US Pharm. 2020;45(6):HS-11-HS-16.
ABSTRACT: Prostatitis, an inflammation of the prostate gland, is a common condition, with prevalence peaking in middle-aged men. Chronic prostatitis/chronic pelvic pain syndrome is characterized by pelvic pain, variable urinary symptoms, and sexual dysfunction. Focused multimodal therapy appears to be more successful than empiric monotherapy, which may add complexity to the drug regimen and calls for collaboration with a pharmacist for an effective continuum of care, management of pain, and antibiotic stewardship.
Chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS) is a common condition worldwide, affecting approximately 2% to 10% of men. The prevalence seems to peak in the fifth decade and decline thereafter and shows no apparent racial predisposition. The symptomatic, chronic forms of prostatitis as defined by the National Institutes of Health (NIH; Table 1), are chronic bacterial prostatitis (CBP; NIH category II) and CP/CPPS (NIH category III). Most men diagnosed with prostatitis have CP/CPPS rather than acute or chronic bacterial prostatitis. Despite having a significant negative impact on patients’ quality of life (QoL) and presenting diagnostic and therapeutic challenges for physicians, CP/CPPS has received relatively little attention in the literature, in comparison with other urologic conditions.1-8
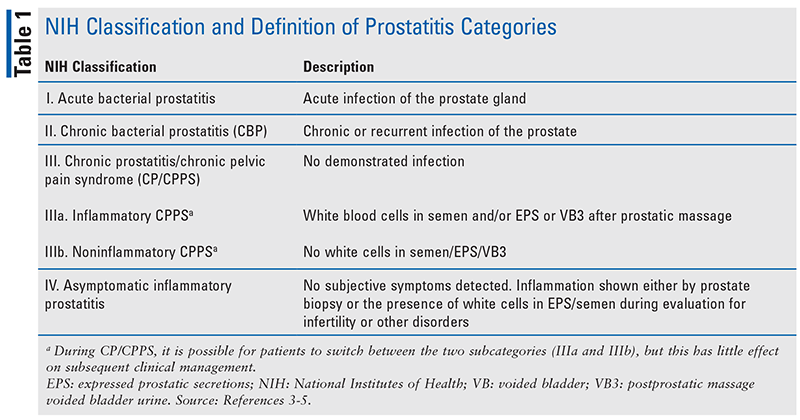
The NIH guidelines define CP/CPPS as chronic pelvic pain that is present for at least 3 of the preceding 6 months in the absence of other identifiable causes.4 This pain is often associated with urinary symptoms and/or sexual dysfunction. This condition, formerly called nonbacterial prostatitis, is generally diagnosed when chronic symptoms are accompanied by midstream urine cultures. What differentiates CP/CPPS from other forms is that there is limited understanding as to its underlying etiology. Interestingly, it is unknown if the prostate is even the main source of pain for this condition and despite the name, its clinical importance is unclear. Additionally, whether or not these patients fall into the inflammatory or noninflammatory classification, there is no evidence that patients in the two subgroups have different symptoms or respond differently to therapy. During a clinical work-up, it is imperative that other causes of pelvic pain are excluded. There are proposed etiologies that suggest CP/CPPS may result from a challenging interplay between psychological factors and dysfunction in the neurologic, immune, and endocrine systems. Given the overall innervation of the bowel and bladder, irritation of the bowel can result in lower abdominal pain and urinary symptoms as well. Moreover, there is evidence that shows, like many other chronic comorbidities, CP/CPPS may lead to problems with daily activities, depression, and overall QoL.4,7-12
Clinical Presentation
CP/CPPS is a diagnosis of exclusion; the evaluation is designed to rule out identifiable causes of pelvic pain and includes a history, physical examination, urinalysis, urine culture, and, in selected cases, diagnostic imaging. The primary symptom of CP/CPPS is usually a dull or sharp pain, which may rage from being variable in duration, lasting minutes, to being constantly present (Table 2). Studies have shown that the most common locations of pain were the perineum, testes, suprapubic area, and penis; yet, given the sensitive area of the pain site, some patients may not disclose the location of their pain to their primary care provider and/or urologist. Pharmacists may take a proactive role in helping patients clarify and describe their pain, which includes dysuria and pain with ejaculation, and in managing pain treatment.3-5
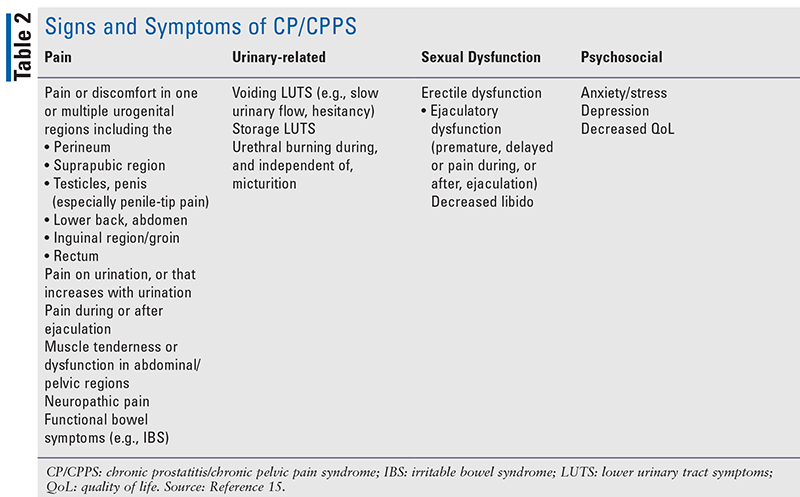
To aid with the clinical assessment of CP/CPPS, both in terms of initial evaluation and during therapeutic monitoring, validated symptom-scoring instruments are used in practice. These include the NIH Chronic Prostatitis Symptom Index (NIH-CPSI, which evaluates pain, voiding, and impact on QoL); the International Prostate Symptom Score (IPSS; urinary symptoms and impact on QoL); and the more recent Urinary, Psychosocial, Organ-specific, Infection, Neurological/systemic, and Tenderness (UPOINT) classification.
Correlation studies of sexual dysfunction symptoms that indicate CP/CPPS patients have lower total QoL scores (lower scores indicate more severe symptoms), suggesting that sexual symptoms can contribute to the morbidity experienced by men with CP/CPPS. Moreover, research using these scales has also shown negative behavioral consequences and psychosocial symptoms, such as depression and anxiety, which can have a significant impact on QoL. Therefore, there is a need for a pharmacist on the interdisciplinary care team for patients with CP/CPPS to assess medication tolerance and adherence, as well as nonpharmacologic management of current symptoms. The inclusion of a pharmacist is critical for medication management and allows patient-centered care teams to consistently deliver evidence-based care.6-8,13-17
Management
There is no uniformly accepted treatment regimen for CP/CPPS; the approach to each patient is individualized based on the predominant symptoms. Many treatments are used in combination, and monotherapy is generally considered to be less effective. The pharmacist-patient relationship is critical in order to educate, counsel, and provide a continuum of pharmaceutical care since both patient preference and symptom management are important determinants of treatment choice.15-17
In most patients, physicians will prescribe either a selective or nonselective alpha-adrenergic antagonist, in combination with a shorter course of an anti-inflammatory medication for CP/CPPS patients who present with voiding lower urinary tract symptoms (LUTS) (Table 3). Pharmacists should keep in mind the side-effect profiles of alpha-blockers and consider offering uroselective alpha-blockers, in collaboration with the physician. These agents work in CP/CPPS by relieving the secondary smooth-muscle spasm within the bladder neck and prostatic urethra, which affords the patient greater comfort in voiding. An evidence-based recommendation is tamsulosin 0.4 mg daily for 6 weeks and ibuprofen 400 mg three times daily for 1 week. Anti-inflammatory medications can also be used alone, as can medications for neuropathic pain. In patients with continued pain and voiding symptoms, it is reasonable to continue an alpha-antagonist beyond the initial treatment duration. Pharmacists working in the continuum of care with the patient should help to ensure the patients are reevaluated after 3 months of continued alpha-blocker therapy.15,16,18-26
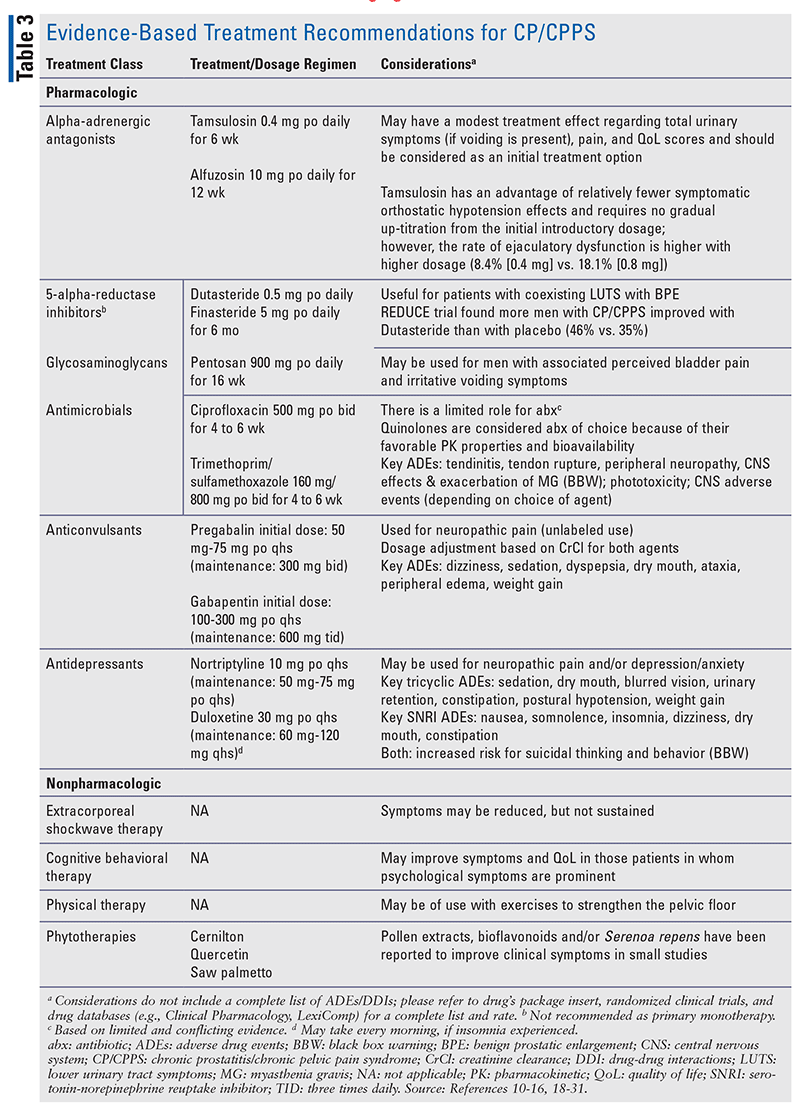
The evidence-base for the use of 5-alpha-reductase inhibitors is limited, and they are not recommended to be used as monotherapy; however, these drugs may serve as an option for patients with voiding difficulties, as treatment for sexual dysfunction, and for patients with benign prostatic hyperplasia with CP/CPPS. While these agents are commonly used in older men, they are not recommended in young men who are still trying to have children, given the effects on semen volume.10,11,15,16,18
In patients who have a negative urine culture and features consistent with CP/CPPS, practitioners generally should not treat with antibiotics due to limited and conflicting available evidence. However, some trials have shown improvement in symptoms after antibiotic treatment, and certain antibiotic classes such as quinolones and tetracyclines have anti-inflammatory and analgesic properties, which may account for symptom improvement, regardless of infection. It is not unreasonable to offer a short course of antibiotics despite a negative urine culture as a one-time trial intervention for naïve early-onset prostatitis patients. Any use beyond this may not be the most favorable approach and is an opportunity for discussion with the patients’ physician, as this could be a contributor to antibiotic resistance. However, in patients with a positive urine culture and symptoms consistent with cystitis, a 7-day course of antibiotics is recommended. 9-12,15,16,19
Role of the Pharmacist
Pharmacists play a critical role in patient-centered care by collaborating with the patient’s primary care provider and specialists (e.g., urologist) to develop and maintain an individualized treatment regimen based on the patient’s CP/CPPS symptoms. Furthermore, pharmacists have a duty to perform medication-therapy management by reviewing the records of these patients to minimize serious drug interactions for CP/CPPS and other comorbid conditions. Lastly, and perhaps most importantly, the pharmacist has an important role in encouraging multidisciplinary collaboration within the health system to ensure that the therapeutic use of antimicrobial treatment results in optimal patient outcomes and to combat antibiotic resistance by the avoidance of overuse of these agents in CP/CPPS.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
REFERENCES
1. Krieger JN, Reilly DE, Cheah PY, et al. Epidemiology of prostatitis: new evidence for a world-wide problem. World J Urol. 2003;21:70.
2. Nickel JC, Downey J, Hunter J, et al. Prevalence of prostatitis-like symptoms in a population-based study using the National Institutes of Health chronic prostatitis symptom index. J Urol. 2001;165(3):842-845.
3. Krieger JN, Nyberg L Jr, Nickel JC. NIH consensus definition and classification of prostatitis. JAMA. 1999;282:236-237.
4. Nickel JC, Nyberg LM, Hennenfent M. Research guidelines for CP: consensus report from the first National Institutes of Health International Prostatitis Collaborative Network. Urology 1999;54:229-233.
5. Nyberg LM, et al. National Institutes of Health Classification of Chronic Prostatitis. In Nickel JC, ed. Textbook of Prostatitis. London: CRC Press. 1999:28.
6. Wagenlehner FM, van Till JW, Magri, V, et.al. National Institutes of Health Chronic Prostatitis Symptom Index (NIH-CPSI) symptom evaluation in multinational cohorts of patients with chronic prostatitis/chronic pelvic pain syndrome. Eur Urol. 2013;63:953.
7. McNaughton Collins M, Pontari MA, O’Leary MP, et.al. Quality of life is impaired in men with CP: the Chronic Prostatitis Collaborative Research Network. J Gen Intern Med. 2001;16:656-662.
8. Walz J, Perrotte P, Hutterer G, et al. Impact of chronic prostatitis-like symptoms on the quality of life in a large group of men. BJU Int. 2007;100:1307.
9. Schaeffer AJ. Clinical practice. CP and CPPS syndrome. N Engl J Med. 2006;355(16):1690-1698.
10. Murphy AB, Macejko A, Taylor A, Nadler RB. CP: management strategies. Drugs. 2009;69(1):71-84.
11. Sharp VJ, Takacs. EB, Powell CP, et al. Prostatitis: diagnosis and treatment. Am Fam Physician. 2010;82(4):397-406.
12. Grabe M, Bartoletti R, Bjerklund Johansen TE, et al. Guidelines on urological infections, 2013. European Association of Urology. http://uroweb.org/wp-content/uploads/18_Urological-infections_LR.pdf. Accessed March 2020.
13. Nickel JC, Mullins C, Tripp DA. Development of an evidence-based cognitive behavioural treatment program for men with CP/CPPS. World J Urol. 2008;26:167-172.
14. Naliboff BD, Stephens AJ, Afariet N. al. Widespread psychosocial difficulties in men and women with urologic CPPS: case-control findings from the multidisciplinary approach to the Study of CPP Research Network. Urology. 2015;85:1319.
15. Rees J, Abrahams M, Doble A, et al. Diagnosis and treatment of CBP and CP/CPPS: a consensus guideline. BJU International. 2015;116:509-525.
16. Magistro G, Wagenlehner FME, Grabe M. Contemporary management of CP/CPPS. Eur Urol. 2016;69:286-297.
17. Locke A, Kamo N. Utilizing clinical pharmacists to improve delivery of evidence-based care for depression and anxiety in primary care. BMJ Qual Improvem Rep. 2016;5:u211816.w4748.
18. Nickel JC, Roehrborn C, Montorsi F. Dutasteride reduces prostatitis symptoms compared with placebo in men enrolled in the REDUCE study. J Urol. 2011;186:1313-1318.
19. Thakkinstian A, Attia J, Anothaisintawee T, Nickel JC. alpha-blockers, antibiotics and anti-inflammatories have a role in the management of CP/CPPS BJU Int. 2012;110:1014-1022.
20. Pontari MA, Krieger JN, Litwin MS. Pregablin for the treatment of men with CP/CPPS: a randomized controlled trial. Arch Intern Med. 2010;170:1586-1593.
21. Aboumarzouk OM, Nelson RL. Pregablin for chronic prostatitis. Cochrane Database Syst Rev. 2012; CD009063.
22. Agarwal MM, Sy ME. Gabapentenoids in pain management in urological CPPS: Gabapentin or pregabalin? Neurourol Urodyn. 2017;36:2028.
23. Giannantoni A, Porena M, Gubbiott M, et al. The efficacy and safety of duloxetine in a multidrug regimen for CP/CPPS. Urology. 2014;83:400.
24. Barbalias GA, Nikiforidis G, Liatsikos EN. Alpha-blockers for the treatment of CP in combination with antibiotics. J Urol. 1998;159(3):883-887.
25. Flomax (tamsulosin hydrochloride) prescribing information. Ridgefield, CT; Boehringer Ingelheim Pharmaceuticals, Inc; 2002.
26. Nickel JC, Touma N. Alpha-blockers for the treatment of chronic prostatitis/chronic pelvic pain syndrome: an update on current clinical evidence. Rev Urol. 2012;14(3/4):56-64.
27. Elist J. Effects of pollen extract preparation prostat/poltit on lower urinary tract symptoms in patients with chronic nonbacterial prostatitis/CPPS: a randomized, double-blind, placebo-controlled study. Urology. 2006;67:60-63.
28. Shoskes DA, Zeitlin SI, Shahed A. Quercetin in men with category III chronic prostatitis: a preliminary prospective, double-blind, placebo-controlled trial. Urology. 1999;54:960-963.
29. Morgia G, Mucciardi G, Galì A. Treatment of CP/CPPS category IIIA with Serenoa repens plus selenium and lycopene (Profluss) versus S. repens alone: an Italian randomized multicenter-controlled study. Urol Int. 2010;84:400-406.
30. Cai T, Mazzoli S, Bechi A. Serenoa repens associated with Urtica dioica (ProstaMEV) and curcumin and quercitin (FlogMEV) extracts are able to improve the efficacy of prulifloxacin in bacterial prostatitis patients: results from a prospective randomised study. Int J Antimicrob Agents. 2009;33:549-553.
31. Nickel JC, Forrest JB, Tomera K, et al. Pentosan polysulfate sodium therapy for men with chronic pelvic pain syndrome: a multicenter, randomized, placebo-controlled study. J Urol. 2005;173(4):1252-1255.
To comment on this article, contact rdavidson@uspharmacist.com.






