US Pharm. 2015;40(10):46-50.
ABSTRACT: Infection with HIV, which is the cause of AIDS, is a major public-health concern throughout the world, especially in developing countries. Once a person is infected with HIV, he or she remains infected for life, even with treatment. Despite the success of standard regimens, antiretroviral therapy is complicated by potential drug-drug interactions, patient nonadherence, and adverse effects. Therefore, developing a safe, effective vaccine that prevents HIV infection is the best strategy for containing the epidemic. Despite progress in vaccine development, there is still no effective vaccine for preventing HIV. Because HIV is highly diverse, it integrates into the host genome and develops ways to evade the immune system. Many roadblocks persist in the field of HIV vaccine development.
HIV is the cause of AIDS. Once a person is infected with HIV, he or she remains infected for life, even with treatment. The World Health Organization reported approximately 2 million new cases of HIV worldwide in 2014, and about 37 million people are living with HIV.1 In 2014, approximately 15 million HIV-infected persons were receiving antiretroviral therapy (ART).1
Historically, HIV has been classified into two major strains, HIV-1 and HIV-2, based on comparative analysis of the amino acid sequences and structures of HIV-1 and HIV-2 proteases.2,3 The HIV-1 strain was transmitted from apes to humans; HIV-2 was transmitted from sooty mangabey monkeys to humans.4 The clinical manifestations of HIV-2 infection are similar to those of HIV-1 infection; however, there are some important differences. HIV-1 exists worldwide, whereas HIV-2 is seen mainly in a few West African countries. HIV-2 progresses more slowly than HIV-1 and has lower virulence and transmissibility.4
HIV infection occurs primarily through three modes of transmission: sexual, parenteral, and perinatal. The CDC estimates that 8% of the 47,500 new HIV infections in the United States in 2010 occurred through parenteral drug use.5 Mucosal tissues, including the cervicovaginal and rectal mucosa, the foreskin, and the oral epithelia, are involved in the sexual transmission of HIV. HIV can affect anyone, regardless of sexual orientation, race, ethnicity, gender, or age. Individuals with a history of sexually transmitted infections, mental illness, or sexual abuse are at higher risk for HIV infection, as are African American and Latino populations.5
Pathogenesis
HIV weakens the immune system and destroys cells that fight disease and infection. HIV is distinct from most viruses in that once the body is infected, it cannot clear the infection. HIV can hide in the body for extended periods of time.6
Cluster of differentiation 4–positive (CD4+) T lymphocytes are the main target of HIV infection, but other cells can be infected.7-9 CD4+ T-cell response plays a critical role in the prevention or control of HIV infection and replication.10 HIV invades CD4+ T cells, using them to make more copies of the virus and eventually destroying host cells.6 It has been shown that patients with the highest CD4+ T-cell response have the lowest viral load.11
CD8+ T cells, or cytotoxic T lymphocytes (CTLs), also play an important role in the adaptive immunity, as they destroy virally infected cells by various mechanisms.12 Patients with a robust CD4+ T-cell response have a stronger CTL response.10 Despite this strong interaction, viral mutants may emerge and evade the immune response, a process known as viral escape.13 The humoral immune response to HIV also contributes to the reduction of viral load through antibody responses directed against various viral antigens.14 Antibodies with the ability to neutralize HIV are called neutralizing antibodies (NAbs).15 These antibodies are directed mainly against viral envelope proteins, which mediate viral binding to host cells, and they can block HIV replication of infection by virus neutralization or antibody-dependent cellular toxicity of HIV-infected cells.15 Natural killer cells (NKs), another important component of the innate immune system, are capable of killing infected cells and producing chemokines with antiviral action.16 As is the case with adaptive immune responses, viral escape can develop and restrict the antiviral effects of NKs.17
Antiretroviral Therapy
Current guidelines recommend ART for all HIV-infected patients to reduce the risk of disease progression and prevent transmission of the virus.18 Understanding the HIV life cycle is the key to understanding ART. FIGURE 1 shows the virus life cycle with potential targets where replication may be interrupted.19 Included in FIGURE 1 are examples of antiretroviral drugs, including entry inhibitors; reverse transcriptase inhibitors (nucleoside reverse transcriptase inhibitors [NRTIs], non-NRTIs [NNRTIs], and nucleotide reverse transcriptase inhibitors; integration, transcription, and translation inhibitors, also known as integrase strand transfer inhibitors (InSTIs); and protease inhibitors (PIs). Recommended ART for treatment-naïve patients consists of two NRTIs in combination with a third antiretroviral from one of the following classes: InSTI, NNRTI, or PI with a pharmacokinetic enhancer such as cobicistat or ritonavir.18 This triple combination therapy provides a durable response, significantly reduces the incidence of opportunistic infections, and improves survival.20
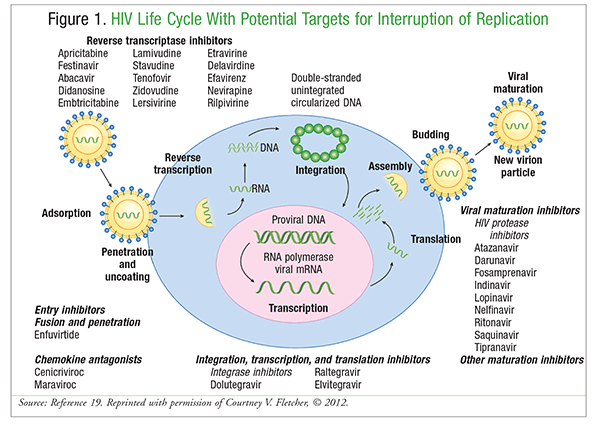
In the selection of ART, patient-specific factors such as virologic efficacy, toxicity, pill burden, dosing frequency, drug-drug interaction potential, resistance, comorbidities, and cost should be considered.18 ART extends the lifespan of HIV-infected patients. A decrease in CD4+ T cells puts patients at risk for opportunistic infections or HIV-associated diseases such as tuberculosis, toxoplasmosis, hepatitis C virus, cytomegalovirus, herpes simplex, Pneumocystis fungal infections, and others. Despite the success of standard regimens, ART is complicated by potential drug-drug interactions (especially through CYP3A-mediated metabolism and clearance), patient adherence, and adverse effects. Therefore, developing a safe and effective vaccine that prevents HIV infection would be the best strategy for containing the epidemic.
Vaccine Development and Challenges
Vaccine development is crucial to viral disease control. It is estimated that, in 2014, $841 million was spent on HIV vaccine research and development worldwide.21 Despite much research on vaccine development, there is no effective HIV vaccine to date. In most cases of viral infection, when a person becomes infected, the body is able to mount an immune response to clear the virus. The body then keeps the memory of that immune response so that immunity to the virus can develop.
The safest and most effective vaccine will elicit an appropriate adaptive immune response stimulating broadly neutralizing antibodies (bNAbs) that will later mitigate viral infection and stimulate cell-mediated immune response, which provides long-term protection via a memory response. Multiple approaches have been attempted in HIV vaccine development.
A peptide vaccine is composed of tiny pieces of proteins, whereas a subunit protein vaccine consists of bigger pieces.22 Once vaccinated, the body responds to the protein and alerts the immune system to recognize the proteins in the HIV virus if exposed.22 Data suggest that the HIV envelope protein Env is a necessary component to elicit bNAbs.23 Vaccines consisting of protein pieces that are made in a laboratory and resemble the HIV virus have been used in an attempt to elicit an antibody response.22
Viral vectors such as canarypox have also been used to carry HIV immunogens. In this type of vaccine, non-HIV viruses carry genes into the cells, causing the body to produce HIV-like proteins. The hope is that if a person is exposed to HIV in the future, the immune system will mount a response to destroy HIV.22
DNA-based vectors have also been used as a target in vaccine design. HIV genes are inserted into pieces of DNA called plasmids. HIV-like proteins are produced, and the body reacts to them after vaccination. It is surmised that, when exposed to the real HIV virus, the body will again mount an immune response.22 DNA vaccines are less effective because of their poor ability to enter cells.24
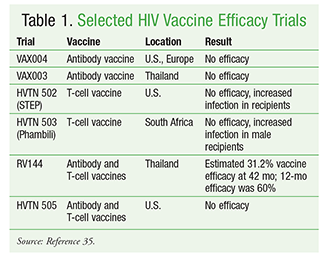
Adjuvant proteins, or a combination of two or more modalities for antigen delivery, have been used to boost vaccine efficacy.25 This strategy was attempted in the large-scale HIV vaccine trial of RV144 in Thailand.26 This trial, published in 2009, demonstrated that the vaccine was safe and prevented 31% of new infections in the >16,000 adults enrolled.26 In the RV144 trial, four doses of priming canarypox virus were administered, and on two visits volunteers also received glycoprotein subunits 120 or 160 as a prime-boost methodology. Research is ongoing to improve upon the RV144 trial, studying alternative priming and dosing schedules, different vectors and adjuvants, and participants’ immune responses.27
Epitopes are antigenic determinants (parts of an antigen that are recognized by the immune system). One focus of new vaccine development is on engineering novel epitope vaccines that are capable of inducing long-term protective immunity by using computational tools for vaccine design and evaluation.28 Another promising area of research involves the use of bNAbs, which appear in patients after 2 to 4 years of HIV infection. Studies examining passive transfer of bNAbs to prevent viral infection in animals have yielded positive results.27 Alternative routes of vaccine delivery, such as mucosal administration, are also being explored.
Despite considerable progress in vaccine development, there is no effective vaccine to date for preventing HIV infection. TABLE 1 summarizes some pivotal HIV vaccine efficacy trials.29-37 A safe and effective vaccine must induce specific CD4+ T-cell responses, which will enhance CD8+ T-cell and humoral immune responses.
Hindrances to Development of an Effective HIV Vaccine
Historically, vaccine development for HIV prevention has been extremely difficult. The human immune system does not easily recognize the virus and initiate a response. The virus also quickly mutates while multiplying in the body. This requires a robust vaccine that can initiate antibody production against a wide range of HIV strains.38
HIV-1 and HIV-2 have many differences, including several different subtypes,4 which makes vaccine development challenging. In addition, there is a high mutation rate of HIV reverse transcriptase, the enzyme used to transcribe the viral genome during replication. Technical difficulties in trial design, such as mucosal sample collection and processing, must be addressed in order to achieve the best evaluation of novel vaccines. Many studies have focused on developing a vaccine against HIV-1; these vaccines also need to be tested for prevention of HIV-2 infection. Some patients develop simultaneous HIV-1 and HIV-2 infections, so research is focusing on understanding the mechanism of this comorbidity. Since HIV-2–infected patients are located mainly in Africa, an efficacious yet cost-effective, affordable, and safe vaccine is the key to controlling the disease. The stigma surrounding HIV disease must be addressed to encourage robust participation in trials.
Conclusion
HIV infection is a major public-health concern. The use of ART merely extends the lifespan of HIV-infected patients, and disease eradication is not yet possible. A safe and cost-effective vaccine that prevents HIV infection is considered the best strategy for containing the epidemic. Despite all the challenges of vaccine development, the RV144 trial proved that an HIV vaccine can be a reality. Additional studies are needed to develop a vaccine with optimal safety and efficacy to positively affect HIV disease prevention and control, as well as patient health and survival.
REFERENCES
1. World Health Organization. HIV/AIDS. www.who.int/mediacentre/factsheets/fs360/en. Accessed August 24, 2015.
2. Tong L, Pav S, Pargellis C, et al. Crystal structure of human immunodeficiency virus (HIV) type 2 protease in complex with a reduced amide inhibitor and comparison with HIV-1 protease structures. Proc Natl Acad Sci U S A. 1993;90:8387-8391.
3. Whittle HC, Ariyoshi K, Rowland-Jones S. HIV-2 and T cell recognition. Curr Opin Immunol. 1998;10:382-387.
4. Sharp PM, Hahn BH. Origins of HIV and the AIDS pandemic. Cold Spring Harb Perspect Med. 2011;1:a006841.
5. CDC. HIV Surveillance Report, 2013. Vol. 25. Published February 2015. www.cdc.gov/hiv/library/reports/surveillance. Accessed September 10, 2015.
6. AIDS.gov. What is HIV/AIDS? www.aids.gov/hiv-aids-basics/hiv-aids-101/what-is-hiv-aids. Accessed August 19, 2015.
7. Douek DC, Brenchley JM, Betts MR, et al. HIV preferentially infects HIV-specific CD4+ T cells. Nature. 2002;417:95-98.
8. Li S, Juarez J, Alali M, et al. Persistent CCR5 utilization and enhanced macrophage tropism by primary blood human immunodeficiency virus type 1 isolates from advanced stages of disease and comparison to tissue-derived isolates. J Virol. 1999;73:9741-9755.
9. Mehandru S, Tenner-Racz K, Racz P, Markowitz M. The gastrointestinal tract is critical to the pathogenesis of acute HIV-1 infection. J Allergy Clin Immunol. 2005;116:419-422.
10. Kalams SA, Buchbinder SP, Rosenberg ES, et al. Association between virus-specific cytotoxic T-lymphocyte and helper responses in human immunodeficiency virus type 1 infection. J Virol. 1999;73:6715-6720.
11. Rosenberg ES, Billingsley JM, Caliendo AM, et al. Vigorous HIV-1-specific CD4+ T cell responses associated with control of viremia. Science. 1997;278:1447-1450.
12. Yang OO, Walker BD. CD8+ cells in human immunodeficiency virus type I pathogenesis: cytolytic and noncytolytic inhibition of viral replication. Adv Immunol. 1997;66:273-311.
13. Goonetilleke N, Liu MK, Salazar-Gonzalez JF, et al. The first T cell response to transmitted/founder virus contributes to the control of acute viremia in HIV-1 infection. J Exp Med. 2009;206:1253-1272.
14. Letvin NL, Walker BD. Immunopathogenesis and immunotherapy in AIDS virus infections. Nat Med. 2003;9:861-866.
15. Goméz-Román VR, Patterson LJ, Venzon D, et al. Vaccine-elicited antibodies mediate antibody-dependent cellular cytotoxicity correlated with significantly reduced acute viremia in rhesus macaques challenged with SIVmac251. J Immunol. 2005;174:2185-2189.
16. Forthal DN, Landucci G, Daar ES. Antibody from patients with acute human immunodeficiency virus (HIV) infection inhibits primary strains of HIV type 1 in the presence of natural-killer effector cells. J Virol. 2001;75:6953-6961.
17. Richman DD, Wrin T, Little SJ, Petropoulos CJ. Rapid evolution of the neutralizing antibody response to HIV type 1 infection. Proc Natl Acad Sci U S A. 2003;100:4144-4149.
18. AIDSinfo. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. https://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-treatment-guidelines/0. Accessed August 19, 2015.
19. Fletcher C. Human immunodeficiency virus infection. In: DiPiro JT, Talbert RL, Yee G, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 9th ed. New York, NY: McGraw-Hill Education; 2014:2034.
20. Michaels SH, Clark R, Kissinger P. Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. N Engl J Med. 1998;339:405-406.
21. AVAC. HIV prevention research & development funding trends, 2000–2014. www.avac.org/sites/default/files/resource-files/ResourceTracking2015.pdf. Accessed September 10, 2015.
22. HIV Vaccine Trials Network. Types of vaccines. www.hvtn.org/en/science/hiv-vaccine-basics/types-vaccines.html. Accessed August 19, 2015.
23. van Gils MJ, Sanders RW. Broadly neutralizing antibodies against HIV-1: templates for a vaccine. Virology. 2013;435:46-56.
24. Chhatbar C, Mishra R, Kumar A, Singh SK. HIV vaccine: hopes and hurdles. Drug Discov Today. 2011;16:948-956.
25. Robinson HL. HIV/AIDS vaccines: 2007. Clin Pharmacol Ther. 2007;82:686-693.
26. Rerks-Ngarm S, Pitisuttithum P, Nitayaphan S, et al. Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N Engl J Med. 2009;361:2209-2220.
27. Fauci AS, Marston HD. Toward an HIV vaccine: a scientific journey. Science. 2015;349:386-387.
28. He L, Zhu J. Computational tools for epitope vaccine design and evaluation. Curr Opin Virol. 2015;11:103-112.
29. Buchbinder SP, Mehrotra DV, Duerr A, et al. Efficacy assessment of a cell-mediated immunity HIV-1 vaccine (the Step Study): a double-blind, randomised, placebo-controlled, test-of-concept trial. Lancet. 2008;372:1881-1893.
30. Lewis GK, DeVico AL, Gallo RC. Antibody persistence and T-cell balance: two key factors confronting HIV vaccine development. Proc Natl Acad Sci U S A. 2014;111:15614-15621.
31. Pitisuttithum P, Berman PW, Phonrat B, et al. Phase I/II study of a candidate vaccine designed against the B and E subtypes of HIV-1. J Acquir Immune Defic Syndr. 2004;37:1160-1165.
32. Haynes BF, Gilbert PB, McElrath MJ, et al. Immune-correlates analysis of an HIV-1 vaccine efficacy trial. N Engl J Med. 2012;366:1275-1286.
33. Hammer SM, Sobieszczyk ME, Janes H, et al. Efficacy trial of a DNA/rAd5 HIV-1 preventive vaccine. N Engl J Med. 2013;369:2083-2092.
34. Tomaras GD, Ferrari G, Shen X, et al. Vaccine-induced plasma IgA specific for the C1 region of the HIV-1 envelope blocks binding and effector function of IgG. Proc Natl Acad Sci U S A. 2013;110:9019-9024.
35. Haynes BF. New approaches to HIV vaccine development. Curr Opin Immunol. 2015;35:39-47.
36. McElrath MJ, De Rosa SC, Moodie Z, et al. HIV-1 vaccine-induced immunity in the test-of-concept Step Study: a case-cohort analysis. Lancet. 2008;372:1894-1905.
37. Gray G, Buchbinder S, Duerr A. Overview of STEP and Phambili trial results: two phase IIb test-of-concept studies investigating the efficacy of MRK adenovirus type 5 gag/pol/nef subtype B HIV vaccine. Curr Opin HIV AIDS. 2010;5:357-361.
38. Collins F. Vaccine research: new tactics for tackling HIV. http://directorsblog.nih.gov/2015/06/30/vaccine-research-new-tactics-for-tackling-hiv. Accessed September 10, 2015.
To comment on this article, contact rdavidson@uspharmacist.com.





