32. Shakoor H, Feehan J, Al Dhaheri AS, et al. Immune-boosting role of vitamins D, C, E, zinc, selenium and omega-3 fatty acids: could they help against COVID-19? Maturitas. 2021;143:1-9.
33. Zhou Y, Hou Y, Shen J, et al. A network medicine approach to investigation and population-based validation of disease manifestations and drug repurposing for COVID-19. PLoS Biol. 2020;18(11):e3000970.
34. Farnoosh G, Akbariqomi M, Badri T, et al. Efficacy of a low dose of melatonin as an adjunctive therapy in hospitalized patients with COVID-19: a randomized, double-blind clinical trial. Arch Med Res. 2022;53(1):79-85.
35. Wichniak A, Kania A, Sieminski M, Cubala WJ. Melatonin as a potential adjuvant treatment for COVID-19 beyond sleep disorders. Int J Mol Sci. 2021;22(16):8623.
36. Marcinowska-Suchowierska E, Kupisz-Urbanska M, Lukaszkiewicz J, et al. Vitamin D toxicity—a clinical perspective. Front Endocrinol (Lausanne). 2018;9:550.
37. Dosedel M, Jirkovsky E, Macakova K, et al. Vitamin C—sources, physiological role, kinetics, deficiency, use, toxicity, and determination. Nutrients. 2021;13(2):615.
38. Andres S, Pevny S, Ziegenhagen R, et al. Safety aspects of the use of quercetin as a dietary supplement. Mol Nutr Food Res. 2018;62(1).
39. National Institutes of Health Office of Dietary Supplements. Zinc. https://ods.od.nih.gov/factsheets/Zinc-HealthProfessional/. Accessed March 8, 2022.
40. Meng X, Li Y, Li S, et al. Dietary sources and bioactivities of melatonin. Nutrients. 2017;9(4):367.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
To comment on this article, contact rdavidson@uspharmacist.com.






