Restless legs syndrome (RLS) is a common sensorimotor disorder of the central nervous system. It is characterized by an irresistible urge or need to move the limbs that occurs as a result of uncomfortable limb sensations. Although RLS usually starts in the legs, it can spread to other parts of the body, such as the arms or torso. RLS sufferers often describe the disorder as feeling like water flowing inside the legs, "bugs in the bones," or electricity in the legs. These symptoms may be present all day, but usually worsen during periods of inactivity and in the evening. People with RLS usually must move their limbs for complete or partial relief of the uncomfortable sensations. At night, RLS often is associated with periodic limb movements (PLMs), which can cause sleep problems and also make it difficult to perform sedentary activities such as sitting comfortably in a car or on an airplane for extended periods.
The cause of RLS remains unclear, but research suggests that impaired dopamine activity in the brain due to iron insufficiency may induce RLS in people with a genetic predisposition. RLS can be classified as primary or as secondary due to another medical condition, such as end-stage renal disease (ESRD) or pregnancy. Dopamine agonists like ropirinole and pramipexole are first-line therapy for RLS with daily symptoms. Alternative therapies are available for refractory cases or persistent symptoms. This article explores the prevalence, etiology, and diagnosis of RLS, as well as available treatments.
Prevalence
Epidemiologic studies report that 7.2% to 11% of the general population in Europe and North America suffers from some form of RLS.1,2 In the United States alone, RLS is believed to affect more than 10 million adults and an estimated 1.5 million children and adolescents, with similar rates between boys and girls.1,3 Women are approximately twice as susceptible to RLS as men (9% vs. 5.4%).1,2 RLS is more common in older adults, but after age 79 its incidence tends to decrease.1 Clinically significant RLS cases exist in 2.7% to 3 % of the population.1,2 Despite its high prevalence, this syndrome remains underdiagnosed and troubling for many patients.
Etiology
The etiology of RLS is not fully understood. Studies have demonstrated that it is not a structural neurodegenerative problem, but rather involves the dopamine system, iron homeo stasis, or genetics. The following discussion sums up current thought on each of these three potential causes.
Dopamine System: Attempts to show that the dopamine system is abnormal in people with RLS have not yielded definitive results. Findings from neuroimaging studies suggest dopamine dysfunction, rather than insufficiency.4 Dopamine's involvement in RLS is supported by the clinical response to dopamine agonists in the treatment of primary RLS.5,6 The exact mechanism of action of dopamine agonists in alleviating RLS symptoms is unknown.
Iron Homeostasis: An autopsy study demonstrated reduced iron stores in the substantia nigra of patients with RLS.7 Thus, one possible cause for RLS might be the inability of the brain to maintain normal iron levels. Iron is also a cofactor in the production of dopamine. Another study demonstrated a possible link between impaired iron homeostasis and dopamine dysfunction.8 Further studies are needed to determine the role of iron in RLS.
Genetics: Clinical experience has shown a strong familial connection in RLS. Several loci have been reported to date, but a disease-causing gene has not yet been identified.9,10 It is generally accepted that RLS follows an autosomal-dominant mode of inheritance that requires only one affected parent to pass the trait to the offspring. More genetics studies are needed to shed light on the genotype of RLS sufferers.
Diagnosis
The diagnosis of RLS is based entirely upon interviews with the patient and symptom history. Diagnostic criteria were established in 1995 by the International Restless Legs Syndrome Study Group (IRLSSG) and revised in 2003.11,12 They are divided into essential, supportive, and associated criteria (TABLE 1). All four essential criteria must be present to establish a diagnosis of RLS. The supportive clinical features assist in the diagnosis of more complicated cases. Associated features are present in the majority of RLS cases and can further guide clinicians in diagnosis.

Primary RLS
Primary RLS has been identified as having a more insidious symptom onset than secondary RLS. It is more common in people younger than age 40. These patients also are more likely to have other family members with the disorder. A critical step in treating patients with primary RLS is to establish the frequency and severity of their symptoms. One treatment algorithm divides primary RLS into three categories according to symptom severity13:
Intermittent RLS requires occasional treatment of symptoms, but is not severe enough to necessitate daily medication therapy. People with intermittent RLS have milder symptoms and are easier to treat. Nondrug treatment is usually the preferred first-line approach.
Daily RLS is characterized by persistent symptoms that have to be managed with daily medication therapy. Drug therapy is the treatment of choice.
Refractory RLS is daily RLS that fails to respond to treatment with at least one first-line drug at the usual dose. At least one of the following criteria needs to be met: inability to achieve adequate medication response; unsatisfactory response over time despite dose increases of the preferred agent; intolerable side effects; and worsening symptoms despite additional doses of medication.
Secondary RLS
Secondary RLS is associated with a more abrupt onset and typically occurs in conjunction with or as a result of another condition, such as iron deficiency, ESRD, or pregnancy. The symptoms of secondary RLS are not distinguishable from those of primary RLS, but treatment targets the underlying condition.
Treatment of Primary RLS
Nonpharmacologic Therapy
In mild RLS, nonpharmacologic approaches should be tried first. In all other cases, nondrug therapy can be used in addition to drug therapy to relieve sensorimotor symptoms and sleep disturbances. Nonpharmacologic treatment includes developing good sleep habits, such as having a regular sleep schedule; ensuring a quiet, dark environment in the bedroom; and avoiding RLS triggers (television, caffeine, nicotine, alcohol) close to bedtime.13 Mild-to-moderate mental activities (video games, puzzles) and physical activities (housework, exercise) are recommended during times when patients are most prone to exhibiting symptoms, such as in the evening and during periods of inactivity or rest. Sedentary activities such as airplane flights, long car rides, and movies are recommended for the morning, when RLS symptoms tend to subside in intensity.
Pharmacologic Therapy
Pharmacologic therapy for RLS is symptomatic. It does not cure the disorder, but it manages its troubling manifestations. Pharmacologic agents used for the treatment of RLS are levodopa (l-dopa), dopamine agonists, gabapentin, opioids, and benzodiazepines.
l-Dopa: l-Dopa is a dopamine precursor. It is formulated together with the decarboxylase inhibitor carbidopa (Sinemet), which prevents the peripheral plasma breakdown of l-dopa. Carbidopa helps reduce the amount of l-dopa needed by 75%. Once l-dopa crosses the blood-brain barrier, it is converted to dopamine. For Parkinson's disease, carbidopa/l-dopa therapy is generally initiated at 25 mg/100 mg three times daily; for intermittent RLS, the combination of l-dopa and carbidopa has been used in lower doses on an as-needed basis due to its immediate action and lack of need for titration.13 Sustained-release formulations of l-dopa with longer half-lives have been investigated for alternative use in patients who are frequently awakened by symptoms at night owing to the short action of the immediate-release form.14 There are currently no single-entity l-dopa products on the U.S. market.
In RLS patients, l-dopa produces typical dopaminergic side effects such as nausea, vomiting, somnolence, dizziness, hypotension, and headache. The most detrimental side effect, augmentation, prevents its use in daily RLS.15 Augmentation involves a worsening of RLS symptoms earlier in the day after an evening dose of medication, an increase in symptom severity, and the spread of symptoms to other parts of the body, such as arms or torso.13
Dopamine Agonists: Dopamine agonists are first-line therapy for daily RLS.13 Nonergot agonists such as ropinirole and pramipexole are preferred to ergot agonists such as pergolide and cabergoline because of their more favorable side-effect profiles. Pergolide and cabergoline, which have been shown to increase the risk of cardiac-valve regurgitation, currently are not available in the U.S. because of this harmful adverse effect.16
Ropinirole (Requip) was the first medication approved by the FDA (in 2005) for the treatment of moderate-to-severe primary RLS. It is a full intrinsic nonergoline agonist for dopamine D2 and D3 receptor subtypes. Three large clinical trials have demonstrated ropinirole's efficacy and tolerability in treating RLS.5,17,18 In all three studies, patients were randomized to ropinirole or placebo on a flexible titration schedule of 0.25 mg to 4 mg/day as tolerated, taken once daily one to three hours before bedtime. Results were assessed with the International RLS Rating Scale (IRLS)--the primary outcome measure--and the Clinical Global Impressions-Improvement (CGI-I) scale. The IRLS comprises 10 sections that evaluate the severity of sensory and motor symptoms, sleep disturbances, impact on activities of daily living, and mood associated with RLS. Based on the IRLS, all three trials found that ropinirole effectively reduced the core symptoms of RLS--such as symptom severity and frequency, as well as sleep problems--and effectively increased overall well-being. The most commonly reported side effects were nausea and headache, typical for dopamine agonists. Augmentation was not formally assessed in these studies, and no reports of disease augmentation were documented.
Pramipexole (Mirapex), approved by the FDA in 2006 for the treatment of moderate-to-severe daily RLS, is another full nonergoline D2 and D3 receptor agonist. The efficacy of pramipexole was evaluated in several clinical trials.6,19,20 All patients received pramipexole (0.125 mg-0.75 mg) as needed and tolerated or placebo once daily two to three hours before bedtime. Across all studies, the two outcome measures used to assess efficacy were the IRLS and the CGI-I. Improvements were seen in both subjective and objective measures of RLS. Commonly reported adverse effects were nausea, headache, and fatigue. Augmentation was not formally evaluated, although some documented reports of augmentation with pramipexole appeared in retrospective studies.21,22
Other Treatments: Gabapentin, benzodiazepines, and opioids are helpful second-line agents that are appropriate alternative therapy for RLS cases involving specific symptoms like continuous sleep disturbances or painful sensations in the extremities.
Most research concerning RLS has been on the anticonvulsant gabapentin (Neurontin), which has been shown to be useful for RLS and peripheral neuropathy.23,24 Gabapentin, a structural analogue of gamma-aminobutyric acid, works by reducing the subjective symptoms of RLS.23 Because it can cause somnolence, gabapentin can be tried as an adjunct agent for RLS that involves persistent sleep problems.24 Additional studies are needed to examine the long-term effects of chronic monotherapy with gabapentin.
Relatively few published studies exist on the use of benzodiazepines for RLS. Only clonazepam and alprazolam have been investigated in clinical trials. Because of their sedative properties, these two agents have been used mainly to induce sleep. A small study of clonazepam showed that the drug was effective for nocturnal RLS because of its ability to induce and maintain sleep; however, it did not reduce PLMs during sleep.25 Higher incidences of daytime sleepiness and cognitive disturbances reportedly were linked to the longer action of the drug. Overall, benzodiazepines are considered a second-line option for RLS that involves significant insomnia.26
How opioid analgesics work in RLS is unknown. One retrospective clinical trial of patients receiving long-term opioid monotherapy (oxycodone, propoxyphene, codeine, or methadone) for primary RLS demonstrated that opioids were very effective for alleviating motor symptoms and sleep disturbances and produced virtually no tolerance or physical dependence (occurring in just one of 36 monotherapy patients).27 The authors recommended selecting an opioid based on the severity of the patient's complaints. Another study suggested that tramadol (Ultram) has a lower abuse potential and fewer side effects than other opioids and that it should be the opioid of choice for the treatment of RLS.28 In severe, refractory cases, oral methadone is recommended because of its long half-life.29
Treatment of Secondary RLS
Some patients develop RLS as a consequence of another condition. Up to 26% of pregnant women are affected by RLS symptoms, especially in the third trimester.30 A high prevalence of RLS occurs in people with ESRD and in those with iron deficiency.31,32 Treatment of secondary RLS focuses on identifying and treating the underlying cause. In pregnancy, therapy is withheld since women experience complete reversal of symptoms after giving birth. Kidney transplantation--but not dialysis--is curative for RLS in ESRD. Intravenous iron can be administered if iron stores are low.33 Clinicians should evaluate patients for other substances that have the potential to aggravate RLS symptoms, such as metoclopromide, all neuroleptics, many antidepressants, and antihistamines. These agents should be discontinued, if possible.
Pharmacist's Role in RLS
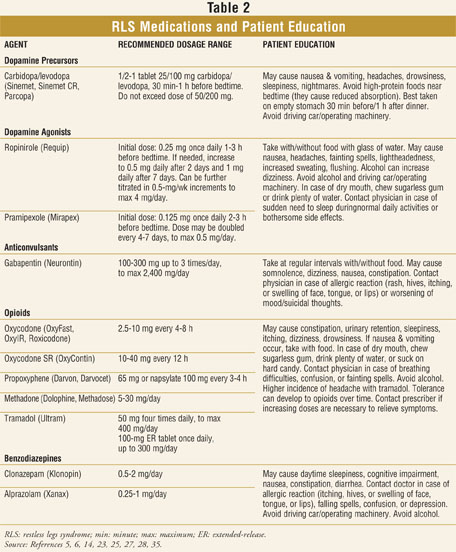
Pharmacists should be knowledgeable about and able to recognize the typical signs and symptoms of RLS, such as unpleasant sensations in the calves, thighs, feet, or even arms. These sensations are often described by patients as "crawling," "creeping," "tingling," "jittery," "deep-seated," or "burning." Although the description of RLS can vary significantly among patients, the common element is a desire or need to move the extremities that worsens in the evenings and causes difficulty in falling asleep or staying asleep. People affected by RLS also may complain of fatigue, depressed mood, or lack of concentration.34 Pharmacists may consider recommending that patients with RLS symptoms speak to their primary care physician regarding referral to a neurologist or an appropriate sleep specialist based on their physician's assessment and recommendation. It also is important for pharmacists to be familiar with the most common medications for RLS and to offer adequate counseling on their proper use and possible adverse effects (TABLE 2).
Various online resources exist that can provide additional information about RLS. The RLS Foundation ( www.rls.org) is the best overall source of RLS information. The site contains a list of support groups, a chat room, up-to-date reviews of RLS treatments, and expert advice. Other online resources are WE MOVE ( www.wemove.org) and Talk About Sleep ( www.talkaboutsleep.com).
REFERENCES
1. Allen RP, Walters AS, Montplaisir J, et al. Restless legs syndrome prevalence and impact: REST general population study. Arch Intern Med. 2005;165:1286-1292.
2. Hening W, Walters AS, Allen RP, et al. Impact, diagnosis and treatment of restless legs syndrome (RLS) in a primary care population: the REST (RLS epidemiology, symptoms, and treatment) primary care study. Sleep Med. 2004;5:237-246.
3. Picchietti D, Allen RP, Walters AS, et al. Restless legs syndrome: Prevalence and impact in children and adolescents--the Peds REST study. Pediatrics. 2007;120:253-266.
4. Wetter TC, Eisensehr I, Trenkwalder C. Functional neuroimaging studies in restless legs syndrome. Sleep Med. 2004;5:401-406.
5. Trenkwalder C, Garcia-Borreguero D, Montagna P, et al. Ropinirole in the treatment of restless legs syndrome: results from the TREAT RLS 1 study, a 12 week, randomised, placebo controlled study in 10 European countries. J Neurol Neurosurg Psychiatry. 2004;75:92-97.
6. Winkelman JW, Sethi KD, Kushida CA, et al. Efficacy and safety of pramipexole in restless legs syndrome. Neurology. 2006;67:1034-1039.
7. Connor JR, Boyer PJ, Menzies SL, et al. Neuropathological examination suggests impaired brain iron acquisition in restless legs syndrome. Neurology. 2003;61:304-309.
8. Wang X, Wiesinger J, Beard J, et al. Thy1 expression in the brain is affected by iron and is decreased in restless legs syndrome. J Neurol Sci. 2004;220:59-66.
9. Mathias RA, Hening W, Washburn M, et al. Segregation analysis of restless legs syndrome: possible evidence for a major gene in a family study using blinded diagnoses. Hum Hered.
10. Desautels A, Turecki G, Xiong L, et al. Mutational analysis of neurotensin in familial restless legs syndrome. Mov Disord. 2004;19:90-94.
11. Walters AS. Toward a better definition of the restless legs syndrome. The International Restless Legs Syndrome Study Group. Mov Disord. 1995;10:634-642.
12. Allen RP, Picchietti D, Hening WA, et al. Restless legs syndrome: diagnostic criteria, special considerations, and epidemiology. A report from the restless legs syndrome diagnosis and epidemiology workshop at the National Institutes of Health. Sleep Med. 2003;4:101-119.
13. Silber MH, Ehrenberg BL, Allen RP, et al. An algorithm for the management of restless legs syndrome. Mayo Clin Proc. 2004;79:916-922.
14. Trenkwalder C, Collado Seidel V, Kazenwadel J, et al. One-year treatment with standard and sustained-release levodopa: appropriate long-term treatment of restless legs syndrome? Mov Disord.
15. Allen RP, Earley CJ. Augmentation of the restless legs syndrome with carbidopa/levodopa. Sleep.
16. Schade R, Andersohn F, Suissa S, et al. Dopamine agonists and the risk of cardiac-valve regurgitation. N Engl J Med. 2007;356:29-38.
17. Walters AS, Ondo WG, Dreykluft T, et al. Ropinirole is effective in the treatment of restless legs syndrome. TREAT RLS 2: a 12-week, double-blind, randomized, parallel-group, placebo-controlled study. Mov Disord. 2004;19:1414-1423.
18. Bogan RK, Fry JM, Schmidt MH, et al. Ropinirole in the treatment of patients with restless legs syndrome: a US-based randomized, double-blind, placebo-controlled clinical trial. Mayo Clin Proc.
19. Oertel WH, Stiasny-Kolster K, Bergtholdt B, et al. Efficacy of pramipexole in restless legs syndrome: a six-week, multicenter, randomized, double-blind study (effect-RLS study). Mov Disord.
20. Trenkwalder C, Stiasny-Kolster K, Kupsch A, et al. Controlled withdrawal of pramipexole after 6 months of open-label treatment in patients with restless legs syndrome. Mov Disord.
21. Ferini-Strambi L. Restless legs syndrome augmentation and pramipexole treatment. Sleep Med.
22. Winkelman JW, Johnston L. Augmentation and tolerance with long-term pramipexole treatment of restless legs syndrome (RLS). Sleep Med. 2004;5:9-14.
23. Happe S, Klösch G, Saletu B, Zeitlhofer J. Treatment of idiopathic restless legs syndrome (RLS) with gabapentin. Neurology. 2001;57:1717-1719.
24. Garcia-Borreguero D, Larrosa O, de la Llave Y, et al. Treatment of restless legs syndrome with gabapentin: a double-blind, cross-over study. Neurology. 2002;59:1573-1579.
25. Saletu M, Anderer P, Saletu-Zyhlarz G, et al. Restless legs syndrome (RLS) and periodic limb movement disorder (PLMD): acute placebo-controlled sleep laboratory studies with clonazepam. Eur Neuropsychopharmacol. 2001;11:153-161.
26. Schenck CH, Mahowald MW. Long-term, nightly benzodiazepine treatment of injurious parasomnias and other disorders of disrupted nocturnal sleep in 170 adults. Am J Med.
27. Walters AS, Winkelmann J, Trenkwalder C, et al. Long-term follow-up on restless legs syndrome patients treated with opioids. Mov Disord. 2001;16:1105-1109.
28. Lauerma H, Markkula J. Treatment of restless legs syndrome with tramadol: an open study. J Clin Psychiatry. 1999;60:241-244.
29. Ondo WG. Methadone for refractory restless legs syndrome. Mov Disord. 2005;20:345-348.
30. Manconi M, Govoni V, De Vito A, et al. Restless legs syndrome and pregnancy. Neurology.
31. Gigli GL, Adorati M, Dolso P, et al. Restless legs syndrome in end-stage renal disease. Sleep Med. 2004;5:309-315.
32. Sun ER, Chen CA, Ho G, et al. Iron and the restless legs syndrome. Sleep. 1998;21:371-377.
33. Earley CJ, Heckler D, Allen RP. Repeated IV doses of iron provides effective supplemental treatment of restless legs syndrome. Sleep Med. 2005;6:301-305.
34. Kushida C, Martin M, Nikam P, et al. Burden of restless legs syndrome on health-related quality of life. Qual Life Res. 2007;16:617-624.
35. Facts & Comparisons. Facts & Comparisons 4.0 [subscription required].
www.factsandcomparisons.com. Accessed August 31, 2008. 2006;62:157-164. 2003;18:1184-1189. 1996;19:205-213. 2006;81:17-27. 2007;22:213-219. 2006;21:1404-1410. 2002;3(suppl):S23-S25. 1996;100:333-337. 2004;63:1065-1069.





