US Pharm. 2010;35(9):32-40.
Human papillomavirus (HPV)—a small, double-stranded DNA virus—is the most common sexually transmitted infection in the United States.1 Approximately 20 million Americans between the ages of 15 and 49 years are currently infected, and an estimated 6.2 million new infections occur each year.2 It is estimated that 50% of sexually active males will acquire a genital HPV infection in their lifetime, and the vast majority of women, at least 80%, will have acquired the infection by the age of 50 years.3 More than 120 different genotypes of HPV have been identified thus far based on genetic sequence of the outer capsid protein L1, of which approximately 40 express preference for the anogenital mucosa.4
Classification
HPV infection has a variable presentation that may be divided into two main classifications—cutaneous and mucosal. Cutaneous infections include common, plantar, or flat warts, while mucosal infections include both genital and anogenital diseases.3 This classification is further subdivided as low risk (nononcogenic) and high risk (oncogenic) according to the propensity to cause cervical cancer. The low-risk HPV may be responsible for genital warts or mild cervical dysplasia. Although 12 genotypes have been defined as low risk, types 6 and 11 account for 90% of cases of anogenital warts. A larger number of genotypes are associated with high-risk HPV, which may lead to low- and high-grade dysplasia; however, types 16 and 18 are responsible for approximately 70% of cervical cancers.3
Disease Burden
Since HPV is one of the most widespread diseases in the U.S., it bears a considerable burden. It is estimated that the cost of successfully treating one episode of genital warts is approximately $460, and annual health care costs exceed $210 million.5 HPV that causes cancer has a much higher cost, both financial and emotional. Long-term consequences of HPV in males include perianal warts and anal cancer. There is a significantly higher risk of anal cancer in the “men who have sex with men” (MSM) population, especially in those with concurrent HIV infection.2 Almost all cervical cancers are associated with persistent infection from high-risk types of HPV.2 In the U.S., over 50 million Pap tests are performed every year, with an estimated $3.9 billion spent on the evaluation, treatment, and follow-up of HPV-related abnormal results.3 Additionally, $146 million is spent on medical costs associated with invasive cervical cancer. The American Cancer Society (ACS) estimated that 11,700 women would develop cervical cancer in 2008, with approximately 3,900 deaths from the disease.1 With the high burden of this disease, studies are being conducted to evaluate the cost-effectiveness of preventive measures, including the HPV vaccines.
Prevention With Vaccines
HPV is highly communicable and is transmitted by sexual contact during both acute and latent infection. The use of barrier methods such as condoms can reduce, but not eliminate, transmission. Abstinence from sexual activity or monogamy with an uninfected partner is the best method to prevent infection.1
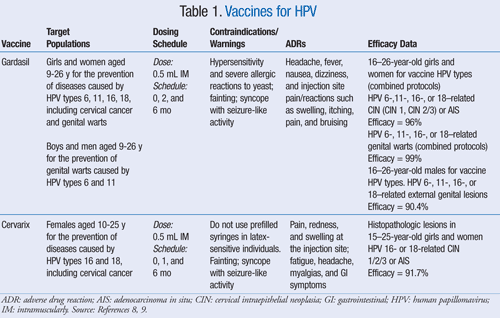
Currently, there are two FDA-approved vaccines available to provide protection against the most common genotypes of HPV. Clinical trails found both Gardasil (Merck & Co., Inc.) and Cervarix (GlaxoSmithKline) highly efficacious (>90%) in preventing cervical intra-epithelial neoplasia (CIN) grades 1, 2, and 3, and anogenital cancers caused by oncogenic genotypes 16 and 18.6 Limitations of the vaccines include diminished efficacy in preventing cervical disease in women who were already HPV seropositive at the time of administration. Thus, it is optimal to administer the vaccine prior to sexual debut.6 Immunocompromised patients may also show a diminished response to both Gardasil and Cervarix. Safety has not been established in pregnant women; therefore, the vaccine is currently not recommended for use in this population.7 However, if a pregnant woman receives the vaccine, she should contact the pregnancy registry set up by the manufacturer in order to monitor maternal and fetal outcomes.8,9 Both vaccines are thimerosal free. One notable difference is that the quadrivalent vaccine, Gardasil, has recently been approved for use in boys and young men to diminish the spread of genital warts, as it also confers protection against genotypes 6 and 11.8 Additional information regarding both vaccines can be found in TABLE 1.8,9
The vaccines cost approximately $100 per dose and require a three-dose series.6 Although HPV vaccinations are part of the immunization schedule recommended by the American Committee on Immunization Practices, they are not yet covered by many insurance plans. In response, government programs were created to assist families who cannot afford to vaccinate their children.
Screening and Diagnosis
Regular cervical cancer screenings are one of the greatest preventive measures a woman can take to lower her risk of cancer-related death. The CDC does not have specific recommendations regarding cervical cancer screenings, but guidelines have been published from professional organizations such as the American College of Obstetricians and Gynecologists, the ACS, and the U.S. Preventive Services Task Force. Most organizations agree that Pap test screenings should begin approximately 3 years after a woman begins having sexual intercourse (but no later than 21 years of age), and women should be rescreened every 2 to 3 years. Between the ages of 65 and 70 years, women with normal results on previous Pap tests may discontinue screenings.10 Most importantly, it should be stressed that receiving a vaccination against HPV does not eliminate the need for women to have regular cervical cancer screenings.
Clinical Presentation
Most cases of HPV infection are asymptomatic and will resolve within 1 year even without treatment. However, in some cases, the infection persists and can lead to external warts or cervical abnormalities. Genital warts (condylomata acuminata) are hyperkeratotic papules that vary in size, shape, and color. Warts can occur on the labia, introitus, and vagina of women or on the penile shaft of circumcised men.10 Most often, genital warts develop within 3 to 4 months after exposure and are usually asymptomatic; however, itching, burning, pain, and tenderness can occur as well. Cervical neoplasias are also asymptomatic and are usually first detected after an abnormal Pap test.
Risk factors for developing HPV infection are related to sexual behavior, including a high number of sexual partners, partners’ sexual history, young age (<25 years), and early age at first sexual intercourse (16 years or younger).2 While drug therapy exists to treat genital warts, only recently have vaccines been approved that try to prevent cervical cancer from developing in patients with persistent, high-risk (oncogenic) HPV infections.
Treatment
Several therapies exist for the treatment of genital warts, but none have proven to be extremely effective or to decrease transmission of the disease. Most are designed to eradicate superficial lesions, not to cure the infection itself. This leads to a relatively high relapse rate and the necessity for alternative therapy. Choice of treatment can be dependent on many factors, including size, number, and location or morphology of warts; patient preference; patient compliance; cost; side-effect profile; and provider experience and preference.3
The most convenient and preferred treatments are topical products that can be administered by patients at home. However, proper technique and compliance must be established in order to ensure that the treatment will achieve optimal efficacy. The two most common medications available are: 1) podofilox (Condylox), a derivative of podophyllin (a resin extract) with antimitotic properties but an unknown mechanism of action against genital warts; and 2) imiquimod (Aldara), a cytokine inducer that activates the cell-mediated immune system (TABLE 2). Both treatments initially have clearance rates >50%, but recurrence is common.11-13
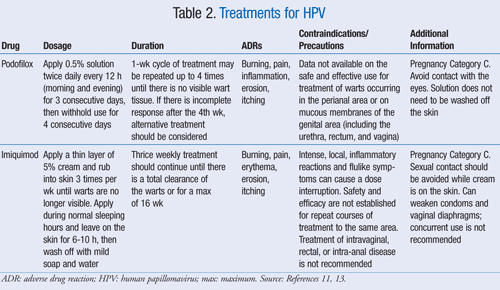
Other options for wart removal must be performed by a physician in a medical office or hospital. One such option is topical podophyllin resin, which comes in a 10% to 25% solution. No more than 0.5 mL should be applied per treatment, and the solution should not come in contact with an open wound, as systemic absorption leading to toxicity can occur.14 Trichloroacetic acid (TCA) and bichloroacetic acid (BCA) 80% to 90% solutions are both caustic agents that destroy warts by chemical coagulation of proteins. The acid is applied directly to the wart and allowed to dry. This treatment can be repeated weekly if necessary. Cryotherapy using liquid nitrogen destroys the wart through thermal-induced cytolysis. Cryotherapy can be repeated every 1 to 2 weeks as needed.14 Genital warts in the immunocompromised patient are largely refractory to topical treatments. Surgery to excise the wart and/or combination therapy with adjunctive imiquimod are better options for these patients.14 The advantage of surgery is that it usually eliminates warts in a single visit and is most beneficial for patients with numerous warts over a large area.
Treatment for cervical cancer involves chemotherapy with adjunctive radiation therapy. This type of treatment must be performed by a physician in a monitored setting due to adverse events associated with the administration of cytotoxic agents. Each cycle of chemotherapy/radiation can last multiple weeks, and patients may require several cycles in an attempt to induce remission.
Patient Counseling
Pharmacists play an important role in the care of patients with HPV. One major component is to serve as educators about the disease and methods of prevention, which includes vaccinations. Other areas should include assisting prescribers in selecting the most appropriate treatment for genital warts and counseling patients on proper administration. A summary of key points is provided in TABLE 3.1,13,15,16

REFERENCES
1. Centers for Disease Control and Prevention (CDC). Human papillomavirus. www.cdc.gov/vaccines/pubs/
2. CDC. Human papillomavirus: HPV information for clinicians. April 2007. www.cdc.gov/std/hpv/common-
3. Paik C, Savino LB. Prevention and treatment options for genital human papillomavirus. Am Pharm. 2007;(8):31-39.
4. Hayney MS. Vaccines, toxoids, and other immunobiologics. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 7th ed. New York, NY: McGraw-Hill; 2008:2047.
5. Chesson H; Advisory Committee on Immunization Practices, CDC. Overview of cost-effectiveness of quadrivalent and bivalent HPV vaccination. October 2009. www.cdc.gov/vaccines/recs/
6. Hutchinson DJ, Klein KC. Human papillomavirus disease and vaccines. Am J Health Syst Pharm.
7. CDC. Guidelines for vaccinating pregnant women. Human papillomavirus (HPV). January 2010. www.cdc.gov/vaccines/pubs/ 2008;65:2105-2111.
8. Gardasil (human papillomavirus quadrivalent [types 6, 11, 16, and 18] vaccine, recombinant) package insert. Whitehouse Station, NJ: Merck & Co. Inc; October 2009.
9. Cervarix (human papillomavirus bivalent [types 16 and 18] vaccine, recombinant) package insert. Research Triangle Park, NC: GlaxoSmithKline; June 2010.
10. Bonnez W, Reichman RC. Papillomaviruses. In: Mandell GC, Bennett JE, Dolin R, eds. Principles and Practice of Infectious Diseases. 7th ed. Elsevier Inc; 2010:2035-2046.
11. Condylox (podofilox) package insert. Corona, CA: Watson Pharmaceuticals; November 2007.
12. Goad JA, Hess KM. Sexually transmitted diseases. In: Koda-Kimble MA, Young LY, Kradjan WA, et al, eds. Applied Therapeutics: The Clinical Use of Drugs. 9th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2009;65:29-30.
13. Aldara (imiquimod) package insert. Bristol, TN: Grace Pharmaceuticals, LLC; April 2009.
14. CDC. Sexually transmitted diseases treatment guidelines 2006. Genital warts. August 4, 2006. www.cdc.gov/STD/treatment/
15. CDC. Human papillomavirus (HPV). Signs and symptoms. February 5, 2010. www.cdc.gov/hpv/signs-
16. CDC. Vaccines and immunizations. HPV vaccine—questions & answers. December 22, 2009. www.cdc.gov/vaccines/vpd-vac/
To comment on this article, contact rdavidson@uspharmacist.com.





