US Pharm. 2019;44(3):28-32.
ABSTRACT: Rheumatoid arthritis (RA) is a chronic disease affecting up to 1% of populations in developed countries. Certain genetic and environmental factors appear to promote the development of RA, but patients can present with heterogeneous signs and symptoms. The goals of any treatment for RA are the reduction of inflammation and remission of disease. Historic treatments, such as aspirin and colloidal gold, were inadequate and did not affect the course of RA. In current practice, there are several biological and synthetic disease-modifying antirheumatic drugs that can reduce disease severity. Guidelines provide point systems for disease classification, stepwise algorithms for treatment, and recommendations for follow-up and monitoring based on response and comorbidities. As RA is a long-term condition, pharmacists can work together with providers to ensure proper drug dosing, titration, and monitoring.
Rheumatoid arthritis (RA) is an autoimmune, inflammatory disease that causes pain, swelling, stiffness, and loss of function in various joints (most commonly in the hands, wrists, and knees).1-3 The respective joint’s lining becomes inflamed, leading to tissue damage, as well as chronic pain, unsteadiness, and deformity.3 There is generally a bilateral/symmetrical pattern of disease progression (e.g., both hands or both knees are affected).2 RA can also affect extra-articular sites, including the eyes, mouth, lungs, and heart.1 Patients can experience an acute worsening of their symptoms—disease flare—but with early intervention and appropriate treatment, symptoms can be ameliorated for a certain duration (disease remission).3,4
EPIDEMIOLOGY
In 2005, RA was prevalent in about 1.3 million adults in the United States, and 2 years later, it affected an estimated 1.5 million adults.2,5 More recent data on RA prevalence in the U.S. are not available yet in the literature. RA can occur in all races and ethnic groups.2 The prevalence of RA in developed countries is 0.5% to 1% of the population (0.6% in the U.S.).5-7 Women have a two- to- three times greater predisposition for developing RA compared with men.1,2,5 RA onset generally occurs in middle age and is more common in older adults, but it can also develop in children and young adults.1,2 The lifetime risk of developing an inflammatory autoimmune rheumatic disease is 1 in 12 (8.3%) for women and 1 in 20 (5%) for men.5 Specifically, the lifetime risk of developing adult-onset RA is 1 in 28 (3.6%) for women and 1 in 59 (1.7%) for men.8 Over time, RA severity has declined, particularly owing to earlier diagnosis and more effective drug regimens, but trends in RA incidence, prevalence, and mortality vary based on the studied population.5,6
ETIOLOGY
The exact cause of RA is still unknown, but genes, environmental factors, and hormones may be involved in its autoimmune development and progression.1-3 Certain risk factors appear to increase the risk of RA, including older age (highest incidence in people aged 60 years); gender (higher incidence in women); genetics (especially human leukocyte antigen [HLA] class II genotypes, such as HLA-DRB1); smoking (tobacco, cigarettes); history of live births (higher RA risk with nulliparity); early life exposures (if mother smoked, child has greater risk of RA); and obesity (higher risk with increasing body weight).3,7,8 Patients who are seropositive for anticitrullinated protein antibodies (ACPAs) or rheumatoid factors (RFs) also have an increased risk of RA.7 Interestingly, women who breastfeed their children appear to have a lower risk of RA.3 Before the advent of effective disease-modifying antirheumatic drugs (DMARDs) and biological therapies, patients with RA had a higher likelihood of dying from premature atherosclerosis, cancer, and infection.2
PATHOPHYSIOLOGY
Joint swelling in RA is usually synovial-membrane inflammation, with cytokine and chemokine involvement.7 The most relevant components in the inflamed space include tumor necrosis factor (TNF), interleukin-6 (IL-6), and granulocyte-macrophage colony-stimulating factor.7 Cytokines and chemokines induce or aggravate the inflammatory response by activating endothelial cells and promoting immune-system cell accumulation within the synovial compartment.7 Activated fibroblasts, B cells, T cells, monocytes, and macrophages can eventually trigger osteoclast generation via receptor activator of nuclear factor kappa-B ligand (RANKL), which is expressed on B cells, T cells, and fibroblasts.7 The RANK receptor is present on macrophages, dendritic cells, and preosteoclasts.7 In addition, the cartilage matrix within joints is eventually degraded by metalloproteinases and other enzymes.7
DIAGNOSIS
In RA, signs and symptoms may overlap with other rheumatic diseases, but classification criteria can assist with diagnosis.7,8 Patients with RA will generally complain of multiarticular pain/aching, morning stiffness, tenderness/swelling, and bilateral/symmetrical joint involvement (e.g., both hands, both knees).3 Patients may also present with weight loss, fever, fatigue, and/or weakness.3 In addition to physical symptoms, the laboratory diagnosis (measurable sign) of RA has improved with the identification of highly specific biomarkers.2 Along with an elevated C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR), the presence of autoantibodies (e.g., ACPA, RF) typically indicates more severe joint damage and increased mortality.7 Of note, RF is directly involved in mechanisms related to cytokine and macrophage activation.7 ACPAs form immune complexes that interact with RF, enhancing the effects of inflammation and subsequent joint destruction.7 While on treatment for RA, the concentrations of ACPA and RF should decrease.7 Patients will rarely become ACPA seronegative, but they can become RF seronegative.7
These findings and laboratory values are incorporated in the classification criteria for RA, with joint involvement, serology, acute-phase reactants, and symptom duration as the main categories for consideration.9 An increasing number of affected joints (≥1 small joint), positivity of RF and/or ACPA, abnormal ESR and/or CRP, and duration of symptoms (≥6 weeks) are scored and calculated, with a score of ≥6 out of 10 being a definitive RA diagnosis.9
TREATMENT
Reversal of inflammation is the typical goal of therapy for RA, and without adequate treatment, patients can develop irreversible disabilities.7,8 Interventions for RA include drugs, lifestyle modifications, and surgical procedures, all of which can ameliorate joint damage and reduce pain and swelling.1 With each approach, there should also be a regular assessment of response (via disease activity), permitting a strategy of adaptation or treating to target.7 Mild-to-moderate RA can be adequately treated in certain patients: The disease can be controlled without flares.1 On the other hand, in severe RA, the signs/symptoms may be more long-term.1
Historic treatments for RA included aspirin and colloidal gold.2 These therapies relieved symptoms, but did not significantly slow or modify disease progression.2 Steroids permit rapid symptom improvement and some disease-modifying benefits, but long-term use is typically associated with serious adverse effects (e.g., hypertension, diabetes, osteoporosis, and cataracts).2,7 Cyclosporine had also been used, but it can be insufficient or ineffective in certain patients, with various side effects.2 Nonsteroidal anti-inflammatory drugs (NSAIDs; e.g., ibuprofen, naproxen) were included as possible treatments for RA.These will reduce pain, swelling, and stiffness, as well as improve physical function, but have no discernable effect on joint damage (i.e., NSAIDs are not DMARDs).7 These drugs are no longer preferred, according to recent guidelines.4
There is now a better understanding of the mechanisms/pathways involved in RA, permitting site-specific drug development.2 There are two major types of DMARDs: biological and synthetic (see Table 1).7 Conventional, synthetic DMARDs (csDMARDs) are not site-specific, and they have unclear mechanisms of action for relieving RA.7 On the other hand, targeted, synthetic DMARDs focus on a specific site (e.g., inhibiting the Janus kinase [JAK] enzyme).7 Biological DMARDs are also site-specific (e.g., targeting TNF, IL-6, IL-1, B cells, or T cells).2,7
GUIDELINES
Two major groups have generated RA guidelines: the American College of Rheumatology (ACR) and the European League Against Rheumatism (EULAR).4,10 The ACR guideline has algorithms for early and established RA, while the EULAR guideline uses a three-phase approach.4,10 In addition, “DMARD” in the ACR guideline implies csDMARD.
ACR 2015
For early RA (symptom duration <6 months), regardless of disease activity level, a treat-to-target strategy is preferable over a nontargeted approach.4 See Figure 1 for an abbreviated treatment algorithm for early RA.4
Regardless of disease activity (low/moderate/high) in DMARD-naïve patients, DMARD monotherapy (methotrexate [MTX] preferable) should be used, instead of upfront double or triple DMARD therapy.4 If there is moderate-to-high disease activity (MHDA) despite DMARD monotherapy (+/- steroids), DMARD combinations or TNF inhibitor (TNFi) (+/-MTX) or non-TNFi (+/- MTX) should be used.4 Of note, TNFi monotherapy is preferable over tofacitinib, and TNFi + MTX is preferable over tofacitinib + MTX.4
For established RA (symptom duration ≥6 months), regardless of disease activity, a treat-to-target strategy is still preferable over a nontargeted approach.4 See Figure 2 for an abbreviated treatment algorithm for established RA.4
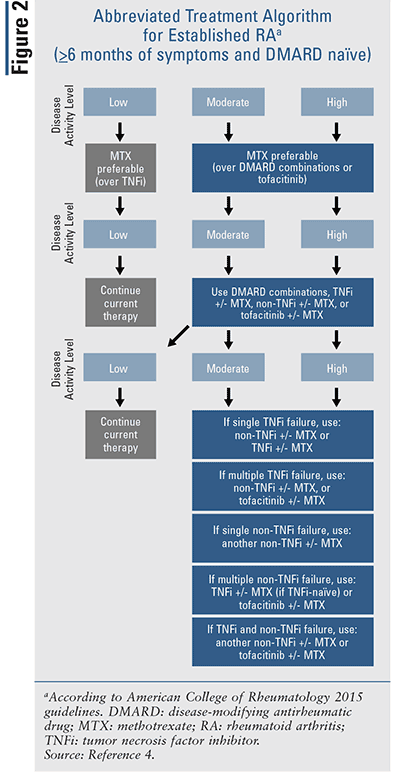
If disease activity is low in DMARD-naïve patients, DMARD monotherapy (MTX preferable) should be used over a TNFi.4 If MHDA occurs in DMARD-naïve patients, DMARD monotherapy (MTX preferable) should be used over DMARD combinations or tofacitinib.4
If MHDA occurs despite DMARD monotherapy, DMARD combinations or a TNFi (+/- MTX) or non-TNFi (+/- MTX) or tofacitinib (+/- MTX) can be used.4
If MHDA occurs despite aTNFi in DMARD-naïve patients, DMARDs (≤2) can be combined with TNFi.4 Alternatively, a non-TNFi (+/- MTX) can be used, instead of another TNFi (+/- MTX) or tofacitinib (+/- MTX).4 If MHDA occurs despite non-TNFi monotherapy, another non-TNFi (+/- MTX) can be used, instead of tofacitinib (+/- MTX).4 If MHDA occurs despite multiple (>2) sequential TNFi therapies, a non-TNFi (+/- MTX) can be used initially, instead of another TNFi or tofacitinib.4
If a non-TNFi cannot be used, and if MHDA occurs despite multiple TNFi, tofacitinib (+/- MTX) can be used, instead of another TNFi (+/- MTX).4 If MHDA occurs despite using >1 TNFi and >1 non-TNFi, another non-TNFi (+/- MTX) can be used initially, instead of tofacitinib; alternatively, tofacitinib (+/- MTX) can be used instead of trying another TNFi.4 If MHDA occurs despite DMARDs, TNFi, or non-TNFi, short-term/low-dose steroids can be added.4
EULAR 2016
In phase I, MTX with upfront steroids is recommended, since steroids should be used when bridging/titrating csDMARD doses.8,10 Furthermore, MTX should be rapidly titrated to a goal of 25 mg/week (with folic acid 1 mg daily or folinic acid 2.5 mg daily supplementation), along with low/intermediate-dose steroids for up to 5 months (when MTX or another csDMARD should have reached maximal effectiveness).8,11 If there is a contraindication to MTX, patients can begin treatment with leflunomide or sulfasalazine (along with steroids).10
In phase II, for patients who do not respond to initial csDMARD therapy (owing to efficacy +/- toxicity issues) and have prognostically unfavorable factors present (e.g., RF, ACPA, high disease activity, early joint damage, or failure of >2 csDMARDs), a biological DMARD (e.g., TNFi or non-TNFi) or JAK inhibitor such as tofacitinib should be used.10 On the other hand, if patients do not have such factors present, the csDMARD can be switched to another csDMARD, or a combination of csDMARDs can be used.10
In phase III, for patients who do not respond to therapies from phase II, the initial biological DMARD should be switched to another biological DMARD (e.g., abatacept, IL-6 inhibitor, rituximab, or another TNF-alpha inhibitor) or a JAK inhibitor.10 The biological DMARD or JAK inhibitor should be combined with a csDMARD.10
Disease Flares
If disease flares occur while patients receive DMARDs, TNFi, and/or a non-TNFi, short-term steroids can be added, but at the lowest possible dose/duration.4
Remission
If patients are in remission, DMARDs, TNFis, non-TNFis, and/or tofacitinib can be tapered (but should not be discontinued altogether).4 On the other hand, if there is still low-level disease activity, DMARDs, TNFis, non-TNFis, and/or tofacitinib should be continued at the current doses.4
Vaccination Considerations
All therapies for RA suppress the immune system to some degree, which can be concerning for vaccination.4 There are four types of vaccines: (1) live-attenuated; (2) inactivated; (3) subunit, recombinant, polysaccharide, and conjugate; and (4) toxoid.12 The ACR guideline mentions that inactivated (specifically, pneumococcal, influenza, hepatitis B) and recombinant (specifically, human papilloma virus) vaccines are permissible for administration prior to or during all types of DMARD treatment (e.g., DMARD monotherapy, DMARD combinations, TNFi biological, or non-TNFi biological).4 Live-attenuated vaccines (specifically: herpes zoster) are permissible prior to initiating DMARD treatment, but are not recommended for administration while patients are on biological DMARDs.4 The ACR guideline does not explicitly mention recommendations for MMR, hepatitis A, polio, Haemophilus influenzae type B, meningococcal, diphtheria, tetanus, or pertussis vaccines.4
Limitations
Current guidelines do not address sarilumab (IL-6 inhibitor; approved in 2017) or baricitinib (JAK inhibitor; approved in 2018) yet.4,10,13,14 An update to the ACR guideline is pending.15
ROLE OF THE PHARMACIST
The majority of RA cases are diagnosed and treated on an outpatient basis. Pharmacists in ambulatory care settings (e.g., clinics, community pharmacies) have pivotal opportunities to counsel patients and ensure that RA drugs are administered appropriately. An example of such a drug is MTX, which should be taken once weekly, along with folic acid supplementation. During disease flares, patients may be prescribed steroids, which have myriad side effects, including hypertension, hyperglycemia, mood changes, and insomnia; pharmacists can confirm steroid dose and duration, as well as clarify whether a taper would be required. For biological DMARDs, pharmacists can reconfirm that patients are capable of self-administration, especially if they have severe RA and possible deformities in their hands or wrists.
CONCLUSION
RA is a chronic disease that requires interventions to modify disease progression. While initial presentations are related to joint inflammation, long-term sequelae can include extra-skeletal manifestations. The most recent RA guidelines are from ACR 2015 and EULAR 2016. There are specific differences between the guidelines, based on the respective region/population studied. A future update of the ACR guidelines may contain commentary regarding the roles of baricitinib and sarilumab, as well as other promising therapies for RA.
REFERENCES
1. U.S. National Library of Medicine. Rheumatoid arthritis.
https://medlineplus.gov/rheumatoidarthritis.html. Accessed January 13, 2019.
2. U.S. Department of Health & Human Services. Rheumatoid arthritis.
https://report.nih.gov/nihfactsheets/ViewFactSheet.aspx?csid=63. Accessed January 13, 2019.
3. CDC. Rheumatoid arthritis (RA). www.cdc.gov/arthritis/basics/rheumatoid-arthritis.html. Accessed January 13, 2019.
4. Singh JA, Saag KG, Bridges SL, et al. 2015 American College of Rheumatology guideline for the treatment of rheumatoid arthritis. Arthritis Rheumatol. 2016;68(1):1-26.
5. Arthritis Foundation. Arthritis by the numbers/book of trusted facts & figures. www.arthritis.org/Documents/Sections/About-Arthritis/arthritis-facts-stats-figures.pdf. Accessed January 13, 2019.
6. Minichiello E, Semerano L, Boissier MC. Time trends in the incidence, prevalence, and severity of rheumatoid arthritis: a systematic literature review. Joint Bone Spine. 2016;83(6):625-630.
7. Smolen JS, Aletaha D, McInnes IB. Rheumatoid arthritis. Lancet. 2016;388(10055):2023-2038.
8. Smolen JS, Aletaha D, Barton A, et al. Rheumatoid arthritis. Nat Rev Dis Primers. 2018;4:18001.
9. Aletaha D, Neogi T, Silman AJ, et al. 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010;62(9):2569-2581.
10. Smolen JS, Landewe R, Bijlsma J, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann Rheum Dis. 2017;76(6):960-977.
11. Van Ede AE, Laan RF, Rood MJ, et al. Effect of folic or folinic acid supplementation on the toxicity and efficacy of methotrexate in rheumatoid arthritis: a forty-eight week, multicenter, randomized, double-blind, placebo-controlled study. Arthritis Rheum. 2001;44(7):1515-1524.
12. U.S. Department of Health & Human Services. Vaccine types. www.vaccines.gov/basics/types/index.html. Accessed January 13, 2019.
13. Kevzara (sarilumab injection) prescribing information. Bridgewater, NJ: Sanofi-Aventis U.S. LLC; April 2018.
14. Olumiant (baricitinib tablet) prescribing information. Indianapolis, IN: Lilly USA, LLC; May 2018.
15. American College of Rheumatology. Updated guideline for the management of rheumatoid arthritis. Project plan—October 2018. Accessed January 13, 2019.
To comment on this article contact rdavidson@uspharmacist.com.








