US Pharm. 2012;37(8):56-59.
In the use of medications—particularly antibiotics—in pediatric patients, it is imperative to remember that pharmacokinetic and pharmacodynamic processes may be different in children compared with adults. These physiological changes can result in unfavorable outcomes for the pediatric patient. This article aims to outline those changes, focusing on the use of two antibiotic classes historically contraindicated in children: fluoroquinolones and tetracyclines.
Physiological Changes in Pediatric Patients
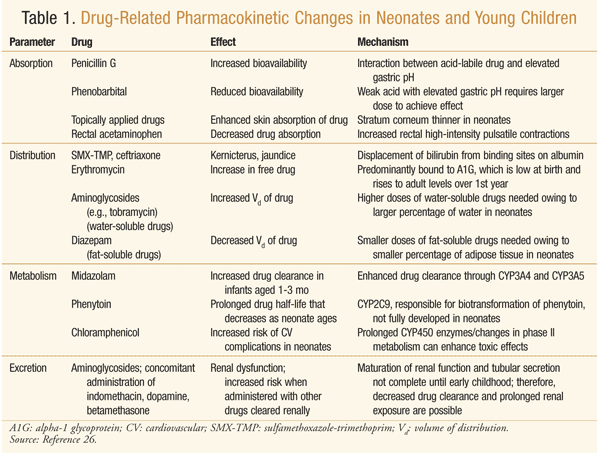
Throughout childhood, significant changes in the absorption, distribution, metabolism, and excretion of medications take place, thereby necessitating age-dependent dosage adjustments.1 Drug absorption is largely affected by developmental transformations, such as changes in gastrointestinal pH, or by lung development, in the case of inhaled antibiotics.1 Reduced protein binding can result in an increased half-life for certain antibiotics. Changes in the expression of individual CYP450 enzymes, phase II metabolism, and renal function significantly alter the metabolism and plasma clearance of numerous antibiotics.1-3 Specific examples of pharmacokinetic changes and their effects on various medications are given in TABLE 1. Although numerous pharmacokinetic differences in pediatric patients are known, little information has been published concerning pharmacodynamic changes.
Fluoroquinolones
Because of their excellent tissue penetration and broad spectrum of activity, fluoroquinolones have become a staple of antimicrobial pharmacotherapy for adult patients. The first fluoroquinolone, nalidixic acid, was introduced in 1964 and for the last two decades has been approved for the treatment of urinary tract infections (UTIs) in children aged 3 months and older; however, it is rarely used.4,5 The more commonly used fluoroquinolones ciprofloxacin and levofloxacin were approved for use in children solely for the postexposure treatment of inhalation anthrax prior to 2004.4,5 In 2004, ciprofloxacin gained approval for treatment of complicated UTIs and pyelonephritis in children aged 1 to 17 years.5 Currently, moxifloxacin is not approved for pediatric use.5 Although the last decade has seen increasing use of fluoroquinolones in the pediatric population, significant controversy exists.
Controversy: Fluoroquinolones have demonstrated adverse effects on cartilage development in juvenile animals through the inflammation and destruction of weight-bearing joints.4,5 These arthropathies were often irreversible, and their potential occurrence in children limited the use of fluoroquinolones in this population.4 In one pediatric study, ciprofloxacin had a 3.3% (9.3% vs. 6.0%) absolute risk increase in musculoskeletal events within 6 weeks of treatment compared with control agents used to treat complicated UTIs or pyelonephritis.4 Adefurin and colleagues found a 57% increased relative risk of arthropathy in children given ciprofloxacin (21% overall) versus those in a nonfluoroquinolone comparator arm.6 In contrast to animal models, neither dose nor duration had an effect on the rate or severity of arthropathy.6 A 2007 study by Noel and colleagues determined the incidence of musculoskeletal events (primarily arthralgias) to be greater in children treated with levofloxacin compared with nonfluoroquinolone-treated children at 2 months (2.1% vs. 0.9%; P = .04) and 12 months (3.4% vs. 1.8%; P = .03).7 These results and the severity of the effects should be weighed heavily when initiation of fluoroquinolones is being contemplated in pediatric patients.
Another significant concern with fluoroquinolone use is increased bacterial resistance. Resistance patterns to fluoroquinolones, which have consistently worsened in adults over the last 20 years, can occur via multiple mechanisms, such as changes in target enzymes (DNA gyrase and DNA topoisomerase) or the expression of efflux pumps.4,5,8,9 The synthesis of newer fluoroquinolones, such as moxifloxacin, created expectations that resistance to gram-positive bacteria would be limited. However, resistance is often considered a class effect and may become a significant concern in the pediatric population with continued use.
Potential Benefits: Fluoroquinolones have been used successfully in a variety of pediatric infections, including cystic fibrosis exacerbations, complicated UTIs, and otitis media.4 Use in children with neutropenic fever or multidrug-resistant gram-negative infections has also increased.4 However, most of the literature supporting the use of fluoroquinolones in children comes from retrospective or small, uncontrolled studies.
Evidence of fluoroquinolone-induced arthropathies in pediatric patients is well documented, but no cases of clinician-diagnosed cartilage destruction in children have been noted in either controlled clinical trials or unsolicited FDA reports.5 Additionally, one retrospective study involving more than 20,000 children demonstrated no significant difference in joint or tendon disorders with fluoroquinolones versus azithromycin.10 Although fluoroquinolones carry a black box warning regarding tendon rupture, there are no published reports of this adverse event in children.5,11
Regarding fluoroquinolone resistance, the overall resistance pattern in pediatric gram-negative isolates (excluding cystic fibrosis patients) has been documented at less than 5%.12 However, it is likely that resistance rates will rise with increased pediatric fluoroquinolone use.8
Tetracyclines: Tetracyclines are a class of broad-spectrum antibiotics with activity against gram-positive, gram-negative, anaerobic, acid-fast (atypical), and Rickettsia bacteria.13 Useful for numerous infections, tetracyclines were first introduced in 1948, with initial concerns of tooth discoloration in children being reported in 1956.14 This adverse event is most likely during the tooth-calcification process, which is completed by age 8 years.15 Other adverse events include light hypersensitivity, gastrointestinal effects, and rare hepatotoxicity.13 Despite these adverse events, tetracyclines have been successfully used for respiratory infections, community-acquired methicillin-resistant Staphylococcus aureus, malaria, and acne in patients aged 8 years and older. However, tetracyclines are relatively contraindicated in children aged less than 8 years, and their use in this age group is examined in the following section.
Controversy: The routine use of tetracyclines in children aged less than 8 years is not recommended because of a long-standing association of these agents with permanent tooth discoloration. Tetracyclines, which bind to cations, can result in the formation of tetracycline–calcium complexes that irreversibly deposit in developing bones and teeth.16 Once calcification is complete, no further calcium exchange occurs, which limits tetracycline’s ability to deposit in the teeth.15 The degree of tetracycline exposure, number of courses, total dosage, and exact timing of tooth development may affect the risk of occurrence.15-17 Staining may vary by agent, with doxycycline potentially having the lowest incidence owing to its decreased affinity for calcium binding compared with other tetracyclines.16,18 Volovitz and colleagues reported no cases of tooth discoloration in a study of 31 children (mean age 4 years) treated with doxycycline for atypical pneumonia.19
Tetracycline-induced photosensitivity, another adverse event in the pediatric population, usually manifests as a light-sensitive rash similar to an exaggerated sunburn. This occurs because of the extensive absorption of ultraviolet light by the tetracyclines.20 In a study of 106 adults and children treated for acne, Layton and colleagues reported a 36% incidence of light-sensitive rash from doxycycline, with increased risk at higher doses (>150 mg/day) and in subjects with lighter complexions.21
While phototoxicity can be a significant concern, the permanent nature of tooth discoloration underscores the importance of avoiding the use of these agents in young children whenever possible. However, when tetracycline use is imperative, limited evidence suggests that minimizing the total dose and length of exposure and using a tetracycline with decreased calcium binding may reduce the risk of tooth discoloration.
Potential Benefits: In certain difficult-to-treat infections with limited alternative therapies, tetracycline may be efficacious and relatively safe in children aged less than 8 years. Tetracyclines are useful for the treatment of both Rocky Mountain spotted fever (RMSF) and ehrlichiosis, a separate rickettsial disease that can mimic RMSF and should be empirically covered whenever RMSF is suspected.22 Additionally, a retrospective study suggested that chloramphenicol, the only other antibiotic with activity against RMSF, may be less effective than doxycycline, with a fatality rate of 8.9% versus 1.6% in the doxycycline group.23 Given the adverse effects of chloramphenicol, including peripheral neuropathy and aplastic anemia, tetracyclines may be considered a more desirable option for RMSF even in children aged younger than 8 years.
Another indication for the use of tetracyclines and fluoroquinolones in children is the treatment of Bacillus anthracis infection or exposure.24 While penicillin can be efficacious, it should be used only in cases where the susceptibility of the isolates is known, leaving the choice of empiric therapy to ciprofloxacin or doxycycline.24 In deciding which agent to use, the potential risk factors of the foregoing medications should be weighed against patient-specific concerns.
Current Recommendations on Pediatric Fluoroquinolone and Tetracycline Use
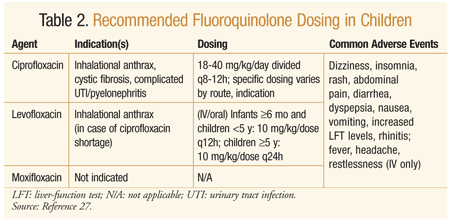
The 2006 American Academy of Pediatrics (AAP) recommendations on fluoroquinolones limit their use to three major circumstances: 1) FDA-approved indications; 2) multidrug-resistant pathogens with no safe or effective alternative; and 3) oral fluoroquinolone sensitivity when all other options are IV only (see dosing recommendations, TABLE 2).4 Fluoroquinolones may be considered in pediatric patients with acute otitis media, sinusitis, lower respiratory tract infections, pneumonia, gastrointestinal infections, or Mycobacterium infections.5 Additionally, topical fluoroquinolones are approved and recommended for acute conjunctivitis in children aged older than 12 months and for external otitis.5
The AAP recommends the use of tetracyclines in infections where the benefits outweigh the risks of adverse events. These include rickettsial infections such as RMSF or ehrlichiosis, and other infections such as cholera and anthrax.25 The AAP suggests the use of doxycycline specifically in these patients, as it has a lower risk of dental staining with less frequent dosing intervals (see dosing recommendations, TABLE 3).25 Additionally, the CDC recommends doxycycline or ciprofloxacin for any patient with B anthracis infection or exposure.24

Conclusion
In children, the pharmacokinetic and pharmacodynamic processes of many drugs, especially antibiotics, may be different compared with those in adults. These physiological changes can result in unfavorable outcomes for the pediatric patient. Because of the ongoing safety concerns surrounding the use of fluoroquinolones and tetracyclines in children, these agents should continue to be limited to treatment of FDA-approved indications or for infections for which no safe and effective alternative exists.
REFERENCES
1. Kearns GL, Abdel-Rahman SM, Alander SW, et al. Developmental
pharmacology—drug disposition, action and therapy in infants and
children. N Engl J Med. 2003;349:1157-1167.
2. Routledge PA. Pharmacokinetics in children. J Antimicrob Chemother. 1994;34(suppl A):19-24.
3. Strolin Benedetti M, Baltes EL. Drug metabolism and disposition in children. Fundam Clin Pharmacol. 2003;17: 281-299.
4. American Academy of Pediatrics Committee on Infectious Diseases. The use of systemic fluoroquinolones. Pediatrics. 2006;118:1287-1292.
5. Bradley JS, Jackson MA; Committee on Infectious Diseases, American
Academy of Pediatrics. The use of systemic and topical
fluoroquinolones. Pediatrics. 2011;128:e1034-e1045.
6. Adefurin A, Sammons H, Jacqz-Aigrain E, Choonara I. Ciprofloxacin safety in paediatrics: a systematic review. Arch Dis Child. 2011;96:874-880.
7. Noel GJ, Bradley JS, Kauffman RE, et al. Comparative safety
profile of levofloxacin in 2523 children with a focus on four specific
musculoskeletal disorders. Pediatr Infect Dis J. 2007;26:879-891.
8. Zervos MJ, Hershberger E, Nicolau DP, et al. Relationship between
fluoroquinolone use and changes in susceptibility to fluoroquinolones of
selected pathogens in 10 United States teaching hospitals, 1991-2000. Clin Infect Dis. 2003;37:1643-1648.
9. Ho PL, Tse WS, Tsang KW, et al. Risk factors for acquisition of levofloxacin-resistant Streptococcus pneumoniae: a case-control study. Clin Infect Dis. 2001;32:701-707.
10. Yee CL, Duffy C, Gerbino PG, et al. Tendon or joint disorders in
children after treatment with fluoroquinolones or azithromycin. Pediatr Infect Dis J. 2002;21:525-529.
11. Khaliq Y, Zhanel GG. Fluoroquinolone-associated tendinopathy: a critical review of the literature. Clin Infect Dis. 2003;36:1404-1410.
12. Fedler KA, Jones RN, Sader HS, Fritsche TR. Activity of
gatifloxacin tested against isolates from pediatric patients: report
from the SENTRY Antimicrobial Surveillance Program (North America,
1998-2003). Diagn Microbiol Infect Dis. 2006;55:157-164.
13. Smith MW, Unkel JH, Fenton SJ, et al. The use of tetracyclines in pediatric patients. J Pediatr Pharmacol Ther. 2001;6:66-71.
14. Sánchez AR, Rogers RS III, Sheridan PJ. Tetracycline and other
tetracycline-derivative staining of the teeth and oral cavity. Int J Dermatol. 2004;43:709-715.
15. Conchie JM, Munroe JD, Anderson DO. The incidence of staining of permanent teeth by the tetracyclines. Can Med Assoc J. 1970;103:351-356.
16. Tan KR, Magill AJ, Parise ME, et al. Doxycycline for malaria
chemoprophylaxis and treatment: report from the CDC expert meeting on
malaria chemoprophylaxis. Am J Trop Med Hyg. 2011;84:517-531.
17. Grossman ER, Walchek A, Freedman H. Tetracyclines and permanent teeth: the relation between dose and tooth color. Pediatrics. 1971;47:567-570.
18. Forti G, Benincori C. Doxycycline and the teeth. Lancet. 1969;1:782.
19. Volovitz B, Shkap R, Amir J, et al. Absence of tooth staining with doxycycline treatment in young children. Clin Pediatr (Phila). 2007;46:121-126.
20. Moore DE. Drug-induced cutaneous photosensitivity: incidence, mechanism, prevention and management. Drug Safety. 2002;25:345-372.
21. Layton AM, Cunliffe WJ. Phototoxic eruptions due to doxycycline—a dose-related phenomenon. Clin Exp Dermatol. 1993;18:425-427.
22. Rocky Mountain spotted fever. In: Pickering LK, Baker CJ, Kimberlin DW, Long SS, eds. Red Book: 2009 Report of the Committee on Infectious Diseases. 28th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2009:573-575.
23. Dalton MJ, Clarke MJ, Holman RC, et al. National surveillance for
Rocky Mountain spotted fever, 1981-1992: epidemiologic summary and
evaluation of risk factors for fatal outcome. Am J Tropo Med Hyg. 1995;52:405-413.
24. CDC. Update: interim recommendations for antimicrobial
prophylaxis for children of breastfeeding mothers and treatment of
children with anthrax. MMWR Morb Mortal Wkly Rep. 2001;50:1014-1016.
25. Tetracyclines. In: Pickering LK, Baker CJ, Kimberlin DW, Long SS, eds. Red Book: 2009 Report of the Committee on Infectious Diseases. 28th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2009:739.
26. Mulla H. Understanding developmental pharmacodynamics: importance for drug development and clinical practice. Paediatr Drugs. 2010;12:223-233.
27. Taketomo CK, Hodding JH, Kraus DM. Pediatric & Neonatal Dosage Handbook. 18th ed. Hudson, OH: Lexi-Comp; 2011.
To comment on this article, contact rdavidson@uspharmacist.com.





