US Pharm. 2018;43(6):29-33.
ABSTRACT: Oral phosphodiesterase type 5 (PDE5) inhibitors remain the standard pharmacologic treatment for erectile dysfunction (ED). ED, a pervasive disorder that is common in men older than 40 years, can have significant consequences for quality of life and self-esteem. The brand and generic markets for these medications are changing, allowing patients and providers more flexibility in their treatment decisions. As a first-line advocate for patients and a source of drug information, the pharmacist—along with other healthcare professionals—will benefit from an understanding of the subtle differences between the oral PDE5 inhibitors in order to optimize patient therapy.
Erectile dysfunction (ED) is a common sexual-arousal disorder primarily affecting men over the age of 40 years. ED is clinically defined as the inability to attain or maintain a penile erection sufficient for sexual intercourse. In patients without trauma or surgery, a 3-month duration of this symptom is usually accepted as grounds for diagnosis.1 The landmark population-based Massachusetts Male Aging Study reported that up to 52% of men aged 40 to 69 years may experience ED to varying degrees. Although age does not directly precipitate ED, a correlation exists.2 Men with ED exhibit decreased quality of life in both physical and psychological dimensions, including self-esteem, compared with men without ED.3 ED impacts not only the patient with ED; partners of men with ED may experience poorer quality of life and report their own sexual dysfunction more than those whose partners do not have ED.4 An emerging field of evidence has suggested that ED is also a marker for cardiovascular disease and may be correlated with general male health status regardless of etiology.5 Compounding the significant prevalence of ED and the reduced quality of life that accompanies it, studies have suggested that men often underreport ED because of embarrassment or lack of awareness of medical causes.6 Therefore, optimal nonpharmacologic and/or pharmacologic treatment for ED is necessary.
During a normal penile erection, parasympathetic stimulation leads to nitric oxide (NO) release from endothelial cells within the penis.7 After sexual stimulation, NO concentration is significantly increased and contributes to the conversion of guanosine triphosphate to cyclic guanosine monophosphate (cGMP). Downstream, cGMP decreases intracellular Ca2+ (calcium ions) in the cavernosal smooth muscles, leading to smooth-muscle relaxation. Once relaxed, the smooth muscles collapse the veins, which causes reduced drainage of arterial blood, thus sustaining an erection. Given the complexity and regulated coordination of this process, multiple etiologies may contribute to the inability to attain or maintain a penile erection sufficient for intercourse. The etiology of ED is generally classified as psychogenic, organic (i.e., neurogenic, endocrinologic, vasculogenic, or drug-induced systemic disease), or mixed psychogenic and organic, with mixed etiology the most common type.7
One of the mainstays of ED treatment is the oral phosphodiesterase type 5 (PDE5) inhibitor class. The four major PDE5 inhibitors are sildenafil (Viagra), tadalafil (Cialis), vardenafil (Levitra), and avanafil (Stendra). During the penile erection process, cGMP is metabolized through the PDE5 enzyme and cannot exert its downstream erectile effects. PDE5 inhibitors are selective, competitive, and reversible, generally working to decrease cGMP metabolism and ultimately leading to successful attainment and maintenance of an erection.8 Comprehension of the subtle differences between the PDE5 inhibitors will help the healthcare professional select the most appropriate pharmacologic therapy for a patient. One significant consideration is cost, which is a significant consideration and, along with the patient’s insurance coverage or limitations, may restrict choice. Because generic PDE5 inhibitors will soon enter the market, an understanding of the unique differences between therapies will be beneficial for the pharmacist—as a first-line advocate for patients and a source of drug information—as well as other healthcare professionals.
This article reviews the roles of the four major oral PDE5 inhibitors in the treatment of ED, evidence supporting their use, cost comparisons, and distinctions between therapies.
A Brief History of Oral PDE5 Inhibitors
In the mid-1980s, the association between NO and the PDE family sparked an increase in drug innovation. The numerous physiological effects of NO had dramatic implications for a number of diseases. The PDE enzyme is ubiquitous in the body, with 11 distinct recognized isoenzymes expressed in different concentrations in various tissues. The PDE5 enzyme is widespread but is more prevalent in penile tissue. Nonselective PDE inhibitors (e.g., theophylline) were used prior to the discovery of the link between NO and PDEs, but selective PDE inhibitors were not yet developed. Since then, a number of selective PDE inhibitors have been approved to treat a variety of disorders ranging from ED to pulmonary hypertension.9
Sildenafil (Viagra) was first studied in clinical trials for coronary heart disease in 1991, but the drug serendipitously had favorable effects on penile erections. By 1998, Viagra was FDA-approved as the first oral treatment for ED. As realization of the market potential for oral PDE5 inhibitors dawned, pharmaceutical companies set out to make new products and improvements. Different potencies, durations of action, and onsets of action played into the creation of alternative PDE5 inhibitor therapies, resulting in the approval of Cialis (2003), Levitra (2003), and Stendra (2012) for treatment of ED.10 In 2008, Cialis earned the first market approval for once-daily dosing, which may appeal to individuals desiring more spontaneity. Although Viagra’s patent was extended to 2020, Pfizer entered an agreement with a generic manufacturer to market sildenafil in 2017, making this drug the first generic PDE5 inhibitor. Generic versions of both Cialis and Levitra are anticipated to be introduced later in 2018.
ED Treatment Guidelines
Based on the 2007 American Urological Association Update for the Management of Erectile Dysfunction, first-line standard management of ED includes identifying and treating organic comorbidities and psychogenic dysfunction. Oral PDE5 inhibitors remain a first-line pharmacologic treatment.11
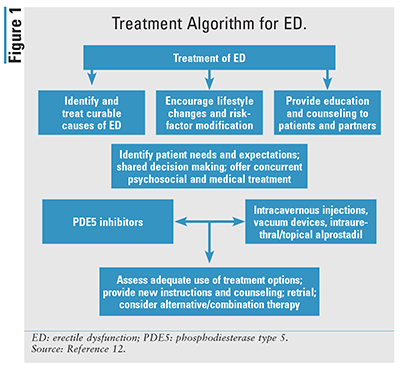
As FIGURE 1 illustrates, the general course of therapy for ED begins with identifying and treating ED with a curable etiology, such as primary testicular failure or secondary pituitary/hypothalamic causes.12 Beyond the curable causes of ED, lifestyle modification through risk-factor reduction and education remains paramount. In addition to these nonpharmacologic standards, the use of an oral PDE5 inhibitor is the standard medical treatment following a shared decision-making agreement. Assessment of treatment options includes a patient-focused discussion incorporating information about risk factors and lifestyle needs. Treatment options should be tailored to patient and partner satisfaction, quality-of-life factors, and treatment-related safety and efficacy considerations. Emphasis is placed on shared decision making and on attempting a new therapy if the current therapy fails.12
Efficacy in Clinical Trials
A number of trials have examined the efficacy and safety of PDE5 inhibitors with regard to objective efficacy parameters and subjective patient-preference parameters. Factors associated with improved long-term treatment success include enhanced patient-provider communication, increased knowledge of drug characteristics, and optimal prescribing patterns.13
A 2013 systematic review and traditional and network meta-analysis by Yuan and colleagues compared the effectiveness and safety of the four PDE5 inhibitors across 118 trials involving 31,195 subjects.14 Effectiveness criteria were risk ratio of Global Assessment Questionnaire 1 (GAQ-1) score (GAQ-1: improvement of erections), mean difference in International Index of Erectile Function erectile-function domain, and mean difference in Sexual Encounter Profile (SEP) questions 2 and 3 (SEP-2: ability to insert; SEP-3: successful completion of intercourse). Network meta-analysis results generally indicated that all of the oral PDE5 inhibitors were more effective than placebo in all studied domains of ED. Network meta-analysis compared tadalafil, vardenafil, and avanafil with sildenafil across all domains. Of particular note, avanafil was associated with significantly lower GAQ-1 responses (relative risk [RR], 0.63 [95% CI, 0.33-0.90]). In the mean difference (MD) assessment of SEP-2, tadalafil (MD, 19.00 [95% CI, 11.34-26.76]) and vardenafil (MD, 18.67 [95% CI, 10.81-26.64]) were superior to sildenafil. In the MD assessment of SEP-3, tadalafil (MD, 18.92.00 [95% CI, 6.66-30.79]) and vardenafil (MD, 19.01 [95% CI, 6.51-31.54]) were also superior to sildenafil. In a rank-of-effectiveness analysis, tadalafil was most likely to be effective across domains, followed by vardenafil. However, comparisons were evaluated as low-grade evidence limited by poor original reporting and indirect comparisons.14
This meta-analysis was a more comprehensive adaptation of a 2009 systematic review. In the earlier trial, the authors found there to be no significant difference in effectiveness and adverse effects (AEs) in a review of four head-to-head PDE5 inhibitor trials.15
Patient Preferences
As is commonly emphasized by ED guidelines, the choice of oral PDE5 inhibitor is made in a shared clinical decision-making process between patient and provider. Several studies and review articles have evaluated patients’ general preference for, adherence to, and satisfaction with a certain PDE5 inhibitor. A comparison of preference studies conducted on sildenafil, vardenafil, and tadalafil, however, found a high degree of bias and flawed methodology in the studies.16
A 2009 review of seven preference trials found that five trials comparing tadalafil with sildenafil reported a general preference for tadalafil based on a longer duration of action.17 In a 6-month observational study involving 8,047 men with ED, patients taking sildenafil or vardenafil were significantly more likely to switch prescriptions compared with those taking tadalafil (odds ratio, 4.43 and 4.14, respectively; P <.0001).18 Of patients who switched from tadalafil, 25% switched back to tadalafil compared with a switch-back rate of less than 10% in the sildenafil and vardenafil cohorts.18 Across studies of patient preference, tadalafil is consistently preferred over sildenafil or vardenafil. One of the preferential factors may be that, unlike sildenafil or vardenafil, tadalafil may be taken with fatty foods without disrupting bioavailability; longer duration of action and more spontaneity surrounding sexual activity may also play into the slight preference for tadalafil.19
Given the relatively recent market history of avanafil, comparative trials are lacking. Avanafil has been shown to have similar efficacy and safety considerations as the other three major PDE5 inhibitors. A unique benefit of avanafil, however, is its quick onset of action. Cost is an important consideration with avanafil because of the impending availability of generic PDE5 inhibitors.20
Safety and Administration Considerations
The most commonly reported AEs for oral PDE5 inhibitors are headache, flushing, dyspepsia, dizziness, and rhinitis.21-26 In the aforementioned 2013 meta-analysis by Yuan and colleagues, safety was statistically similar between the oral PDE5 inhibitors, with the exception of a higher incidence of myalgia with tadalafil versus sildenafil (RR, 4.69 [95% CI, 1.39-14.21]).14 Incidents of visual abnormalities with sildenafil and vardenafil are common.
Oral PDE5 inhibitors have slight off-site binding affinity to other PDE enzymes. Vasodilation and cardiovascular safety are pertinent concerns that are reflected in the drugs’ contraindications, warnings, and precautions.21-26
Contraindications: Any concurrent use of organic nitrates, including sublingual nitroglycerin, amyl nitrite, and isosorbide mononitrate or dinitrate, is contraindicated. Nitroglycerin must be withheld at least 24 hours following the last dose of sildenafil or vardenafil, 48 hours following that of tadalafil, and 12 hours following that of avanafil, owing to the drugs’ variable half-lives.21-26
Precautions: All PDE5 inhibitors have precautions in the following cardiovascular categories: 1) myocardial infarction, stroke, or life-threatening arrhythmia in the last 6 months; 2) resting hypotension (blood pressure [BP] <90/50 mmHg) or hypertension (BP >170/100 mmHg); and 3) unstable angina or angina during sexual intercourse or congestive heart failure categorized as New York Heart Association Class IV.21-26
Antihypertensive Coadministration: Alpha-blockers are a significant concern—to varying degrees—among the PDE5 inhibitors; dosing recommendations differ based on alpha-blocker use. This is especially significant given the increased occurrence of benign prostatic hyperplasia. All PDE5 inhibitors administered concomitantly with antihypertensives or alcohol use may lower BP and therefore should be used with caution.21-26
CYP Considerations: All PDE5 inhibitors are CYP3A4 substrates and have varying dose adjustments based on CYP inhibition. Drugs that affect CYP3A4 will work to increase or decrease total PDE5 inhibitor bioavailability, thereby affecting the activity of these drugs. Although there is variability in dosage requirements between the drugs, all PDE5 inhibitors require decreased doses if the drug is coadministered with a moderate or potent CYP3A4 inhibitor.21-26
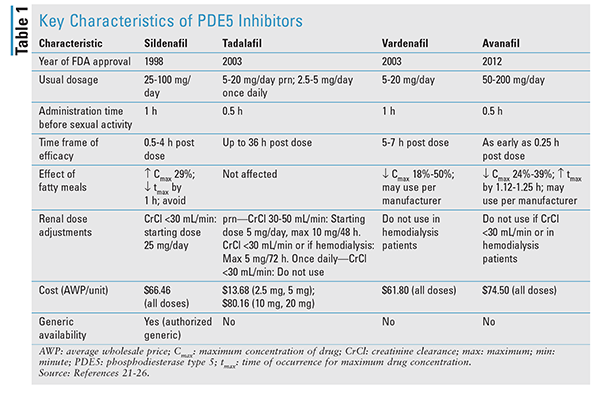
TABLE 1 summarizes some of the key characteristics of the four major PDE5 inhibitors.
Conclusion
PDE5 inhibitors represent a major first-line oral therapy option for men with ED. The prevalence and health implications of ED are far-reaching. Based on the impending shift in the oral PDE5 inhibitor market from brand to generic products, patients and providers will have more freedom of choice, and cost will be a lesser consideration. Although multiple reports suggest general equivalency of efficacy and safety between the four major PDE5 inhibitors, tadalafil with optimal use has been suggested to be preferable, providing increased efficacy in the ability to insert and the successful completion of intercourse. AEs, onset and duration of action, administration considerations, precautions, and cost are all key factors in individualizing therapy. Choice of oral PDE5 inhibitor therapy should be guided by a discussion between patient and provider that focuses on understanding the various characteristics, which can increase the likelihood of satisfaction.
REFERENCES
1. Impotence. NIH Consensus Development Panel on Impotence. JAMA. 1993;270:83-90.
2. Johannes CB, Araujo AB, Feldman HA, et al. Incidence of erectile dysfunction in men 40 to 69 years old: longitudinal results from the Massachusetts male aging study. J Urol. 2000;163:460-463.
3. Sánchez-Cruz JJ, Cabrera-León A, Martín-Morales A, et al. Male erectile dysfunction and health-related quality of life. Eur Urol. 2003;44:245-253.
4. Wagner G, Fugl-Meyer KS, Fugl-Meyer AR. Impact of erectile dysfunction on quality of life: patient and partner perspectives. Int J Impot Res. 2000;12(suppl 4):S144-S146.
5. Salonia A, Castagna G, Saccà A, et al. Is erectile dysfunction a reliable proxy of general male health status? The case for the International Index of Erectile Function-Erectile Function domain. J Sex Med. 2012;9:2708-2715.
6. Baldwin K, Ginsberg P, Harkaway RC. Under-reporting of erectile dysfunction among men with unrelated urologic conditions. Int J Impot Res. 2003;15:87-89.
7. Gratzke C, Angulo J, Chitaley K, et al. Anatomy, physiology, and pathophysiology of erectile dysfunction. J Sex Med. 2010;7(1 pt 2):445-475.
8. Shamloul R, Ghanem H. Erectile dysfunction. Lancet. 2013;381:153-165.
9. Elhwuegi AS. The wonders of phosphodiesterase-5 inhibitors: a majestic history. Ann Med Health Sci Res. 2016;6:139-145.
10. Connelly D. Three decades of Viagra. The Pharmaceutical Journal. www.pharmaceutical-journal.com/news-and-analysis/infographics/three-decades-of-viagra/20202847.article. Accessed April 26, 2018.
11. American Urological Association. The management of erectile dysfunction: an update. www.auanet.org/documents/education/clinical-guidance/Erectile-Dysfunction.pdf. Accessed February 18, 2018.
12. European Association of Urology. EAU guidelines on erectile dysfunction, premature ejaculation, penile curvature and priapism. https://uroweb.org/wp-content/uploads/EAU-Guidelines-Male-Sexual-Dysfunction-2016-3.pdf. Accessed February 18, 2018.
13. Smith WB II, McCaslin IR, Gokce A, et al. PDE5 inhibitors: considerations for preference and long-term adherence. Int J Clin Pract. 2013;67:768-780.
14. Yuan J, Zhang R, Yang Z, et al. Comparative effectiveness and safety of oral phosphodiesterase type 5 inhibitors for erectile dysfunction: a systematic review and network meta-analysis. Eur Urol. 2013;63:902-912.
15. Tsertsvadze A, Fink HA, Yazdi F, et al. Oral phosphodiesterase-5 inhibitors and hormonal treatments for erectile dysfunction: a systematic review and meta-analysis. Ann Intern Med. 2009;151:650-661.
16. Raheem AA, Kell P. Patient preference and satisfaction in erectile dysfunction therapy: a comparison of the three phosphodiesterase-5 inhibitors sildenafil, vardenafil and tadalafil. Patient Prefer Adherence. 2009;3:99-104.
17. Mirone V, Fusco F, Rossi A, et al. Tadalafil and vardenafil vs sildenafil: a review of patient-preference studies. BJU Int. 2009;103:1212-1217.
18. Hatzichristou D, Haro JM, Martín-Morales A, et al. Patterns of switching phosphodiesterase type 5 inhibitors in the treatment of erectile dysfunction: results from the Erectile Dysfunction Observational Study. Int J Clin Pract. 2007;61:1850-1862.
19. Martín Morales A, Casillas M, Turbi C. Patients’ preference in the treatment of erectile dysfunction: a critical review of the literature. Int J Impot Res. 2011;23:1-8.
20. Evans JD, Hill SR. A comparison of the available phosphodiesterase-5 inhibitors in the treatment of erectile dysfunction: a focus on avanafil. Patient Prefer Adherence. 2015;9:1159-1164.
21. Huang SA, Lie JD. Phosphodiesterase-5 (PDE5) inhibitors in the management of erectile dysfunction. P T. 2013;38:407,414-419.
22. Levitra (vardenafil hydrochloride) package insert. Whippany, NJ: Bayer HealthCare Pharmaceuticals Inc; April 2014.
23. Stendra (avanafil) package insert. Cranford, NJ: Mist Pharmaceuticals; September 2017.
24. Cialis (tadalafil) package insert. Indianapolis, IN: Lilly USA, LLC; February 2018.
25. Viagra (sildenafil citrate) package insert. New York, NY: Pfizer Inc; December 2017.
26. Red Book Online [Micromedex Solutions; subscription database]. Greenwood Village, CO: Truven Health Analytics, Inc. www.micromedexsolutions.com. Accessed February 19, 2018.
To comment on this article, contact rdavidson@uspharmacist.com.






