US Pharm. 2014;39(6):34-38.
ABSTRACT: Onychomycosis (fungal infection of the nail) is a common dermatologic condition. Despite often being thought of as a purely cosmetic issue, it may be a source of morbidity and thus warrants treatment. Pharmacists, as highly accessible healthcare professionals, are able to play an important role in the prevention and treatment of onychomycosis. This article summarizes the prevalence, risk factors, prevention, diagnosis, and nonpharmacologic treatment of onychomycosis and also provides a review of available pharmacotherapy options.
Onychomycosis is a fungal infection of the nail apparatus that is caused by dermatophytes, most often Trichophyton rubrum, or nondermatophyte molds and yeasts, most often Candida species.1 It is a common dermatologic condition; prevalence in the general population in North America is estimated to be 7% to 15%, with about 90% of cases involving the toenails.1-3 Although some practitioners continue to view onychomycosis as a purely cosmetic issue, serious complications such as cellulitis can occur, particularly among patients with risk factors such as diabetes and peripheral vascular disease.1 Even patients at low risk for complications often report reduced quality of life; therefore, treatment should be considered whenever possible.1 Both topical and oral antifungal agents are available to treat onychomycosis, and it is important that pharmacists be aware of the efficacy, tolerability, and appropriate treatment duration of each antifungal.
Risk Factors and Prevention
Several risk factors are associated with predisposition to onychomycosis, including increasing age, exposure to damp communal areas (e.g., gyms, swimming pools), poor foot hygiene, impaired immune status, diabetes mellitus, poor peripheral circulation, tinea pedis, and nail trauma.2-4 Patients at risk for onychomycosis should be counseled on appropriate prevention techniques. Footwear should be worn when in damp communal areas, toenails should be kept short and clean, feet should be dried completely immediately after bathing, socks should be made of absorbent material (e.g., cotton) and changed if wet, and symptoms of tinea pedis should be addressed in a timely fashion.5
Diagnosis
Onychomycosis of the toenails may be clinically characterized by thickening of the toenails, onycholysis (separation of the nail plate from the nail bed), subungual hyperkeratosis (buildup of debris in the space created by onycholysis), and nail discoloration (usually yellow-brown or white) .1,4 Only about half of nail dystrophy cases are caused by fungi, making appropriate diagnostic technique necessary to rule out disease states with similar presentations (e.g., psoriasis, nail trauma).4 Currently, the most efficient screening test for onychomycosis is direct microscopy of a potassium hydroxide–prepared nail specimen; however, this technique has a false-negative rate of 5% to 15% and lacks differentiation between fungal pathogens.4 Fungal pathogens can only be identified by performing in vitro laboratory culture, which facilitates also antifungal susceptibility testing. Unfortunately, this technique suffers from a false-negative rate of nearly 50% .4 Due to their limitations, direct microscopy and in vitro laboratory culture of nail material should optimally be used together when diagnosing suspected onychomycosis. Despite this recommendation, onychomycosis is often treated empirically based on clinical presentation.
Treatment
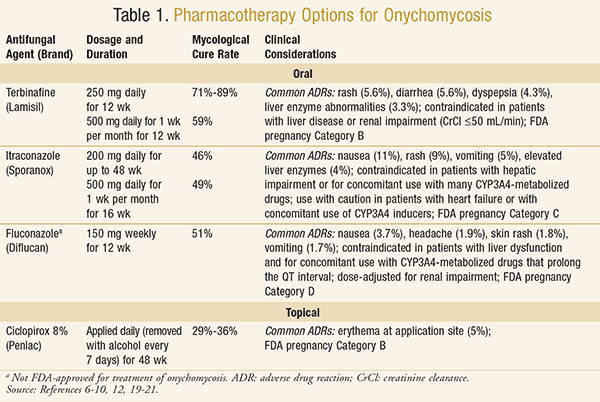
Dosage and duration, mycological cure rate, and clinical considerations (including adverse effects) for each antifungal agent discussed are listed in
TABLE 1.![]()
![]()
Oral Pharmacotherapy: In general, using an oral antifungal is the most effective treatment option for onychomycosis.1 However, oral antifungals may be inappropriate for some populations, particularly those with liver dysfunction.1 Griseofulvin, which has an FDA indication for onychomycosis, and ketoconazole, which does not, were commonly used before the introduction of the newer antifungals discussed in this article.4 However, the newer antifungals have shorter treatment durations, improved efficacy, and improved safety profiles, leading to the replacement of griseofulvin and ketoconazole, which are no longer recommended for use in this indication. For any agent chosen, treatment duration will be long (at least 12 weeks), and time to complete regrowth of healthy, disease-free nails can take up to 6 months for fingernails and 12 to 18 months for toenails.4
Terbinafine is a fungicidal allylamine derivative with an antifungal spectrum that includes dermatophytes and molds.4 In one study, FDA-approved continuous terbinafine therapy was found to be significantly more effective than pulse terbinafine therapy (patients take the drug for only 1 week per month), which is not an FDA-approved regimen (71% and 59%, respectively; P = .03) .6 Continuous terbinafine therapy offers the highest efficacy of any available therapy (71%-89% when used for 12 weeks for onychomycosis of the toenails) and should be recommended first-line for most patients without contraindications to therapy.6-8
Itraconazole is a fungistatic triazole with an antifungal spectrum that includes dermatophytes and molds.4 A continuous itraconazole regimen is FDA-approved for onychomycosis involving the toenails, and a pulse-dosing regimen (patients take the drug for only 1 week per month) is FDA-approved for onychomycosis with only fingernail involvement.9 Itraconazole therapy is generally longer than terbinafine therapy and has been investigated for up to 48 weeks of continuous use for onychomycosis of the toenails; however, it is still less effective than terbinafine.10 Itraconazole also has a higher rate of adverse drug reactions than terbinafine and fluconazole. For these reasons, it is recommended that itraconazole be reserved for use as a second-line therapy for patients who do not have success with terbinafine.
Fluconazole is a fungistatic triazole with an antifungal spectrum that is largely limited to Candida species and some dermatophytes.4 Fluconazole is not FDA-approved for the treatment of onychomycosis but has been studied for use off-label. One study compared a 12-week treatment regimen of fluconazole 150-mg once weekly to a 24-week regimen and found no significant difference in efficacy.7 Another study found no significant difference in efficacy between 150-mg, 300-mg, and 450-mg weekly regimens.11 Based on these results, to minimize risk of adverse effects while maintaining efficacy, the recommended dosing regimen for fluconazole is 150 mg once weekly for 12 weeks for onychomycosis of the toenails. Because of its similar efficacy, lower incidence of adverse events, and once-weekly dosing regimen, fluconazole may be considered a second-line option alongside itraconazole, despite its lack of FDA-approval for onychomycosis.
Topical Pharmacotherapy: Nonprescription topical antifungals marketed for athlete’s foot (e.g., terbinafine, tolnaftate) are not recommended for treating onychomycosis of the toenails.4 These topical antifungals are unable to penetrate the nail, a requirement to eradicate the infection; however, they may be useful for prophylaxis.4 ,5
Ciclopirox is a fungicidal pyridone with an antifungal spectrum that includes dermatophytes, molds, and yeasts.12 Although less effective than the oral antifungal agents, ciclopirox nail lacquer is associated with a lower incidence and severity of adverse effects than the oral antifungals.12 Ciclopirox represents a valid option for patients who cannot tolerate other options due to adverse effects or contraindications or for those who are hesitant to start long-term oral antifungal therapy.12 Concomitant use of systemic therapy (e.g., terbinafine) with topical ciclopirox may offer improved efficacy and decreased time to cure compared to either agent alone, although further research is required before this practice can be recommended routinely.4
Nonpharmacologic Treatment: Nail trimming and debridement can be used concurrently with pharmacologic treatments including topical therapies, to increase efficacy.
Several laser devices are FDA-approved for the treatment of onychomycosis, including neodymium :yttrium-aluminum-garnet ( Nd:YAG) devices and dual-wavelength near-infrared lasers.13 In one study, 61% of participants achieved mycological and cosmetic cure at week 16 of Nd:YAG therapy (mycological cure alone was not assessed).14 A second study demonstrated that 30% of participants with onychomycosis of the toenails achieved mycological cure at day 180 of dual-wavelength infrared laser therapy.15 Participants reported no adverse effects in either study.14,15
These therapies are much more expensive than pharmacologic methods and typically are not covered by insurance plans.13 For this reason, it may be prudent to recommend laser therapy for those patients who are unable or unwilling to use conventional pharmacologic options, those who have experienced treatment failure, or those for whom out-of-pocket expense is not an issue.
Alternative Options: Dosing and duration, mycological cure rate, and clinical considerations for each alternative option are listed in TABLE 2.
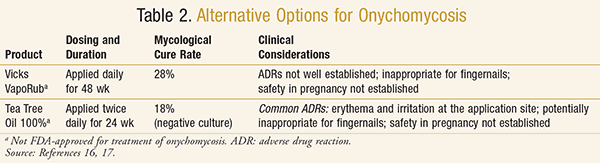
Vicks VapoRub (Procter & Gamble), which contains camphor, eucalyptus oil, menthol, and thymol, was investigated in a small study of toenail onychomycosis (N = 18) .16 The authors noted better outcomes for participants with positive cultures for Trichophyton mentagrophytes and Candida parapsilosis than for other organisms.16 Adverse effect rates and safety in pregnancy have not been established for this regimen. Although the Vicks VapoRub treatment showed similar efficacy to prescription-only ciclopirox 8% nail lacquer and is relatively low-cost (especially when factoring in appointment-related costs for prescription options), further research is needed before it can be routinely recommended to patients.16 Vicks VapoRub has not been researched for treatment of fingernail onychomycosis and may be inappropriate for that use because of the risk of irritation of the eyes or mucous membranes if touched by a fingernail that is being treated.
Tea tree ( Melaleuca alternifolia) oil has been investigated as a potential onychomycosis treatment. In one study, 18% of participants achieved a negative culture after twice-daily application for 24 weeks.17 Adverse effects associated with tea tree oil treatment included erythema and irritation at the application site.17 The safety of topical tea tree oil during pregnancy has not been well established. Like Vicks VapoRub, tea tree oil has not been tested for treatment of fingernail onychomycosis and may be inappropriate for such use because of the risk of irritation of the eyes or mucous membranes. Further study is needed before this therapy can be routinely recommended to patients.
Future Treatments
Several clinical trials related to onychomycosis treatment are ongoing. New dosage forms being investigated include nail lacquer and topical gel formulations of terbinafine as well as a patch formulation of terbinafine combined with ketoconazole.18 Several topical formulations of entirely new drug candidates are also being explored. If any are shown to be effective, these formulations may represent clinically important alternatives to ciclopirox nail lacquer for patients who are unable to tolerate oral antifungals.
Newer antifungal agents without FDA indications for onychomycosis, such as oral posaconazole and topical luliconazole, are also being investigated. New nonpharmacologic methods are also being researched, including microdrilling techniques to be combined with topical antifungals as well as new laser devices and methods.18
Conclusion
Pharmacists are an important source of information for patients and other healthcare providers regarding the prevention and treatment of onychomycosis, a common disease with several treatment options. Pharmacists should be prepared to discuss the pros and cons of oral and topical antifungal treatments, as well as nonpharmacologic and alternative treatments. In general, using an oral antifungal (particularly terbinafine) is the most effective option, but that may be inappropriate for some patient populations, such as those with liver dysfunction. Due to the long duration of therapy required for any onychomycosis treatment regimen, it is important for pharmacists to emphasize and support adherence to the chosen therapy.
REFERENCES
1. Roberts DT, Taylor WD, Boyle J. Guidelines for treatment of
onychomycosis.
Br J
Dermatol. 2003;
148(3):402-410.
2.
Ghannoum MA,
Hajjeh RA,
Scher R, et al. A large-scale North American study of fungal isolates from nails: the frequency of
onychomycosis, fungal distribution, and antifungal susceptibility patterns. J Am
Acad
Dermatol. 2000;
;43(4):641-648.
3. Gupta AK, Jain HC,
Lynde CW, et al. Prevalence and epidemiology of
onychomycosis in patients visiting physicians’ offices: a multicenter Canadian survey of 15,000 patients. J Am
Acad
Dermatol. 2000
;43(2):244-248.
4.
Elewski
BE.
Onychomycosis: pathogenesis, diagnosis, and management.
Clin
Microbiol Rev. 1998
;11(3):415-429.
5. Gupta AK,
Baran R,
Summerbell R.
Onychomycosis: strategies to improve efficacy and reduce recurrence. J
Eur
Acad
Dermatol
Venereol. 2002;16(6):579-586.
6.
Warshaw EM,
Fett DD, Bloomfield HE, et al. Pulse versus continuous
terbinafine for
onychomycosis: a randomized, double-blind, controlled trial. J Am
Acad
Dermatol. 2005;53(4):578-584.
7.
Havu V,
Heikkilä H,
Kuokkanen K, et al. A double-blind, randomized study to compare the efficacy and safety of
terbinafine (Lamisil) with fluconazole (
Diflucan) in the treatment of
onychomycosis. Br J
Dermatol. 2000;142(1):97-102.
8. Evans EG,
Sigurgeirsson B. Double-blind,
randomised study of continuous
terbinafine compared with intermittent
itraconazole in treatment of toenail
onychomycosis. The LION Study Group. BMJ. 1999;318(7190):1031-1035.
9.
Sporanox (
itraconazole) oral capsules package insert. Titusville, NJ: Janssen Pharmaceuticals,
Inc; 2012.
10. De Backer M, De
Vroey C,
Lesaffre E, et al. Twelve weeks of continuous oral therapy for toenail
onychomycosis caused by
dermatophytes: a double-blind comparative trial of
terbinafine 250 mg/day versus
itraconazole 200 mg/day. J Am
Acad
Dermatol. 1998;38(5):S57-S63.
11.
Scher RK,
Breneman D, Rich P, et al. Once-weekly fluconazole (150, 300, or 450 mg) in the treatment of distal
subungual
onychomycosis of the toenail. J Am
Acad
Dermatol. 1998;38(6):S77-S86.
12. Gupta AK,
Fleckman P,
Baran R.
Ciclopirox nail lacquer topical solution 8% in the treatment of toenail
onychomycosis. J Am
Acad
Dermatol. 2000;43(4
suppl):S70-S80.
13. Westerberg DP,
Voyack MJ.
Onychomycosis: current trends in diagnosis and treatment. Am
Fam Physician. 2013;88(11):762-770.
14. Kimura U, Takeuchi K, Kinoshita A, et al. Treating
onychomycoses of the toenail: clinical efficacy of the sub-millisecond 1,064 nm
Nd:YAG laser using a 5 mm spot diameter. J Drugs
Dermatol. 2012;11(4):496-504.
15. Landsman AS, Robbins AH. Treatment of mild, moderate, and severe
onychomycosis using 870- and 930-nm light exposure: some follow-up observations at 270 days. J Am
Podiatr Med Assoc. 2012;102(2):169-171.
16. Derby R,
Rohal P, Jackson C, et al. Novel treatment of
onychomycosis using over-the-counter mentholated ointment: a clinical case series. J Am Board
Fam Med. 2011;24(1):69-74.
17. Buck DS,
Nidorf DM,
Addino JG. Comparison of two topical preparations for the treatment of
onychomycosis:
Melaleuca
alternifolia (tea tree) oil and
clotrimazole. J
Fam
Pract. 1994;38(6):601-605.
18. Search results:
onychomycosis. U.S. National Institutes of Health Clinical Trials. http://clinicaltrials.gov/ct2/results?term=onychomycosis. Accessed March 30, 2014.
19. Lamisil (
terbinafine hydrochloride) oral tablets package insert. East Hanover, NJ: Novartis Pharmaceuticals Corp; 2005.
20.
Diflucan (fluconazole) oral tablets, powder for oral suspension, solution for intravenous infusion package insert. New York, NY:
Roerig; 2010.
21.
Penlac (
ciclopirox) topical solution package insert. Berwyn, PA:
Dermik Laboratories; 2005.






