US Pharm. 2010;35(12):29-43.
Millions of Americans suffer from peptic ulcer disease (PUD). With approximately 10% of the U.S. population experiencing this condition, it has significantly impacted our health care system.1,2 The prevalence within the United States has become equal for both men and women.1 Death rates over the last 50 years have declined for PUD, primarily due to decreases in men. Regardless of the overall reduction in mortality, death rates of elderly patients on nonsteroidal anti-inflammatory drugs (NSAIDs) have increased.3 PUD is the main cause for upper gastrointestinal (UGI) hemorrhage and other complications. This article is designed to provide background information regarding the disease process and appropriate management of PUD and the eradication of etiologic factors.
Etiology and Pathophysiology
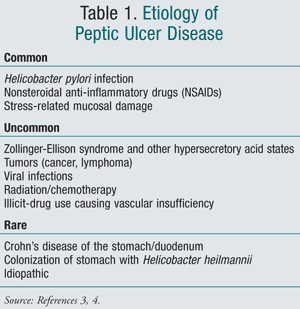
Peptic ulcer disease encompasses gastric, duodenal, and esophageal ulcers, with common etiologies of Helicobacter pylori infection, NSAID use, and stress-related mucosal damage (TABLE 1). Due to the more invasive involvement of the gastric muscularis mucosa, PUD differs from the more superficial acid-related disorders such as erosions and gastritis.3 Peptic ulcers are typically named for their anatomical location, such as gastric or duodenal ulcers, and increased gastric acid is the primary cause. For the purpose of this article, only etiologies of H pylori and NSAID-induced PUD will be discussed. In addition to these etiologies, other controversial environmental risk factors including cigarette smoking, psychological stress, caffeine intake, and alcohol ingestion may increase the risk for the development of PUD.3-5
The disease process of PUD is multifactorial based on etiology and risk factors. Ulcers may occur with hypersecretion of hydrochloric acid and pepsin, causing an imbalance between gastric luminal factors and degradation in the defensive function of the gastric mucosal barrier. Mucosal defenses include mucus, secretion of bicarbonate, mucosal blood flow, and epithelial cell defense. When acid and pepsin invade a weakened area of the mucosal barrier, histamine is released. Histamine will stimulate parietal cells to secrete more acid. With the continuation of this vicious cycle, erosion occurs to form the ulcer.3-5
Although over 50% of the population has chronic H pylori infection, only 5% to 10% develop ulcers.4 H pylori is a pH-sensitive bacterium that can infiltrate the gastric mucosal layer to reside in a neutral-pH environment. Acutely, the infection or colonization may ironically produce a hypochlorhydric environment. It is thought that this protective mechanism for the organism occurs due to the increase of urease, which hydrolyzes urea and converts it to ammonia and carbon dioxide. H pylori contributes to mucosal injury by multiple mechanisms (TABLE 2).4,6

Ulcers induced by nonselective NSAIDs can occur due to a topical irritation of the gastric epithelial cells and reduced protective prostaglandin synthesis.4 Due to their pharmacologic properties, many acidic NSAIDs cause alterations in the hydrophobic mucosal gel layer. The topical irritation may be the first insult to injury; however, inhibition of cyclooxygenase (COX) is the greatest concern. NSAIDs inhibit the rate-limiting enzyme in the conversion of arachidonic acid to prostaglandins. COX-2 exists throughout the body, producing prostaglandins associated with inflammation and pain, whereas COX-1 is located in the stomach, kidney, intestines, and platelets. Isoforms COX-1 and COX-2 are inhibited by nonselective NSAIDs. As a result of COX-1 inhibition, adverse effects such as ulcers or GI bleeds may occur.3-5
Complications
The major complications of H pylori-associated and NSAID-induced ulcers include UGI hemorrhage, gastric or duodenal perforation, stricture at the ulcer site, and gastric outlet obstruction.7 NSAIDs are responsible for a four-fold increased risk of ulcer complications among users compared to nonusers.4 Patients at risk for ulcer complications are those with a history of ulcers or ulcer complications, advanced age, comorbidities, H pylori infection, use of high-dose NSAIDs, and concomitant use of corticosteroids, aspirin, or anticoagulants.4 Elderly patients are particularly susceptible to PUD complications because they may present atypically or with a painless ulcer, and also because NSAID use has risen among this population.5,8
UGI bleeding represents the most common and severe complication of peptic ulcers, with 28% to 59% of all episodes attributed to PUD.7 The mortality rate from peptic ulcer bleeding is 5% to 10% worldwide.9 Peptic ulcer bleeding is managed through supportive care, proton pump inhibitor (PPI) treatment, and endoscopic hemostasis. Surgical interventions are typically reserved for endoscopic treatment failure. Perforation and obstruction related to peptic ulcers occur less frequently than hemorrhage and often require surgical intervention.7
Of concern is the relationship between H pylori and gastric cancer. The International Agency for Research on Cancer and the World Health Organization (WHO) have classified H pylori as a definite carcinogen.10 Thus, treatment and eradication of H pylori should be considered in infected persons.10-12
Clinical Presentation
The most common presenting symptom of patients with PUD is epigastric pain (TABLE 3).4 Stomach pain upon food intake is suggestive of a gastric ulcer, while presentation of a duodenal ulcer includes pain occurring 2 to 5 hours after eating or on an empty stomach and nocturnal pain. It is relieved by food intake, antacids, or antisecretory therapy.4,5 Chronic ulcers may be asymptomatic and are often NSAID-induced, with UGI bleeding or perforation being the first clinical manifestation.4 Patients presenting with alarm symptoms should be suspected of having complications of PUD and referred for endoscopic evaluation.11

Diagnosis
Patients under 45 years of age suspected of having PUD without alarming symptoms should undergo noninvasive testing for H pylori. Endoscopic evaluation should be considered for patients older than 45 years of age and in those presenting with alarming symptoms.11
Noninvasive testing of H pylori includes the urea breath test (UBT), the fecal antigen test (FAT), and antibody testing. The UBT and FAT are the preferred methods for diagnosing an active infection prior to antibiotics and in the absence of PPI administration for 1 to 2 weeks.12 The UBT is the test of choice for confirming eradication and is most accurate at least 4 weeks following completion of treatment. The FAT is often considered unpleasant due to the handling and storage of stool samples and is less validated for posttreatment use as compared to the UBT. Antibody testing is only useful when the result is negative, and confirmation of a positive result is required by performing a UBT or FAT, as the results may remain positive for years after H pylori exposure. Therefore, antibody testing is not recommended to confirm posttreatment eradication.12
In patients who are undergoing endoscopic evaluation for H pylori, the rapid urease test (RUT) is preferred in those who have not taken PPIs within 1 to 2 weeks, or an antibiotic or bismuth within 4 weeks, of the endoscopic examination. Histology is the recommended test in patients who have taken one of the previously mentioned medications prior to endoscopy. Culture and polymerase chain reaction are methods capable of identifying antibiotic sensitivities of H pylori; however, these tests are difficult to perform and are not widely available.12
General Guidelines for Treatment
H pylori Infection
The goal of treating H pylori-infected patients is to eliminate the organism, resulting in greater rates of ulcer healing and lower reoccurrence rates.3 Eliminating H pylori with an antibiotic and adding an anti-secretory agent were found to be more effective than antisecretory therapy alone. Eradication treatment options include triple therapy containing one antisecretory agent and two antibiotics, or quadruple therapy of one antisecretory agent, two antibiotics, and bismuth salt (TABLE 4).13
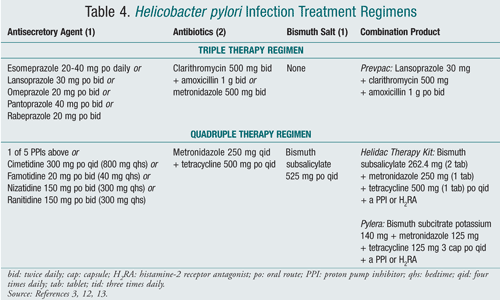
Clarithromycin and amoxicillin are the preferred antibiotics as part of triple therapy (metronidazole is substituted for amoxicillin in patients with a penicillin allergy), while metronidazole and tetracycline are components of quadruple therapy. Triple therapy is the less complex regimen and is usually chosen as first-line treatment. The American College of Gastroenterology recommends both treatment options be continued for 10 to 14 days; 7- and 10-day treatment regimens are equally efficacious, but 14 days is most effective. If a second course of therapy is needed because of treatment failure, the other regimen should be utilized.12
Metronidazole should be chosen instead of amoxicillin as part of triple therapy in patients with penicillin allergies. If the patient has a history of macrolide antibiotic use, quadruple therapy should be used.12 When selecting a PPI, there is not an appreciable difference within the class.14,15 Currently, there are no data to support substitution of ampicillin for amoxicillin, doxycycline for tetracycline, or azithromycin or erythromycin for clarithromycin.12
NSAID-Induced Ulcers
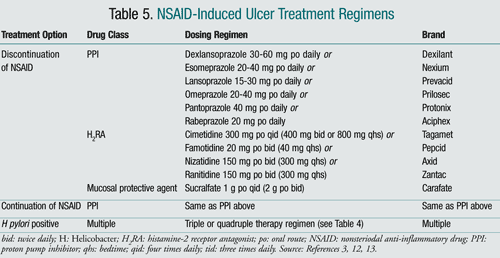
The goals of treating NSAID-induced ulcers are to reduce symptoms and heal the ulcer.3 When treating an NSAID-induced ulcer, the NSAID should be discontinued, if possible, and a histamine-2 receptor antagonist (H2RA), a PPI, or sucralfate should be initiated. PPIs are the optimal choice due to faster relief of symptoms and healing of the ulcer, when compared to H2RAs and sucralfate.16-18 If the NSAID cannot be discontinued, the lowest effective dose and the shortest duration of action should be implemented, and a PPI should be added to the patient's regimen. If the patient is also H pylori positive, eradication therapy should be initiated.6 Patients at high risk for developing NSAID-induced ulcers should be treated concurrently with a PPI or misoprostol to reduce the risk of ulcer development, or be switched to a selective COX-2 inhibitor.19 However, the usefulness of COX-2 inhibitors is limited due to their increased association with myocardial infarction and other thrombotic cardiovascular events. If utilizing a COX-2 inhibitor, the lowest possible dose of celecoxib is recommended to help minimize the risk of cardiovascular events.6
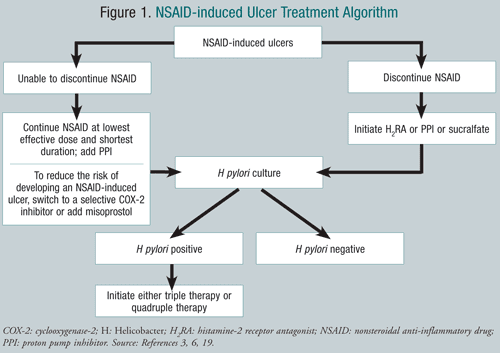
TABLE 5 displays the treatment options and dosing regimens for NSAID-induced ulcers. FIGURE 1 shows an NSAID-induced ulcer treatment algorithm.
Eradication Rates
Peptic ulcers have greater healing rates when NSAIDs are discontinued. When the offending agent is removed, 90% of NSAID-induced ulcers heal with 8 weeks of treatment with H2RAs.4 PPIs have been shown to heal duodenal ulcers after 4 weeks in greater than 93% of patients and to heal gastric ulcers after 8 weeks in 80% to 90% of patients.5,20,21
H pylori eradication rates with first-line triple therapy vary from 70% to 95%.7 Eradication rates are 4% to 5% higher when the treatment duration is increased from 7 days to 10 days or 14 days.22 Clarithromycin resistance has increased in certain geographic regions and should be considered when choosing antimicrobial therapy.10 Bismuth-based quadruple therapy results in eradication rates of 57% to 95%, which is highly dependent on patient adherence.4
Agent Overview
Antisecretory Agents
H2RAs: These agents selectively inhibit the H2 receptors of parietal cells, leading to reduced acid secretion. Reducing acid secretion allows for ulcer healing.23 When comparing H2RAs, all have similar efficacy in reducing acid secretion and promoting ulcer healing. H2RAs are generally well tolerated, with the most common adverse effects being headache, somnolence, dizziness, and diarrhea or constipation. However, cimetidine has an increased risk of causing gynecomastia, impotence, increased serum creatinine, neutropenia, and agranulocytosis, although rare. For all H2RAs, changes in renal function may require dosage adjustment or discontinuation of therapy. Cimetidine has a significant drug interaction profile through inhibition of the CYP450 enzymes 3A4 and 2D6. Cimetidine can potentially increase plasma levels of warfarin, theophylline, phenytoin, quinidine, metronidazole, and dofetilide.13,24
PPIs: There are six FDA-approved PPIs. These agents decrease gastric acid secretions by inhibiting the H+/K+-adenosine triphosphatase pump in the gastric parietal cells, leading to a significant reduction of gastric acid secretion. PPIs also reduce pepsin by increasing the gastric pH, which promotes decreased mucosal damage.25 These agents are generally well tolerated; the most common adverse effects include headache, somnolence, dizziness, diarrhea, constipation, and nausea. PPIs produce elevations in gastric acid pH levels to promote an environment for ulcer healing; a gastric acid pH level greater than 4 has been found to encourage healing.25
Certain medications (i.e., ketoconazole, itraconazole, indinavir, atazanavir, iron salts, mesalamine) are dependent upon the acidic environment of the stomach for absorption and may not be as effective when given in conjunction with an antisecretory agent.25
Antibiotics
H pylori is a gram-negative bacterium. Amoxicillin, clarithromycin, metronidazole, and tetracycline all have antimicrobial activity against H pylori. Amoxicillin is a beta-lactam antibiotic that inhibits bacterial cell wall synthesis, resulting in bactericidal activity against susceptible organisms. Amoxicillin should be avoided in patients with penicillin allergies.26 Amoxicillin can decrease the effectiveness of oral contraceptives; therefore, a back-up method of birth control should be used.
Clarithromycin is a macrolide antibiotic that binds to the 50S ribosomal subunit of susceptible organisms, inhibiting RNA-dependent protein synthesis and resulting in bacteriostatic activity. Serious adverse effects include QTc prolongation, ventricular arrhythmias, and hepatotoxicity.26 Clarithromycin is a potent CYP3A4 inhibitor and can increase plasma levels of warfarin, fluconazole, cyclosporine, digoxin, carbamazepine, and HMG-CoA reductase inhibitors.13,24
Metronidazole is a nitroimidazole antibiotic that disrupts the helical DNA structure and causes strand breakage, leading to inhibition of protein synthesis and cell death of susceptible organisms. It is also considered bactericidal. Serious, but rare, adverse effects include hepatitis, pancreatitis, confusion, seizures, and neuropathy. Patients should avoid alcohol due to the potential for disulfiram-like reactions.26 Metronidazole is a moderate inhibitor of CYP3A4 and may increase plasma levels of warfarin, cyclosporine, lithium, phenytoin, carbamazepine, and amiodarone.13,24
Tetracycline exhibits bacteriostatic activity by binding to the 30S ribosomal subunit of susceptible organisms, thereby inhibiting bacterial protein synthesis. The most common adverse effects are nausea, diarrhea, photosensitivity, and tooth discoloration. Tetracycline is contraindicated in pregnant women and children under 8 years old. Bioavailability is significantly reduced when administered with calcium, iron, antacids, milk, multivitamins, or food. This interaction can be avoided through separation of administration times by at least 2 hours.26 Since tetracycline can decrease the effectiveness of oral contraceptives, a backup method should be implemented.
Mucosal Protective Agents
Bismuth: This agent has multiple ulcer-healing mechanisms, including antibacterial activity against H pylori, inhibition of pepsin activity, and the increase of prostaglandin, mucus, and bicarbonate production. Bismuth does not inhibit acid production.27 The most common adverse effects are blackened tongue and stools.13,24 Caution should be used in the elderly and in patients with renal impairment because of the risk of bismuth accumulation. Bismuth is contraindicated in patients with an aspirin allergy, history of GI bleed, history of coagulopathy, active influenza, or chicken pox infection due to the risk of developing Reye's syndrome.
Misoprostol: This synthetic prostaglandin E1 analogue replaces prostaglandins that are inhibited by NSAIDs and can also induce uterine contractions. Misoprostol inhibits acid secretion and provides mucosal protection.28 For the treatment of NSAID-induced ulcers, it is not as effective as PPIs in ulcer healing rates.29 Misoprostol has been shown to be effective in reducing the risk for NSAID-induced ulcers when administered concurrently with the NSAID.6,19 The most common adverse effects are diarrhea, nausea, headache, and flatulence. Misopros-tol should be administered with meals and at bedtime to reduce the incidence of diarrhea. For the prevention of NSAID-induced ulcers, it should be dosed 200 mcg by mouth four times a day.13,24 Most patients are unable to tolerate the 800 mcg per day dose, due to GI side effects (alternate doses include 100 mcg by mouth four times a day and 200 mcg by mouth two times a day).13,24 The main contraindication is current pregnancy (Pregnancy Category X). If misoprostol is utilized in a female patient in her childbearing years, adequate contraception must be confirmed and a negative pregnancy test should be documented within 2 weeks of initiating treatment. Caution should be used in the elderly and in patients with renal impairment.
Sucralfate: This medication is an aluminum salt from sucrose octasulfate that cannot be absorbed by the body. Sucralfate binds to the gastric mucosa, forming a protective layer that prevents gastric damage from bile salts, pepsin, and peptic acid. This allows healing of the gastric mucosa or the ulcerated tissue.30 The most common adverse effect is constipation. Sucralfate should be taken on an empty stomach to avoid binding with dietary proteins and phosphate. The drug may reduce the plasma levels of fluoroquinolones, phenytoin, digoxin, theophylline, quinidine, warfarin, and ketoconazole.13,24 These medications should be administered at least 2 hours prior to sucralfate. This agent should be used with caution in renal failure patients because of the possibility of aluminum accumulation.
Role of the Pharmacist
Pharmacists play an integral role in the management of patients with PUD. They are in a unique position to provide patient education and management of risk factors associated with peptic ulcer development and recurrence. Patients should be encouraged to abstain from smoking and alcohol consumption, and smoking cessation therapy should be offered when appropriate. Pharmacists can work within the health care team to make recommendations for appropriate NSAID use and recognize patients at risk for ulcer complications. Further, through review of medication histories, pharmacists can identify previous antimicrobial exposure that could increase the likelihood of H pylori resistance. Patients must be made aware that a number of OTC medications and herbal products contain NSAIDs, and that they should consult their health care provider prior to taking these medications. Additional patient education emphasizing the importance of adherence and counseling on proper administration of drug therapy is an essential component for treatment success.
REFERENCES
1. Del Valle J, Chey WD, Schmeiman JM, et al. Acid peptic disorders. In: Yamada T, Aplers DH, Kaplowitz N, et al, eds. Textbook of Gastroenterology. 4th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2003:1321-1376.
2. Helicobacter pylori and peptic ulcer disease. Centers for Disease Control and Prevention.
www.cdc.gov/ulcer/economic.htm
3. Berardi RR, Welage LS. Peptic ulcer disease. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 7th ed. New York, NY: McGraw-Hill; 2008:569-587.
4. Malfertheiner P, Chan FK, McColl KE. Peptic ulcer disease. Lancet. 2009;374:1449-1461.
5. Ramakrishnan K, Salinas RC. Peptic ulcer disease. Am Fam Physician. 2007;76:1005-1012.
6. Lanza FL, Chan FK, Quigley EM; Practice Parameters Committee of the American College of Gastroenterology. Guidelines for prevention of NSAID-related ulcer complications. Am J Gastroenterol. 2009;104:728-738.
7. Napolitano L. Refractory peptic ulcer disease. Gastroenterol Clin North Am. 2009;38:267-288.
8. Thomopoulos KC, Vagenas KA, Vagianos CE, et al. Changes in aetiology and clinical outcome of acute upper gastrointestinal bleeding during the last 15 years. Eur J Gastroenterol Hepatol. 2004;16:177-182.
9. Sung JJ, Tsoi KK, Ma TK, et al. Causes of mortality in patients with peptic ulcer bleeding: a prospective cohort study of 10,428 cases. Am J Gastroenterol. 2010;105:84-89.
10. Suzuki H, Iwasaki E, Hibi T. Helicobacter pylori and gastric cancer. Gastric Cancer. 2009;12:79-87.
11. Dzieniszewski J, Jarosz M. Guidelines in the medical treatment of Helicobacter pylori infection. J Physiol Pharmacol. 2006;57(suppl 3):143-154.
12. Chey WD, Wong BC. American College of Gastroenterology guideline on the management of Helicobacter pylori infection. Am J Gastroenterol. 2007;102:1808-1825.
13. Micromedex Healthcare Series Web site [subscription required]. Version 5.1. Greenwood Village, CO: Thomson Reuters Healthcare; 2010.
www.thomsonhc.com.stlcopisa.
14. Ulmer HJ, Beckerling A, Gatz G. Recent use of proton pump inhibitor-based triple therapies for the eradication of H pylori: a broad data review. Helicobacter. 2003;8:95-104.
15. Vergara M, Vallve M, Gisbert JP, Calvert X. Meta-analysis: comparative efficacy of different proton-pump inhibitors in triple therapy for Helicobacter pylori eradication. Aliment Pharmacol Ther. 2003;18:647-654.
16. Yeomans ND, Tulassay Z, Juhasz L, et al. A comparison of omeprazole with ranitidine for ulcers associated with nonsteroidal anti-inflammatory drugs. Acid Suppression Trial: Ranitidine versus Omeprazole for NSAID-associated Ulcer Treatment (ASTRONAUT) Study Group. N Engl J Med. 1998;338:719-726.
17. Agrawal NM, Campbell DR, Safdi MA, et al. Superiority of lansoprazole vs ranitidine in healing nonsteroidal anti-inflammatory drug-associated gastric ulcers: results of a double-blind, randomized, multi-center study. NSAID-Associated Gastric Ulcer Study Group. Arch Intern Med. 2000;160:1455-1461.
18. Bianchi Porro G, Lazzaroni M, Manzionna G, et al. Omeprazole and sucralfate in the treatment of NSAID-induced gastric and duodenal ulcer. Aliment Pharmacol Ther. 1998;12:355-360.
19. Laine L. Approaches to nonsteroidal anti-inflammatory drug use in the high-risk patient. Gastroenterology. 2001;120:594-606.
20. Vakil N, Fennerty MB. Direct comparative trials of the efficacy of proton pump inhibitors in the management of gastro-oesophageal reflux disease and peptic ulcer disease. Ailment Pharmacol Ther. 2003;18:559-568.
21. Burget DW, Chiverton SG, Hunt RH. Is there an optimal degree of acid suppression for healing of duodenal ulcers? A model of the relationship between ulcer healing and acid suppression. Gastroenterology. 1990;99:345-351.
22. Fuccio L, Minardi ME, Zagari RM, et al. Meta-analysis: duration of first-line proton-pump inhibitor based triple therapy for Helicobacter pylori eradication. Ann Intern Med. 2007;147:553-562.
23. Parsons ME, Ganellin CR. Histamine and its receptors. Br J Pharmacol. 2006;147(suppl 1): S127-S135.
24. Clinical Pharmacology Web site [subscription required]. Tampa, FL: Gold Standard, Inc; 2010.
www.clinicalpharmacology-ip.
25. Horn J. The proton-pump inhibitors: similarities and differences. Clin Ther. 2000;22:266-280.
26. Gallagher JC, MacDougall C. Antibiotics Simplified. Sudbury, MA: Jones and Bartlett Publishers; 2009.
27. Pasricha PJ. Treatment of disorders of bowel motility and water flux; Antiemetics: agents used in biliary and pancreatic disease. In: Brunton LL, Lazo JS, Parker KL, ed. Goodman & Gillman's: The Pharmacological Basis of Therapeutics. 11th ed. New York, NY: McGraw-Hill; 2006:983-1008.
28. Chong YS, Su LL, Arulkumaran S. Misoprostol: a quarter century of use, abuse, and creative misuse. Obstet Gynecol Surg. 2004;59:128-140.
29. Hawkey CJ, Karrasch JA, Szczepanski L, et al. Omeprazole compared with misoprostol for ulcers associated with nonsteroidal anti-inflammatory drugs. Omeprazole versus Misoprostol for NSAID-induced Ulcer Management (OMNIUM) Study Group. N Engl J Med. 1998;338:727-734.
30. Duerksen DR. Stress-related mucosal disease in critically ill patients. Best Pract Res Clin Gastroenterol. 2003;17:327-344.
To comment on this article, contact rdavidson@uspharmacist.com.





