US Pharm. 2013;38(3):HS2-HS6.
ABSTRACT: Patient-controlled analgesia (PCA), defined as a delivery system in which patients self-administer predetermined doses of analgesic medication to relieve their pain, has become a standard form of pain management. IV and epidural PCA are the two most common routes of administration. Advantages of PCA include improved pain relief and greater patient satisfaction. Adverse effects such as respiratory depression, hypotension, and postoperative nausea and vomiting still occur with PCA. Proper patient selection for the use of PCA is imperative, especially among older adults.
Inadequately controlled pain adversely affects a patient’s quality of life and can lead to chronic pain, hampering the recovery process.1 Uncontrolled pain is associated with negative clinical outcomes including delayed hospital discharge, decreased vital capacity and alveolar ventilation, pneumonia, tachycardia, hypertension, myocardial infarction, myocardial ischemia, poor wound healing, and insomnia.2
Patient-controlled analgesia (PCA) is a delivery system in which patients self-administer predetermined doses of analgesic medication to relieve their pain.3 PCA empowers patients to take a proactive role in the management of their pain. Studies have shown that the use of PCA alleviates anxiety and fear surrounding postoperative pain management; offers slightly better pain control; is associated with greater patient satisfaction; and possibly may alleviate the detrimental physiological responses due to pain.4-6 The principles behind PCA involve initially achieving a minimum effective analgesic concentration using a loading dose followed by maintaining a constant plasma concentration, thereby avoiding peaks and troughs.6
The option of PCA is often not offered to older patients because of concern that the elderly may not be able to follow directions for proper use (e.g., due to the presence of cognitive impairment); concern over age-related pharmacokinetic or pharmacodynamic alterations; or concern that older adults might be overly apprehensive about complications associated with opioid use, including addiction.5 While the threshold for the sensation of pain is slightly increased in the elderly, such is not the case for severe postsurgical pain.5 Further, in addition to pain medications possibly inducing delirium, pain itself can result in altered consciousness.5
Routes of PCA Administration
Routes of administration for PCA are numerous and include intravenous (IV), epidural (PCEA), nasal, via the peripheral nerve, and trans-cutaneous/transdermal. IV PCA and PCEA are the two most studied routes of administration, with the former being the route with the most robust body of literature.3 A discussion of routes of administration other than IV PCA and PCEA is beyond the scope of this article.
Elements of a PCA Order
Elements of PCA include an initial loading dose, a demand or bolus dose, a lockout interval (during which time additional analgesic medication cannot be released from the device), an optional background/continuous infusion rate for IV PCA (not PCEA), and an hourly (often 1 or 4 h) maximum administration rate.3,6,7 The purpose of the initial loading dose is to achieve a minimal level of analgesia such that pain assessment scores—i.e. scores on a visual analogue scale—are ≤4 (range 0-10); this may be started while the patient is still in the recovery room. The amount of analgesic available for delivery in the demand or bolus dose needs to be enough to achieve sufficient analgesia while minimizing adverse effects. Use of a bolus dose is optional for peripheral nerve catheter PCA. A typical lockout period is 5 to 10 minutes (preferably the latter to avoid drug accumulation) and should reflect the pharmacokinetic profile of the analgesic.3 The peak effect of most IV opioids is within 7 to 15 minutes with an onset of action of about 5 minutes.8
Although somewhat controversial, the use of a background infusion is intended to provide a constant infusion rate of analgesic delivery whether or not the patient activates the system. Opponents are concerned that patients receiving background infusions will continue to receive medication even while they are sedated; sedation precedes respiratory depression. A background infusion is thought to bypass the inherent safety measure that a sedated patient is unlikely to continue ordering bolus doses. It is best to avoid administration of a background infusion in the elderly because of the concern over respiratory depression. This effect can be compounded by the coadministration of sedative-hypnotics or, in older patients, by the presence of comorbidities (e.g., sleep apnea, renal failure).5,7
Use and lack of use of a background infusion were found to be equally effective in patients undergoing cardiac surgery, although the former group had a greater morphine consumption.9 Opposite dose results were observed in colorectal cancer patients who received a very-low-dose morphine background infusion along with small-dose PCA boluses.10 The use of a background infusion may have a role in the pain management of an opioid-tolerant patient, in which case it would represent their typical daily dose of opioids. The use of an hourly maximal administration rate designed to limit the cumulative doses given, while also controversial, has been viewed as a safety element for PCA.3
Advantages of PCA
Among the advantages of PCA over traditional administration of analgesics, either orally or via intramuscular (IM) injection, include improved pain relief, greater patient satisfaction, less sedation, and possibly fewer postoperative complications. When compared with nurse-controlled analgesia, PCA has been shown to offer slightly better analgesic effects, decreased patient anxiety, and significantly less postoperative pulmonary atelectasis.3,5 PCEA is associated with the most significant improvements in pulmonary complications compared with other forms of analgesia.7
Medications Used for PCA
The opioid most commonly utilized in IV PCA is morphine, although other opioids have often been used as well, including hydromorphone, fentanyl, and meperidine (TABLE 1).3,8,11 PCEA typically employs the use of a small concentration of a long-acting anesthetic such as bupivacaine, levobupivacaine, and ropivacaine plus a lipophilic opioid (e.g., fentanyl or sufentanil).3 For IV PCA in older adults, lower doses may be needed in the elderly due to increased central nervous system (CNS) sensitivity to medications with a centrally mediated mechanism of action. An algorithm on how to adjust IV PCA dosing has been published, which includes morphine or an alternative opioid.6
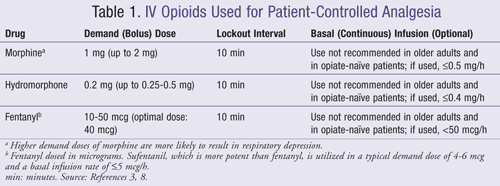
Morphine: Penetration into the blood-brain barrier for morphine may be delayed, resulting in a discrepancy between analgesic effects and respiratory depressive effects. Morphine is metabolized via conjugation with glucuronic acid, and it is eliminated via hepatic and renal sites. Although 75% to 85% of a morphine dose is conjugated to morphine-3-glucuronide (M3G), a therapeutically irrelevant form, 5% to 10% is converted to morphine-6-glucuronide (M6G), which is more potent and has a longer duration of action than the parent compound, morphine. Further, this metabolite is eliminated renally, resulting in accumulation in patients with kidney disease. In a patient with renal impairment, even small doses of morphine may result in unexpected respiratory depression; thus, its use should be avoided in this population. Morphine use is associated with histamine release, which may be troubling to patients.3
The elderly, especially those with underlying pulmonary disease or renal insufficiency, may be especially sensitive to morphine’s respiratory depression. Older adults may experience more intense and prolonged analgesia from morphine due to an altered sensitivity to the drug’s pharmacologic effects (i.e., pharmacodynamic alterations) and altered clearance of the drug (i.e., pharmacokinetic alterations).12 There is great interpatient variability in morphine dosing.5,6
Fentanyl: Fentanyl is a pure opioid receptor agonist that is 75 to 100 times more potent than morphine, has greater lipid solubility resulting in more rapid penetration in the CNS, and has a quicker onset of action compared to morphine. Fentanyl has a short duration of action, as it actively redistributes into inactive tissue such as fat and skeletal muscles. Fentanyl is hepatically metabolized to norfentanyl, which may also possess analgesic properties. Unlike morphine, fentanyl is a good choice in a patient who is experiencing opioid-induced hypotension, as it is not associated with histamine release.3
Sufentanil: Sufentanil, an analogue of fentanyl, is five to 10 times more potent than the latter analgesic; however, it is infrequently used in IV PCA. Sufentanil rapidly penetrates the blood-brain barrier and undergoes extensive first-pass effect. A recommended initial dose for sufentanil is 4 to 6 mcg along with a small background infusion owing to its short half-life.6
Hydromorphone: Hydromorphone is a derivative of morphine that is six to eight times more potent than the parent compound when administered via IV PCA. Since it is metabolized to inactive metabolites, its use may be preferred in patients with renal impairment or those who are intolerant to morphine. It can produce a transient hyperglycemia.3
Local Anesthetics (Bupivacaine and Ropivacaine): Bupivacaine is administered in concentrations of 0.05% to 0.175%, and ropivacaine is utilized in strengths of 0.15% to 0.20% in the elderly. Since there is little to no absorption of drug in the epidural space, plasma concentrations are dependent on the rate of absorption from the site of administration, volume of distribution of the drug, and plasma clearance. Continuous epidural infusion of anesthetics may result in neurotoxicity, depending on the dose. Whereas excessive administration of opioids via the epidural route can result in respiratory depression, the overuse of local anesthetics can cause hypotension and/or motor block; this is more common with a continuous epidural infusion compared with PCEA.5
Meperidine: Meperidine is a synthetic opioid that has also been used for IV PCA; however, its use is not recommended, especially in the elderly. The drug has mild anticholinergic effects, a very short duration of action, and renal elimination, and is associated with significant CNS toxicity including confusion, anxiety, nervousness, hallucinations, and seizures. Meperidine use is contraindicated in patients with renal or hepatic dysfunction, in those with a seizure disorder, or in patients taking a monoamine oxidase inhibitor (MAOI).3,8
For PCEA, a typical bolus dose of a local anesthetic is 2 to 4 mL with a lockout interval of 10 to 20 minutes. For nonelderly patients, a typical continuous infusion rate is 3 to 10 mL/h, with a slower rate and smaller boluses utilized in older adults.6
Adjunctive Medications for PCA
Concern over opioid side effects has resulted in a search for the use of adjuvant agents to help improve analgesia while minimizing opioid-related adverse events. Among the adjuvant agents that have been used along with PCA include ketamine, gabapentin, pregabalin, dexmedetomidine, nonsteroidal anti-inflammatory drugs (NSAIDs), naloxone, tapentadol, lidocaine, and clonidine.2,3 Discussion of adjunctive pain medications is beyond the scope of this article.
IV PCA and PCEA
When comparing IV PCA with an opioid versus PCEA with a local anesthetic and opioid, research has shown that the latter offers superior pain relief and that this benefit is particularly apparent in either high-risk patients (e.g., the elderly)5 or those undergoing higher-risk procedures (e.g., thoracic or thoracoabdominal surgery).13 Additional benefits of PCEA include promotion of early mobilization, improvement of bowel function, shortening of the duration of hospitalization, less sedation, less nausea, and a reduction in cardiovascular morbidity compared with IV PCA. Another possible advantage of PCEA over IV PCA is that PCEA may provide for a more rapid recovery of mental status in older surgical patients with delirium. However, the literature on the use of PCEA in the elderly is not as robust as that for IV PCA.5
The combination of local anesthetic/opioid PCEA has been found to provide better postoperative pain relief compared to epidural or systemic opioid monotherapy. PCEA has also been shown to improve patient satisfaction and safety compared to bolus administration of opioids. In patients undergoing colonic resection, those receiving continuous PCEA compared to those getting IV PCA had increased functional exercise capacity, improved health-related quality of life, and earlier restoration of gastrointestinal function, mobilization, and discharge.3 Further, the use of PCEA as opposed to IV PCA was associated with a reduction in all-cause mortality after rectal cancer surgery.14 In patients undergoing spinal fusion surgery, those who received PCEA has superior pain relief and higher patient satisfaction compared to those who had received IV PCA.3 PCEA is contraindicated in patients with thrombocytopenia, coagulopathy, or anticoagulation.
Disadvantages of PCA
Drug Misadventures: While the use of PCA may offer advantages, there are problems specifically associated with this form of drug administration. Problems with the use of PCA involve both human factors and device malfunction issues. Improper patient selection, inadequate patient monitoring, programming errors, lack of knowledge by the team administering the PCA, drug-interactions, patients’ use of their own home medications, mistaking the call bell for the bolus PCA button, and technical problems are some of the issues that complicate PCA therapy. Additionally, problems with medical devices often go unreported.15 For this reason, it is important to conduct a failure modes and effects analysis to try to prevent problem scenarios.
A U.S. Pharmacopeia analysis of data from July 2000 to June 2005 revealed that 1% of medication errors (9,571 out of a total of 919,241) involved PCA. Of these, 6.5% were associated with patient harm. When age was analyzed, 34.7% of PCA-related adverse events were in patients aged 60 years or older; of these, 36.4% resulted in harm. This is particularly troubling given the vulnerability of this age group.16
One of the greatest human factors associated with adverse effects is unauthorized administration of IV bolus doses of an analgesic by well-meaning family, friends, or hospital staff, which is known as PCA by proxy. At least 15 cases of harm or death have resulted from PCA by proxy due to respiratory depression. Family members of patients receiving PCA must be educated about this risk.17
Adverse Drug Events: Typical opioid-induced side effects such as respiratory depression, sedation, confusion, nausea, vomiting, pruritus, and urinary retention are observed with PCA. Respiratory depression is one of the adverse effects that generates the most concern by health care professionals administering narcotics to the elderly. When examining the role that PCA has in inducing respiratory depression, the literature is plagued with methodological issues such as variations in the criteria defining respiratory depression. Some studies have defined respiratory depression using respiratory rate, depth, and rhythm; oxygen saturation via pulse oximetry; or need for an opioid antagonist.3,6 Risk factors for respiratory depression associated with IV PCA include the use of a background infusion; health professional versus patient-controlled analgesia; advanced age; head injury; hypovolemia; use of hypnotics or sedatives; renal, hepatic, pulmonary (e.g., pneumonia, chronic obstructive pulmonary disease) or cardiac impairment; sleep apnea; and obesity.3,6 However, tolerance does develop to opioid-induced respiratory depression, usually within a few days.18
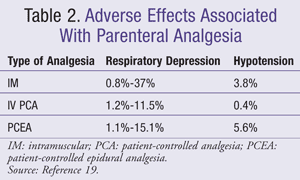
TABLE 2 compares adverse effects associated with IM, IV PCA, and PCEA.19
Postoperative nausea and vomiting is the most common adverse event
associated with IV PCA, which is related to the anesthetic utilized, the
patient’s anxiety level, and the degree of pain experienced.3
The elderly are at greater risk of developing sedation and confusion because of age-related decline in renal function and possible underlying cognitive impairment. In patients with renal impairment, the use of morphine and, most especially, meperidine should be avoided. Twenty-three percent of patients being treated for postoperative pain develop urinary retention, with the highest incidence among those receiving epidural analgesia.3 Risk factors for postoperative urinary retention include male gender, advanced age, and the use of PCA. Both IV PCA and PCEA can increase the risk of infection. Compared with IV PCA, PCEA may be associated with neurologic adverse events. PCEA can result in epidural hematomas, and it appears to offer no advantage in patients undergoing elective cardiac surgery with respect to hospital length of stay, quality of recovery, or morbidity compared with IV PCA. Patients receiving PCEA need to be closely monitored for motor blockade, a condition that may be particularly problematic in the elderly.3
Risk Factors: Patients with cognitive impairment or paralysis, those who are comatose, head trauma victims, those with severe learning difficulties, patients with severely impaired manual dexterity, those with a history of substance abuse, or very young patients (e.g., infants) are not suitable candidates for the use of PCA.3,7,20 Special caution is needed when PCA is used in elderly or frail patients, those with respiratory disease, those with sleep apnea, and the morbidly obese.15 PCA may also mask serious postsurgical complications such as urinary retention, compartment syndrome, pulmonary embolism, and myocardial infarction, although one may postulate that oversedation with more conventional forms of opioid administration may have a similar or worse effect.7
Strategies for Safe PCA Use
Everyone involved in the provision of PCA has a role to play to ensure its safety, including the patient, family and friends, and the health care staff. General educational strategies should include these key players. A detailed, bullet-point discussion on this topic has been published.21 This guide lists safety measures that can be implemented during every step of the PCA utilization process, including the purchase of the pump, actions to be taken prior to prescribing and dispensing PCA, prescribing PCA, dispensing PCA, initiating PCA, and monitoring its effects.21 Criteria for the proper use of PCA should be established, with standardized order forms utilized. Competency assessments for those involved with the administration of PCA should be held on a regular basis.21
Conclusion
While the use of PCA in older adults offers greater autonomy and participation in one’s health care decisions, patient selection is of utmost importance. PCA is not a “one size fits all” or “set it and forget it” intervention. For elderly patients with conditions that profoundly affect cognition or manual dexterity or in those with severely compromised renal or pulmonary function, safer alternatives may be available. Institutions should conduct failure modes and effects analyses before implementing PCA in order to avoid the potential pitfalls with its use.
REFERENCES
1. Lehmann KA. Recent developments in patient-controlled analgesia. J Pain Symptom Manage. 2005;29(suppl):S72-S89.
2. Vadivelu N, Mitra S, Narayan D. Recent advances in postoperative pain management. Yale J Biol Med. 2010;83:11-25.
3. Momeni M, Crucitti M, De Kock M. Patient-controlled analgesia in the management of postoperative pain. Drugs. 2006;66:2321-2337.
4. Hudcova J, McNicol ED, Quah CS, et al.
Patient-controlled opioid analgesia versus conventional opioid analgesia
for controlling postoperative pain. Cochrane Summaries.
http://summaries.cochrane.org/CD003348/patient-controlled-opioid-analgesia-versus-conventional-opioid-analgesia-for-controlling-postoperative-pain.
Accessed November 6, 2012.
5. Mann C, Pouzeratte Y, Eledjam JJ. Postoperative
patient-controlled analgesia in the elderly—risks and benefits of
epidural versus intravenous administration. Drugs Aging. 2003;20:337-345.
6. Grass JA. Patient-controlled analgesia. Anesth Analg. 2005;101(suppl):S44-S61.
7. Macintyre PE. Safety and efficacy of patient-controlled analgesia. Br J Anaesth. 2001;87:36-46.
8. Craft J. Patient-controlled analgesia: is it worth the painful prescribing process? Bayl Univ Med Cent Proc. 2010;23:434-438.
9. Mota FA, Marcolan JF, Pereira MH, et al. Comparison
study of two different patient-controlled anesthesia regimens after
heart surgery. Rev Bras Cir Cardiovasc. 2010;25:38-44.
10. White I, Ghinea R, Avital S, et al. Morphine at
“sub-analgesic” background infusion rate plus low-dose PCA bolus
controls pain better and is as safe as twice a bolus-only PCA regimen: a
randomized, double-blind study. Pharmacol Res. 2012;66:185-191.
11. Hutchinson RW, Anastassopoulos K, Vallow S, et al.
Intravenous patient-controlled analgesia pump and reservoir logistics:
results from multicenter questionnaire. Hosp Pharm. 2007;42:1036-1044.
12. Janora DM, Jermyn RT, Surve SR. Pharmacologic management of pain in older adults. Clin Geriatr. 2010;18:23-32.
13. Ali M, Winter DC, Hanly AM, et al. Prospective,
randomized, controlled trial of thoracic epidural or patient-controlled
analgesia on perioperative quality of life. Br J Anaesth. 2010;104:292-297.
14. Gupta A, Bjornsson A, Fredriksson M, et al. Reduction
in mortality after epidural anaesthesia and analgesia in patients
undergoing rectal but not colonic cancer surgery: a retrospective
analysis of data from 655 patients in Central Sweden. Br J Anaesth. 2011;107:164-170.
15. Doyle DJ. PCA overdose. AHRQ. WebM&M. www.webmm.ahrq.gov/case.aspx?caseID=103. Accessed December 1, 2012.
16. Hicks RW, Sikirica V, Nelson W, et al. Medication errors involving patient-controlled analgesia. Am J Health Syst Pharm. 2008;65:429-440.
17. Pasero C, McCaffery M. Authorized and unauthorized use of PCA pumps. Am J Nurs. 2005;105:30-32.
18. Weber LM, Ghafoor VL, Phelps P. Implementation of standard order sets for patient-controlled analgesia. Am J Health Syst Pharm. 2008;65:1184-1191.
19. Cashman JN, Dolin SJ. Respiratory and haemodynamic
effects of acute postoperative pain management: evidence from published
data. Br J Anaesth. 2004;93:21-23.
20. Patient-controlled analgesia. Enotes. www.enotes.com/patient-controlled-analgesia-reference/patient-controlled-analgesia-172268. Accessed November 15, 2012.
21. Grissinger M. Safety and patient-controlled analgesia. Part 2: How to prevent errors. P T. 2008;33:8-9.
To comment on this article, contact rdavidson@uspharmacist.com.





