US Pharm. 2011;36(12):30-33.
Celiac disease, also known as gluten-sensitive enteropathy or sprue, is a chronic autoimmune disorder caused by a genetic intolerance to gluten.1,2 Although gluten means the entire protein component of wheat, the word is commonly used in reference to gliadin (occurring in wheat) and the related proteins hordein (in barley) and secalin (in rye).3 Patients with celiac disease must avoid the ingestion of wheat, barley, and rye (and perhaps oats)—including any product containing these grains—in order to prevent disease-related pathology. This can be challenging, as ingestion of as little as 50 mg of gluten per day may produce disease-associated pathology, and some clinicians believe that exposure to even smaller amounts may be problematic.4,5 The FDA has suggested that a gluten-free diet be defined as ingestion of no more than 10 mg of gluten (estimated to be equivalent to 1/100 of a peanut) per day.6
Celiac disease was originally believed to be a pediatric syndrome. Today, the disorder is known to occur in adults as well. While pediatric celiac disease should not be overlooked, adult presentations are now more common. In addition, diagnosis is becoming more frequent in patients with nondiarrheal symptoms.1,7 As with other autoimmune disorders, celiac disease occurs more commonly in women than in men. Although the gastrointestinal (GI) system is the primary site of injury, celiac disease is a multisystem disorder; its adverse consequences extend beyond the GI tract, and untreated disease may be associated with the development of many other disorders, including nutritional deficiencies, other autoimmune diseases, and cancer.1,3
As many as 3 million Americans (approximately 1% of the U.S. population) may have celiac disease; however, recent findings suggest that the U.S. prevalence is much greater than originally thought and may be rising.7 Educational and advocacy efforts have helped increase the rate of diagnosis; nevertheless, celiac disease continues to be widely overlooked. It is estimated that well over 90% of individuals with celiac disease remain undiagnosed, leading some experts to refer to the disorder as a hidden epidemic.3 In celiac disease, lack of diagnosis and poor dietary compliance are associated with increased morbidity and mortality; therefore, it is necessary to improve the rate and accuracy of diagnosis.8,9 Pharmacists can play a significant role by identifying patients who may have celiac disease, referring them for evaluation, and assisting with disease management after diagnosis.
Etiology
Although more must be learned about what causes celiac disease, gluten ingestion is known to be required for the disorder to manifest; therefore, gluten has been called the crucial trigger.3 The presence of specific genes is necessary for celiac disease to develop. Approximately 95% of individuals with the disorder carry the human leukocyte antigen (HLA)-DQ2 heterodimer, and most patients who are DQ2 negative carry the DQ8 genotype. However, these HLA haplotypes alone are not enough to cause celiac disease, as they occur in up to 25% of the general population, while only approximately 1% of the population has the disorder.3,10 Other factors, some of them undetermined, are likely involved in the etiology of celiac disease. The consensus is that the pathogenesis of celiac disease involves interaction between gluten and immune, genetic, and environmental factors.
Clinical Manifestations
Many of the signs and symptoms of celiac disease are due to malabsorption caused by damage to the small intestine. These classic symptoms include diarrhea, steatorrhea, weight loss or failure to thrive, bloating, flatulence, and vitamin and nutrient deficiencies. Recognition of celiac disease may be particularly challenging in individuals who do not present with classic symptoms. Therefore, clinicians should always consider atypical manifestations such as illness resulting from single micronutrient deficiencies, nonspecific GI discomfort (e.g., constipation, abdominal pain, secondary lactose intolerance, dyspepsia), and non-GI symptoms (e.g., fatigue, depression, arthralgia, iron-deficiency anemia, osteomalacia or osteoporosis), among others (TABLE 1).11,12
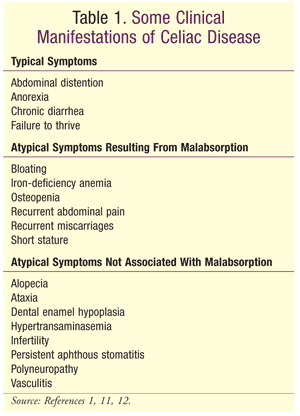
Pediatric celiac disease usually develops early, upon introduction of gluten-containing cereals. The classic presentation of predominantly digestive features with a typical malabsorption syndrome is common in children. In contrast, celiac disease in adults usually presents with fewer digestive manifestations, often involving atypical manifestations without malabsorption.2 Some patients are asymptomatic, but they are still at risk for developing intestinal damage and other disease-related complications.8
Diagnosis
Often, a significant delay occurs between symptom onset and diagnosis of celiac disease. The average time to diagnosis in the U.S. is estimated to be 9 years.3 Pharmacists can help reduce the time to diagnosis by knowing the signs and symptoms of celiac disease and being aware of common diseases that are caused by or associated with this disorder. Pharmacists must also keep in mind that patients with undiagnosed disease may seek symptomatic relief for what is believed to be another ailment. These patients may use inappropriate remedies, often without relief, while continuing to ingest gluten, thereby remaining at risk for celiac disease–related pathology.13 Since so many individuals with celiac disease are undiagnosed, as noted earlier, pharmacists can make a significant contribution by identifying those who may benefit from a diagnostic evaluation.
Two things are required to confirm a diagnosis of celiac disease: a duodenal biopsy demonstrating intraepithelial lymphocytes, crypt hyperplasia, and villous atrophy, and a positive response to a gluten-free diet.1 Serologic tests are useful, noninvasive, adjunctive diagnostic tools that are currently the means by which most celiac disease patients are identified.14 These tests also facilitate the screening of large populations.15,16 Serologic tests for endomysial immunoglobulin A antibodies or antitissue transglutaminase antibodies—considered to be equally accurate—are highly sensitive and selective.14,17
Management
The six fundamental elements of celiac disease management may be identified by the mnemonic CELIAC: Consultation with a skilled dietician, Education about the disease, Lifelong adherence to a gluten-free diet, Identification and treatment of nutritional deficiencies, Access to an advocacy group, and Continuous long-term follow-up by a multidisciplinary team.14,17 Fundamental goals of treatment include relieving symptoms, healing the intestine, and reversing the consequences of malabsorption while enabling the patient to enjoy a healthful, interesting, and practical gluten-free diet.14,18
People with celiac disease must adhere to a permanent gluten-free lifestyle. This includes complete avoidance of foods or other products that contain or have been contaminated with gluten. Fortunately, various gluten-free grains and food products exist, with more products becoming available.14 Patients usually experience clinical improvement within days or weeks of beginning a gluten-free diet.1
In celiac disease, intestinal lesions lead to reduced absorptive capacity in the small intestine, which increases the risk of malabsorptive syndrome. Therefore, it is highly important to assess the patient for vitamin and mineral deficiencies. Although a number of nutrients should be investigated, patients should be evaluated for deficiencies in folic acid, vitamin B12, fat-soluble vitamins, iron, and calcium, in particular.1,19 Sometimes, iron-deficiency anemia may be the patient’s sole manifestation of celiac disease.10,20 Since the majority of adults with celiac disease have some degree of osteopenia or osteoporosis, it is recommended that all patients with the disorder be screened for bone loss.3,21
As many as one-third of patients with celiac disease develop dyspepsia or gastroesophageal reflux, disorders that frequently are treated with proton pump inhibitors. Clinicians should be mindful that the increase in intragastric pH that these drugs cause reduces the digestion of dietary proteins and increases immune-system stimulation. This change in intragastric pH may also facilitate the entry of pathogenic microorganisms and the inactivation of digestive enzymes.10
Adults with celiac disease are at increased risk for certain infectious diseases.22 The risk of infections caused by encapsulated organisms (including pneumococcal pneumonia and meningococcal infection) is related to hyposplenism, which is commonly found in active celiac disease.23 Immunizing pharmacists should be aware that patients older than 50 years should receive pneumococcal vaccine.3 An annual influenza vaccine should be administered to celiac disease patients to reduce the incidence of secondary bacterial infections.23
Gluten in Pharmaceutical Products
Although additional research is needed to determine the adverse events associated with ingestion of trace amounts of gluten, patients and health care providers (HCPs) must verify the gluten content of all drugs, cosmetics, and other pharmaceutical products that may be intentionally or unintentionally ingested.4,24 Patients may not realize that orally ingested prescription and nonprescription drugs, vitamins, nutritional supplements, health and beauty aids, and cosmetics may contain gluten.25 While the potential for oral ingestion of some of these products may be recognized by the patient (e.g., toothpaste, lipstick), other possible means by which some gluten-containing products may be ingested (e.g., swallowing shampoo or soap while showering) may be overlooked.3,8 Although gluten is not absorbed through the skin, gluten-containing topical products may create the potential for gluten ingestion if a patient touches a hand to his or her mouth or handles food after using such products.8
Determining whether a pharmaceutical product is gluten free may be challenging.26-28 Some progress has been made; however, very few labels for prescription and nonprescription drug products, vitamins, supplements, and cosmetics specifically address gluten content. In addition, the ingredient list on the label may be insufficient information to ascertain whether the product is safe for patients with celiac disease. In order to determine whether a product is acceptable, the inherent gluten content of the product, as well as whether the product may have been contaminated with gluten during production, handling, or packaging, must be confirmed. Pharmacists have a great responsibility to help patients and their caregivers ascertain whether the use of a specific pharmaceutical product will result in the ingestion of gluten.
Gluten contained in inactive ingredients has an important, often unrecognized, impact upon the final product’s gluten status. Because a specific gluten-content designation may not appear on the label, it is extremely important to carefully review the list of inactive ingredients, since substances derived from whole grains, grain flour, or starch grain may contain gluten. While excipients such as cornstarch and potato starch are generally gluten free (unless contaminated), unspecified or pregelatinized starch, dusting powder, and flours may be derived from wheat (TABLE 2).15,25-27

It is also important to check sweeteners used in pharmaceutical products. Although uncontaminated sucrose, honey, dextrose, fructose, and corn syrup solids should not be problematic, the ingestion of sweeteners such as wheat- and barley-based syrups must avoided.26
Fillers, thickening agents, and polymers used for liquid and solid drug formulations, such as gums, cellulose and its derivatives, and other polymers, generally do not contain gluten. It is recommended, however, that the source of the solvent or vehicle used in a liquid formulation be verified to ensure that it is free of gluten-containing grains.26 As noted previously, the potential for gluten contamination must always be considered.
HCPs and consumers should be encouraged to contact manufacturers to verify the gluten content of their products. Specifically, it should be asked, “Has this product been confirmed to be gluten free?” Importantly, some manufacturers may state that although they do not add any ingredients derived from gluten when making the product, they do not test to confirm that the final product is gluten free. They may also note that the suppliers of the raw materials used to manufacture the product cannot guarantee that the materials do not contain or are not contaminated with gluten. Manufacturers may state that they use potato- or corn-based starch derivatives in their product, but that they are not absolutely sure that minute amounts of gluten contaminants from other raw materials used at the same location are not present in their inactive ingredients.26 Such nonspecific information often leads to uncertainty as to whether the product should be used by someone with celiac disease.
Published information about the gluten content of foods has become more accessible; however, verifying the gluten content of pharmaceutical products remains a challenge.28 Web-based listings (e.g., www.glutenfreedrugs.com, www.celiaccentral.com, www.ashp.org/gluten) and various publications can assist patients and HCPs in this regard.28-31 Some professional organizations and celiac disease foundations are collaborating (e.g., the American Society of Health-System Pharmacists and the National Foundation for Celiac Awareness) to help patients and HCPs obtain drug information. Although updated lists of gluten-free drugs are available, pharmacists must be aware that formulations may change after the source information is published and that the manufacturer should be contacted to obtain current information.
Conclusion
Pharmacists have a responsibility, as well as many opportunities, to care for patients with celiac disease. Counseling the patient about gluten-free diet, drugs, vitamins, and nutritional supplements and addressing other health-related matters are but two ways in which the pharmacist can help. Pharmacy profiles that clearly identify patients with celiac disease are important. Patients should be reminded that although the amount of gluten in a single exposure to a pharmaceutical product may be small, any ingestion of gluten from any source must be avoided, as the cumulative effect of inadvertent ingestion of gluten from different sources (e.g., unknowingly eating foods contaminated with gluten) may exceed the threshold for causing intestinal damage.
REFERENCES
1. Green PHR, Cellier C. Celiac disease. N Engl J Med. 2007;357:1731-1743.
2. Rodrigo-Sáez L, Fuentes-Álvarez D, Pérez-Martínez I, et al. Differences between pediatric and adult celiac disease. Rev Esp Enferm Dig. 2011;103:238-244.
3. Green PH, Jones R. Celiac Disease: A Hidden Epidemic. New York, NY: HarperCollins; 2006.
4. Catassi C, Fabiani E, Iacono G, et al. A prospective, double-blind, placebo-controlled trial to establish a safe gluten threshold for patients with celiac disease. Am J Clin Nutr. 2007;85:160-166.
5. Akobeng AK, Thomas AG. Systemic review: tolerable amount of gluten for people with coeliac disease. Aliment Pharmacol Ther. 2008;27:1044-1052.
6. Blumer I, Crowe S. Celiac Disease for Dummies. Mississauga, Ontario: John Wiley & Sons Canada, Ltd; 2010.
7. Rampertab SD, Pooran N, Brar P, et al. Trends in the presentation of celiac disease. Am J Med. 2006;119:355.e9-355.e14.
8. Korn D. Wheat Free, Worry Free: The Art of Happy, Healthy Gluten-Free Living. Carlsbad, CA: Hay House, Inc; 2002.
9. Sollid LM. Coeliac disease: dissecting a complex inflammatory disorder. Nat Rev Immunol. 2002;2:247-255.
10. García-Manzanares A, Lucendo AJ. Nutritional and dietary aspects of celiac disease. Nutr Clin Pract. 2011;26:163-173.
11. Murray JA. The widening spectrum of celiac disease. Am J Clin Nutr. 1999;69:354-365.
12. Leeds JS, Hopper AD, Sanders DS. Coeliac disease. Br Med Bull. 2008;88:157-170.
13. Mangione RA, Patel PN. Caring for patients with celiac disease: the role of the pharmacist. Pharmacy Today. 2008;14:50-64.
14. See J, Murray JA. Gluten-free diet: the medical and nutritional management of celiac disease. Nutr Clin Pract. 2006;21:1-15.
15. Drago S, Di Pierro M, Catassi M, Fasano A. Recent developments in the pathogenesis, diagnosis and treatment of celiac disease. Expert Opin Ther Patents. 2002;12:45-51.
16. Catassi C, Kryszak D, Louis-Jacques O, et al. Detection of celiac disease in primary care: a multicenter case-finding study in North America. Am J Gastroenterol. 2007;102:1454-1460.
17. National Institutes of Health. NIH Consensus Development Conference on Celiac Disease.
http://consensus.nih.gov/2004/
18. Ferrell RJ, Kelly CP. Diagnosis of celiac sprue. Am J Gastroenterol. 2001;96:3237-3246.
19. Hallert C, Grant C, Grehn S, et al. Evidence of poor vitamin status in coeliac patients on a gluten-free diet for 10 years. Aliment Pharmacol Ther. 2002;16:1333-1339.
20. Rodrigo L. Celiac disease. World J Gastroenterol. 2006;12:6585-6593.
21. Stazi AV, Trecca A, Trinti B. Osteoporosis in celiac disease and in endocrine and reproductive disorders. World J Gastroenterol. 2008;14:498-505.
22. Freeman HJ, Chopra A, Clandinin MT, Thomson AB. Recent advances in celiac disease. World J Gastroenterol. 2011;17:2259-2272.
23. Walters JR, Bamford KB, Ghosh S. Coeliac disease and the risk of infections. Gut. 2008;57:1034-1035.
24. Patel DG, Krogh CM, Thompson WG. Gluten in pills: a hazard for patients with celiac disease. Can Med Assoc J. 1985;133:114-115.
25. Gluten-free drugs for celiac disease patients. Med Lett Drugs Ther. 2008;50:19-20.
26. Crowe JP, Falini NP. Gluten in pharmaceutical products. Am J Health Syst Pharm. 2001;58:396-401.
27. Cacace JL. Formulating for the gluten-sensitive individual. IJPC. 2005;9:357-358.
28. Jay L. Letter to the editor. Hosp Pharm. 2011;46:389.
29. King AR. Gluten content of the top 200 medications of 2009: a follow-up to the influence of gluten on patients’ medication choices. Hosp Pharm. 2011;46:417-426.
30. Plogsted S. Medications and celiac disease—tips from a pharmacist. Pract Gastroenterol. 2007;31:58-64.
31. Lowell JP. The Gluten-Free Bible: The Thoroughly Indispensable Guide to Negotiating Life Without Wheat. New York, NY: Henry Holt & Co; 2005.
To comment on this article, contact rdavidson@uspharmacist.com.





