US Pharm. 2016(41)22-19.
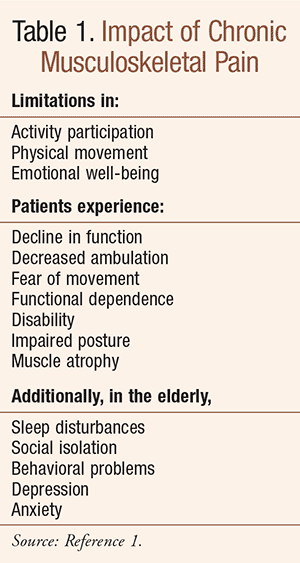
The number of adults suffering from musculoskeletal pain (TABLE 1) has grown exponentially with the increase in the geriatric population; there are now 35 million people aged >65.1,2 Arthritis, a particular type of musculoskeletal disorder, is one of the most common conditions in the United States, and is the leading cause of disability and a major factor in limiting quality of life.3,4 Approximately one in five adults in the U.S. has been diagnosed with some form of arthritis.3 While the word arthritis is a blend of two forms ultimately from Greek—arthr-, meaning “joints,” and -itis, meaning “inflammation,”—the term refers not to one disease, but rather to a variety of diseases that can cause joint pain or stiffness, damage to the structure of a joint, or loss of joint function (TABLE 2).3 Arthritis may affect any joint in the body, causing pain and immobility to Americans by the millions, according to the National Institutes of Health.5,6
There are many factors that contribute to the development of arthritis, including heredity, obesity, trauma, aging, loss of estrogen, occupational factors (i.e., heavy physical labor, such as that performed by dockworkers, miners, and furniture movers), medical conditions (e.g., certain metabolic disorders), and sports injuries.5 Of note, while arthritis is often thought of as one disease, it actually occurs in more than 100 different forms, some that develop gradually secondary to wear of the joints, such as osteoarthritis (OA), and others that manifest acutely and wax and wane.3 It is important for healthcare professionals to understand that OA and other “aches and pains” (TABLE 2) may overlap with inflammatory autoimmune diseases and disorders in an elderly person.

Inflammatory autoimmune diseases such as rheumatoid arthritis (RA) are relatively common conditions, especially among adults aged >50.7 Over 5% of women over 75 years of age have RA.4 Risk factors for developing an autoimmune disease include age and gender, among others.7 A recent study estimating the lifetime risk of RA (3.6% for women and 1.7% for men) and other autoimmune diseases indicates that the second most common autoimmune disorder is polymyalgia rheumatica (PMR), with a lifetime risk of 2.4% for women and 1.7% for men.7,8 People of Northern European and Scandinavian descent are at higher risk for developing PMR; older adults are affected almost exclusively, with an average age at onset of 70 years and a range from 50 to 90 years.3,4
Signs and Symptoms
PMR onset is dramatic and presents with bilateral proximal aching of the shoulder and hip girdle muscles and the back and neck muscles; morning stiffness is typical (lasting >1 hour), and discomfort may be such that patients are prevented from rising out of bed in the morning and performing simple tasks.4,9 Bursitis is primarily responsible for shoulder symptoms; pain may cause subjective weakness, although muscle strength is usually unimpaired.4,9 Synovitis of the knees, wrists, and small joints of the hands—mild, transient, and without erosive changes—is often associated with PMR.4
Of note, both PMR and giant cell arteritis (GCA; also known as temporal arteritis) are related diseases of the elderly (TABLE 3). Patients need to be warned about symptoms of GCA—headache (severe and localized to the temple), muscle pain during chewing, and, in particular, visual disturbances—and should report them immediately to their healthcare provider since this condition may develop at the onset of PMR or much later, and even after the patient’s disorder appears to be resolved.9

Diagnosis, Treatment, and Prognosis
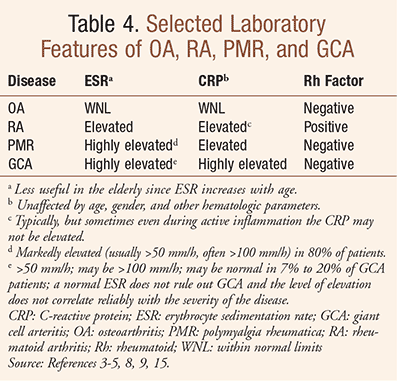
PMR is suspected in geriatric adults with typical symptoms; the differential diagnosis includes RA, polymyositis, hypothyroidism, multiple myeloma, and fibromyalgia.9 The erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) levels are typically elevated in PMR, while rheumatoid factor is usually absent (TABLE 4).3,9 The ESR reading may be less useful in older adults, however; the sedimentation rate increases with age, and a normal level is more difficult to determine.3 Additionally, even during active inflammation, the CRP may not be elevated; the ESR and CRP levels are often helpful in monitoring the degree of inflammation at any given point in time, and in conjunction with symptoms, can be used as a tracking tool for treatment effectiveness.3,9
While aspirin and non-steroidal anti-inflammatory drugs (NSAIDs) are noted to be effective for mild symptoms, NSAIDs are rarely sufficient. The usual treatment is daily, low-dose corticosteroid therapy. While immediate and complete relief of symptoms is usually achieved and resolution occurs in 1 to 2 years, both professional care and self-care are considered essential elements of management.3 Prednisone is initiated at 15 to 20 mg orally once a day and results in dramatic improvement within hours or days; this response may be used to support the diagnosis.9
As a point of reference, the usual labeled dosage range for oral prednisone in adults is 5 mg/day to 60 mg/day administered in one to four divided doses, depending upon the disease being treated, with individualization of dose and titration to response.10 After symptoms are controlled, the dose is slowly decreased every 5 to 7 days.10 Maintenance doses in which prednisone is used for chronic conditions are usually 10 to 20 mg orally once a day, or 20 mg to 40 mg orally every other day.10
If GCA is suspected to be present, 1) the dose of corticosteroids should be higher: prednisone (unlabeled use) is given initially, 40 to 60 mg orally daily, typically requiring 1 to 2 years of treatment; however, a taper may begin after 2 to 3 months; 2) alternative dosing of 30 to 40 mg orally per day has demonstrated similar efficacy; and 3) a temporal artery biopsy should be performed.9,11,12
There are no available specific guidelines for dosage adjustments of prednisone in renal impairment; it appears that no dosage adjustments are required.10 Prednisolone is useful and preferred to prednisone in patients with significant liver disease, since it does not require hepatic activation.10,11 No dosage adjustment of prednisolone is needed in hepatic dysfunction.10 Doses are equivalent (i.e., 1 mg prednisone is equivalent to 1 mg of prednisolone).10
Monitoring parameters include measurement of blood pressure, weight, blood glucose (2-hour postprandial), and electrolytes; intraocular pressure (with use of the drug for >6 weeks); and chest x-ray (if prolonged treatment).11,13 In patients with a history of peptic ulcer disease, upper gastrointestinal (GI) x-ray should be considered.13 Additionally, there are special geriatric considerations regarding the use of systemic prednisone in light of the risk of adverse effects: use cautiously in the elderly, in the smallest dose for the shortest period of time. If long-term use is necessary, monitor bone-mineral density, institute fracture-prevention strategies, and consider the appropriateness of introducing a bisphosphonate to prevent osteoporosis.9,11 In elderly patients, pharmacists should monitor for complications of corticosteroid use (e.g., diabetes, hypertension).9
Regarding the maximum dosage limits of prednisone, it is important to reiterate that the dosage must be individualized and is highly variable depending on the nature and severity of the disease and on patient response.10 Although there is no absolute maximum dosage, the Boston Collaborative Drug Study found that psychiatric events occurred in fewer than 1% of patients when prednisone was prescribed in doses of <30 mg/day, whereas their incidence rose to 18% in patients receiving 80 mg/day.10,14
Some patients who are unable to have their prednisone dose tapered and who have frequent recurrences may benefit from the addition of methotrexate (10-15 mg orally once a week, if renal function is normal) or another immunosuppressant such as azathioprine.9 The addition of a second agent in PMR or GCA is controversial, since minimal benefit has been shown in controlled, randomized trials.9 No benefit has been shown in trials using anti-tumor necrosis factor TNF agents (rituximab, infliximab, and adalimumab).9
Conclusion
It is important for pharmacists to understand that the musculoskeletal pain of osteoarthritis and the “aches and pains” of other conditions may overlap with inflammatory autoimmune diseases and disorders in an elderly person. PMR, with its dramatic onset—and the related GCA—is an example of such an inflammatory condition; it should not be overlooked in the elderly, so that specific treatment may be initiated to provide relief from pain.
REFERENCES
1. Laird R, Udeshi AR. Chronic musculoskeletal pain: initial pharmacological and non-pharmacological therapies. Today’s Geriatric Medicine. 2016; Mar/Apr:10-14.
2. Podichetty VK, Mazanec DJ, Biscup RS. Chronic non-malignant musculoskeletal pain in older adults: clinical issues and opioid intervention. Postgrad Med J. 2003;79(937):627-623.
3. Mayo Clinic on Arthritis. Rochester, Minnesota: Mayo Clinic; 2013:13-80.
4. Scott DL. Arthritis in the elderly. In: Fillit HM, Rockwood K, Woodhouse K, eds. Brocklehurst’s Textbook of Geriatric Medicine and Gerontology. 7th ed. Philadelphia, PA: Saunders Elsevier; 2010:566-576.
5. Vad V. Arthritis Rx. New York, New York: Gotham Books; 2006:19-56.
6. Osteoarthritis. National Institutes of Health. U.S. Department of Health and Human Services. Updated March 29, 2013. https://report.nih.gov/nihfactsheets/ViewFactSheet.aspx?csid=55. Accessed May 16, 2016.
7. Polymyalgia rheumatica (PMR). WebMD. January 11, 2011. www.webmd.com/rheumatoid-arthritis/news/20110110/1-in-12-women-will-have-autoimmune-disease. Accessed April 1, 2016.
8. Crowson C, Matteson EL, Myasoedova E, et al. The lifetime risk of adult-onset rheumatoid arthritis and other inflammatory autoimmune rheumatic diseases. Arthritis Rheum. 2011;63(3): 633-639. www.ncbi.nlm.nih.gov/pmc/articles/PMC3078757/. Accessed April 1, 2016.
9. Polymyalgia rheumatica. Merckmanuals.com. Last full review/revision February 2016. www.merckmanuals.com/professional/musculoskeletal-and-connective-tissue-disorders/vasculitis/polymyalgia-rheumatica. Accessed May 13, 2016.
10. Prednisolone. clinicalpharmacology-ip.com. Revised March 24, 2016. http://clinicalpharmacology-ip.com. Accessed May 8, 2016.
11. Semla TP, Beizer JL, Higbee MD. Geriatric Dosage Handbook. 20th ed. Hudson, OH: Lexicomp; 2015:1199-1201, 1202-1205.
12. Hiratzka LF, Bakris GL, Beckman JA, et al. ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM guidelines for the diagnosis and management of patients with thoracic aortic disease: executive summary. J Am Coll Cardiol. 2010;55(14):1509-1544.
13. Epocrates.com. Epocrates Plus Version 15.12.1. Updated May 9, 2016. Accessed May 10, 2016.
14. The Boston Collaborative Drug Surveillance Program. Acute adverse reactions to prednisone in relation to dosage. Clin Pharmacol Ther. 1972;13:694-698.
15. Seetharaman M, Foster CS, Albertini JG, et al. Giant cell arteritis (temporal arteritis) workup. Medscape.com. Updated November 4, 2015. http://emedicine.medscape.com/article/332483-workup. Accessed May 23, 2016.
To comment on this article, contact rdavidson@uspharmacist.com.






