US Pharm. 2010;35(4)(OTC Trends suppl):9-12.
Any community pharmacist most likely will agree that self-care in the United States continues to be a growing trend. Patient purchases of OTC products have consistently risen, for many reasons. The convenience of treating a condition with an OTC product is appealing to the typically busy patient, who seeks to avoid a lengthy and expensive office visit in favor of meeting the demands of home and work. In addition, patients in the information age have an increased level of awareness about health. Using Internet resources, patients prefer to diagnose and treat themselves at home with the click of a button. Households are inundated with direct-to-consumer advertising, which empowers the average American to “find out if this product is right for you.” Finally, rising health care costs and a growing number of underinsured or uninsured patients have contributed to the increased use of OTC products. During this time of strain on the health care system and uncertainty about health care reform, patients prefer to avoid paying physicians’ fees, traveling to physicians’ offices, and missing work in favor of using an inexpensive OTC product.
The OTC Market
The sale of OTC products constitutes a huge market in the U.S. According to the Consumer Healthcare Products Association (CHPA), U.S. retail sales of OTC medications in 2008 totaled $16.8 billion, excluding Walmart sales.1 TABLE 1 illustrates the growing number of OTC sales by category from 2005 through 2008.2 Approximately 1,000 active ingredients are used in more than 100,000 OTC products available in the U.S. marketplace. Eighty-four ingredients, dosages, or indications have made the switch from prescription to OTC status since 1976. Eighty-two percent of U.S. women and 71% of U.S. men have used an OTC product in the last 6 months to treat at least one common health ailment.3 In addition, 18.9% of Americans have reported taking one or more dietary supplements in the past year, and 37.2 million Americans have stated that they use herbal remedies regularly. The top-selling herbal supplements in the U.S. in 2000 were garlic, ginkgo biloba, and glucosamine.4
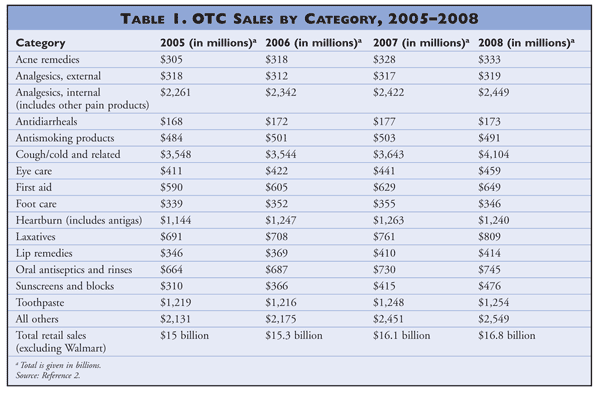
While the wide selection of OTC products may be a blessing for many patients, it also is daunting. Products containing multiple active ingredients, along with line extensions of popular brands, make the OTC marketplace confusing for patients and increase the risk of therapeutic duplication, drug–disease and drug–drug interactions, and accidental misuse. It is no wonder that a study conducted in 2003 for the National Council on Patient Information and Education (NCPIE) found that 66% (n = 1,009) of Americans believe that selecting an OTC medication can be a challenge because of the wide range of competing products available.5
OTC Effects on the Health Care System
The use of OTC products has had a positive effect on the health care system. The self-care movement will continue to be invaluable for future legislation and health care reform, for it is the “foundation of our health care system” from a public-health and economic standpoint.6 One estimate suggests that if only 2% of self-treating patients in the U.S. visited primary care practitioners rather than managing the condition on their own, physician office visits would increase by 62%. This increase in office visits would overwhelm the health care system! The CHPA Web site offers 1997 statistics showing that every dollar spent on nonprescription medications provides $2.47 in health care benefits.3 These savings benefit the health care system as a whole and are especially advantageous for Medicare Part D beneficiaries, underinsured and uninsured patients, and participants in flexible spending accounts who can use pretax dollars to purchase OTC products.
Patient Understanding, Beliefs, and Behaviors About OTC Use
To be considered for OTC status, medications must meet the following FDA criteria7:
• Their benefits outweigh their risks.
• The potential for misuse and abuse is low.
• The patient can use them for self-diagnosed conditions.
• They can be adequately labeled.
• Health practitioners are not needed for the safe and effective use of the product.
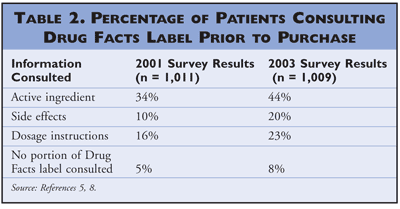
Since May 2002, a standardized Drug Facts label has been required to appear on all nonprescription medications to guide patients about safe and effective use. This label was developed to rectify problems associated with small type and the use of technical language on previous package labeling. The National Opinion Surveys, conducted by NCPIE in 2001 and 2003 (n = 1,011 and 1,009, respectively), sought to determine how well consumers understood and processed essential information about OTC products both before and after the standardized label was enforced.5,8 The results demonstrated progress in safe and effective use of OTC products following the introduction of standardized labels, but there is still room for improvement.
Just more than half (56%) of respondents were aware of the Drug Facts label on OTC products.5,8 More respondents in 2003 paid attention to active ingredients, side effects, and dosage instructions on the Drug Facts label compared with respondents in 2001, but 8% still did not read anything on the Drug Facts label when selecting an OTC product (TABLE 2). When asked about dosing of OTC products, 30% and 48% of respondents in 2001 and 2003, respectively, admitted to taking more than the recommended dose of products (TABLE 3).
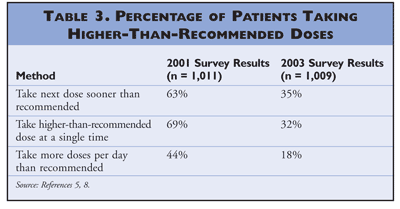
The risk of drug–drug interactions is a concern with use of OTC products. In 2001, 36% of respondents said that they were likely to combine OTC products when they had multiple symptoms (e.g., headache and sore throat).5,8 In addition, in 2003, more than half of respondents (51%) reported having taken an OTC medication and a prescription drug simultaneously.
It is interesting to learn where patients obtain information about OTC products outside of the Drug Facts label. According to a 2001 Roper Starch survey, 27% of patients get their information from friends and family, 20% from a physician, 7% from the Internet, and 7% from a pharmacist.9 Just under half of participants in the 2003 NCPIE survey (43%) said that they consult a pharmacist when buying an OTC product.5 In the same survey, 80% of respondents stated that they would buy a particular OTC product based on their pharmacist’s recommendation, and 82% would not purchase a specific OTC product if their pharmacist advised against it.
The Pharmacist’s Role
In order for the benefits to outweigh the risks of OTC product use, patients must be informed about these products. The benefits of self-treatment include financial advantages for patients and third-party payers; a lightened workload for a tired health care system; convenience; and the wide variety of OTC products available. The risks of self-treatment include the potential for drug–drug and drug–disease interactions; the overuse or misuse of OTC products; incorrect diagnosis of the self-care condition; misinterpretation of the Drug Facts label; and patient confusion caused by combination products and brand line extensions.
The pharmacist plays a key role in ensuring that the balance of OTC use tips toward benefit rather than risk. Who better to serve as the “learned intermediary” to help patients understand and apply health information?6 The pharmacist is available at the point of purchase to help the patient select an appropriate OTC product, suggest nonpharmacologic therapy, or refer the patient to a physician when necessary. Pharmacists are trained to help patients determine when and how to treat themselves; when to see a physician; when diagnostic tests are appropriate; and when to use emergency health care services. As an extension of pharmaceutical care, pharmacists can counsel patients on the appropriate use of OTC products, thereby maximizing the benefits and reducing the risks.
When pharmacists take the time to counsel patients about OTC products, the results are significant. In a 1996 study, fourth-year pharmacy students interacted with 745 patients in community pharmacies in Washington State.10 Consultations averaged 4.6 minutes in length. Following consultation, 42.6% of patients changed their intended purchase, 8% made no purchase, 4.3% were referred to a physician, and 7.1% avoided a potential adverse effect (drug–disease interaction, drug–drug interaction, additive side effects, duplication of therapy). More than half of patients (51%) said that they would be willing to pay $1 or more for an OTC consultation in the future, and 28% would pay $5 or more. Three-quarters of patients (75%) were willing to spend 1 to 5 minutes with a pharmacist to discuss OTC product use.
Pharmacists’ impact on the safe and effective use of OTC products is invaluable. To encourage pharmacists to be more involved in self-care and to feel more comfortable making recommendations, the American Pharmacists Association developed a certificate training program entitled OTC Advisor: Advancing Patient Self-Care.6 This program was designed to “educate pharmacists…about the safe and effective use of nonprescription medications.” Armed with this knowledge and a stepwise approach to self-care, pharmacists can help patients make “appropriate decisions and achieve optimal outcomes from nonprescription therapy.”6 Regardless of the means, all community pharmacists should become familiar with evidence-based knowledge of self-care medications and seize all opportunities to counsel patients regarding the selection and use of OTC products.
QuEST Process: A Stepwise Approach
OTC product consultations between pharmacists and patients take place in various settings throughout the pharmacy: the drive-through window, the register, the OTC aisle, the consultation room, or the telephone. Because these encounters sometimes can be disjointed and rushed, it is important to have a stepwise approach for assessing patients’ self-care needs and developing appropriate recommendations. The QuEST process offers the pharmacist a quick guideline for assessing a patient’s condition systematically and completely. The mnemonic QuEST means: Quickly and accurately assess the patient; Establish that the patient is an appropriate self-care candidate; Suggest appropriate self-care strategies to the patient; Talk with the patient about these strategies.
Another helpful mnemonic, SCHOLAR-MAC, also assists with the first step of assessing the patient: Symptoms; Characteristics of symptoms; History of symptoms; Onset; Location; Aggravating factors; Remitting factors; Medications (prescription, OTC, herbal, dietary supplements); Allergies; Conditions (medical).
After thoroughly exploring the patient’s symptoms and ruling out exclusions to self-care, the pharmacist can recommend appropriate OTC products and provide counseling on the safe and effective use of the products chosen. The last phase of this process is documentation and follow-up. Follow-up may occur formally or informally.
It is important for the pharmacist to use effective questioning skills. The encounter should begin with broad, open-ended questions in order to gain as much information as possible. Closed-ended questions may be used to clarify important points. It is important to avoid leading questions, which may come across as judgmental or condescending and elicit false information. The pharmacist should also use active listening skills. Minimizing distractions, deliberately using nonverbal cues, summarizing the interaction, and empathizing with the patient are essential for establishing a therapeutic alliance and gaining trust. Effective questioning and listening skills are essential to the QuEST process, which offers pharmacists a quick method of thoroughly assessing patients before recommending an OTC product.
Use of OTC Products in Special Populations
While pharmacists should endeavor to provide counseling to all patients regarding OTC use, the following populations require special attention and care when it comes to self-care decisions.
Pediatric Patients: Many parents and caregivers depend on OTC products to treat common childhood illnesses. Unfortunately, many parents and caregivers are unfamiliar with the dosing and administration of these products. The 2001 NCPIE survey determined that only 21% of consumers were aware that a child’s weight is the most accurate guide for determining the correct dose.8 A 1997 study found that only 30% of parents and caregivers were able to correctly determine and measure the weight-based dose for their child.11 This places a large responsibility on pharmacists to ensure safe and effective dosing in pediatric patients. The pharmacist should encourage parents and caregivers to know their child’s weight and counsel them regarding the use of appropriate dosing devices (e.g., syringes, calibrated medicine cups). The pharmacist can calculate the appropriate weight-based dose and mark the dose on a dosing device for the parent or caregiver. In addition, under the direction of a primary care physician, the pharmacist can recommend doses and products for children under the age of 2 years, an age group for which the FDA advises against using OTC products without supervision.
Geriatric Patients: Adults aged 65 years and older consume 33% of all OTC medicines sold in the U.S.3 In addition, older adults are at increased risk for serious drug-related problems. Through patient education, pharmacists play a key role in preventing interactions between OTC products and concurrent medical conditions and medications. Age also alters the absorption, distribution, metabolism, and elimination of certain drugs, which increases susceptibility to therapeutic effects and adverse effects. The pharmacist should counsel geriatric patients about increased susceptibility to adverse effects. Because polypharmacy often plagues this population, the pharmacist should consider whether the patient is seeking treatment for an adverse reaction from another medication.
Pregnant and Breastfeeding Women: The Drug Facts label on most OTC products directs pregnant and breastfeeding women to “ask a health professional before use.” Therefore, the pharmacist should be knowledgeable about the use of these agents in each trimester of pregnancy and in lactating women. Whenever possible, the pharmacist should recommend nonpharmacologic therapy, owing to the ability of most medications to cross the placenta and transfer into breast milk to some extent. Additionally, the pharmacist should identify which trimester of pregnancy a woman is in and recommend older products with an established record of safe use in pregnancy. Topical or local dosage forms are also a safer recommendation for pregnant and lactating women.
Conclusion
Patients’ use of OTC products carries both benefits and risks. When pharmacists proactively counsel patients on OTC product selection and use, the risks virtually disappear. As the economic downturn continues and questions about health care reform remain, more patients will seek ways to self-treat and will rely on pharmacists for health information. Pharmacists should embrace the opportunity (and challenge) to counsel every patient on the safe and effective use of OTC products. The benefits for the individual patient, the pharmacy profession, and the health care system are too great to miss out on!
REFERENCES
1. Consumer Healthcare Products Association. OTC retail sales—1964-2008. www.chpa-info.org/pressroom/Retail_Sales.aspx. Accessed January 18, 2010.
2. Consumer Healthcare Products Association. OTC sales by category—2005-2008. www.chpa-info.org/pressroom/Sales_Category.aspx. Accessed January 18, 2010.
3. Consumer Healthcare Products Association. OTC facts and figures. www.chpa-info.org/pressroom/OTC_FactsFigures.aspx. Accessed January 18, 2010.
4. Consumer Healthcare Products Association. Dietary supplement facts and figures. www.chpa-info.org/pressroom/DS_FactsFigures.aspx. Accessed January 18, 2010.
5. Uses and attitudes about taking over-the-counter medicines: findings of a 2003 national opinion survey conducted for the National Council on Patient Information and Education. www.bemedwise.org/survey/summary_survey_findings.pdf. Accessed January 18, 2010.
6. American Pharmacists Association. OTC advisor: advancing patient self-care. The pharmacist’s role in self-care. www.pharmacist.com. Accessed January 18, 2010.
7. Office of Nonprescription Products. www.fda.gov/AboutFDA/CentersOffices/cder/ucm093452.htm. Accessed January 18, 2010.
8. Attitudes and beliefs about the use of over-the-counter medicines: a dose of reality. www.bemedwise.org/survey/final_survey.pdf. Accessed January 18, 2010.
9. Roper Starch Worldwide. Self-care in the new millennium; March 2001.
10. Sclar DA, Robison LM, Skaer TL. Pharmacy consultation and over-the-counter medication purchasing outcomes. Over-the-Counter Medication Intervention Project Team. J Clin Pharm Ther. 1996;21:177-184.
11. Simon HK, Weinkle DA. Over-the-counter medications. Do parents give what they intend to give? Arch Pediatr Adolesc Med. 1997;151:654-656.
To comment on this article, contact rdavidson@uspharmacist.com.






