US Pharm. 2016;41(11):26-29.
ABSTRACT: Diabetes is one of the most prevalent disease states affecting Americans today. Diabetes is a complex disease, and understanding each drug class and its role in managing diabetes is crucial to pharmacy practice. Sodium-glucose cotransporter 2 (SGLT2) inhibitors provide a novel avenue for hemoglobin A1C reduction. Having a comprehensive understanding of the SGLT2 inhibitors can help practitioners to determine when to use this medication class. SGLT2 inhibitors include canagliflozin, dapagliflozin, and empagliflozin. These medications offer a unique mechanism of action by blocking glucose reabsorption in the kidney. This medication class offers the advantage of once-daily dosing and a low risk of hypoglycemia. Potential disadvantages include genitourinary infections and elevations in low-density lipoprotein.
Diabetes is one of the most common disease states plaguing Americans. Over the past 35 years, the prevalence of diabetes has quadrupled in adults in the United States, with 21.9 million currently diagnosed. This number translates to roughly 9.3% of the U.S. population.1 In 2014 alone, 1.7 million Americans >20 years of age were diagnosed with type 2 diabetes. Type 2 diabetes accounts for nearly 95% of all diagnosed cases of diabetes.2 Diabetes is associated with numerous long-term microvascular and macrovascular complications. Microvascular complications include diabetic nephropathy, retinopathy, and neuropathy. Macrovascular complications include cardiovascular disease and stroke.3
With new medications regularly coming to the market for the treatment of type 2 diabetes, the question of medication selection revolves around hemoglobin A1C reduction potential, risk of cardiovascular adverse effects, and risk of hypoglycemia. The 2016 American Diabetes Association Standard of Medical Care in Diabetes recommends metformin as first-line monotherapy or in combination with adjunctive agents. Multiple second-line agents are available for optimizing treatment, including sulfonylureas, thiazolidinediones (TZDs), dipeptidyl peptidase IV (DPP-IV) inhibitors, glucagon-like peptide-1 (GLP-1) receptor agonists, insulin, and sodium-glucose cotransporter 2 (SGLT2) inhibitors.4
Pharmacology and Dosing
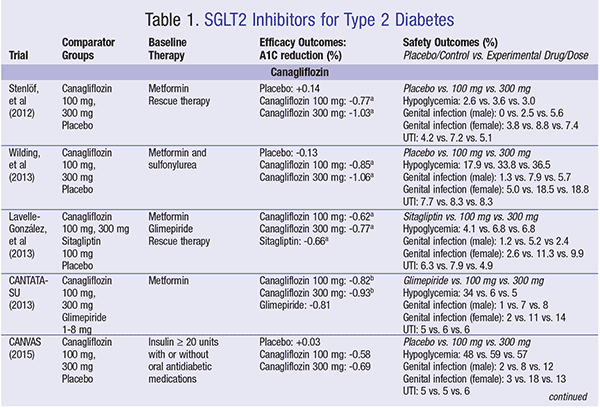
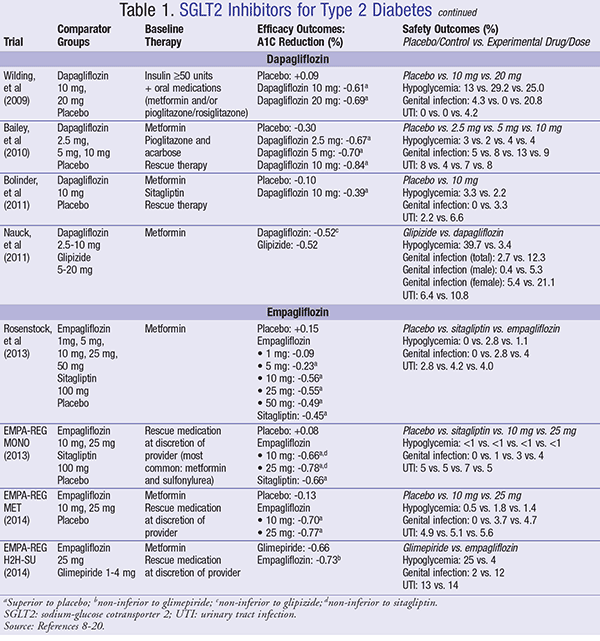
Three SGLT2 inhibitors are currently approved by the FDA for treatment of type 2 diabetes, including canagliflozin, dapagliflozin, and empagliflozin (Table 1). These agents exhibit a unique mechanism of action by blocking glucose reabsorption in the proximal tubule of the kidney. The glucose is then excreted via urination. These medications are administered orally once daily and can be given with or without food.5-7
Efficacy and Safety
Canagliflozin: Numerous studies have assessed the use of canagliflozin as add-on therapy to traditional diabetes regimens. It has also been compared with sulfonylurea and DPP-IV inhibitor therapy. Canagliflozin, in 100 mg and 300 mg doses, provides an A1C reduction ranging from 0.58% to 0.85% and 0.77% to 1.06%, respectively.8-12 When compared with glimepiride, canagliflozin 100 mg and 300 mg were non-inferior in reduction of A1C and achievement of the A1C goal. Canagliflozin also resulted in significantly less hypoglycemia.11 In each trial, fasting plasma glucose and postprandial glucose decreased with canagliflozin use. An additional benefit found with canagliflozin includes a reduction in body weight of 1.9 to 4.0 kg, with higher doses resulting in greater weight reduction. Systolic blood pressure (SBP) also decreased between 3.3 and 5.0 mm Hg. An adverse effect occurring frequently was genital mycotic infection. These infections occurred in 7.4% to 18.8% of women and 2.4% to 12% of men.8-12 In one study, a slight increase in urinary tract infections (UTIs) was found with canagliflozin.9 In general, hypoglycemia was rare; if it did occur, it was not severe.8,9,12
Dapagliflozin: Many studies have assessed the use of dapagliflozin as add-on therapy to traditional diabetes regimens. It has also been compared with sulfonylurea therapy. In four studies, dapagliflozin 10-20 mg provided an A1C reduction ranging from 0.39% to 0.84%.13-16 When compared with glipizide, dapagliflozin was non-inferior at A1C reduction and resulted in significantly less hypoglycemia.16 Dapagliflozin therapy was also assessed in patients taking insulin along with oral medications. The addition of dapagliflozin 10 mg and 20 mg resulted in an A1C decrease of 0.61% and 0.69%, respectively. In this trial, hypoglycemia occurred more frequently in the group receiving dapagliflozin; however, the concomitant use of insulin may have increased the patients’ risk for this adverse effect.16 In each trial, fasting plasma glucose and postprandial glucose decreased with dapagliflozin use. Additional benefits found with dapagliflozin included a reduction in body weight of 2.1-4.5 kg and SBP of 2.7-5.1 mm Hg, depending on dose and duration of use.13-16 In two studies, hypoglycemia was rare and often not severe with dapagliflozin.13,15 Genital infections were an adverse effect occurring more frequently in all dapagliflozin trials. The occurrence of these infections varied widely, from zero to 12.5%.13-16 One study showed these infections happened in 21.1% of women and 5.3% of men.16 In general, UTIs and hypoglycemia occurred infrequently.13-16
Empagliflozin: Multiple studies have assessed the use of empagliflozin as add-on therapy to traditional diabetes regimens. It has also been compared with sulfonylurea and DPP-IV inhibitor therapy. Empagliflozin 10 mg and 25 mg provide an A1C reduction ranging from 0.56% to 0.70% and 0.55% to 0.78%, respectively.17-20 When compared with glimepiride, empagliflozin 25 mg was non-inferior at A1C reduction after 52 weeks of administration. After 104 weeks, empagliflozin was superior to glimepiride. It also resulted in significantly less hypoglycemia.20 When compared with sitagliptin, empagliflozin 10 mg and 25 mg were non-inferior at A1C reduction. Hypoglycemia was rare in both treatment groups.19 In each trial, fasting plasma glucose and postprandial glucose decreased with empagliflozin use. Additional benefits found with empagliflozin included a reduction in body weight of 2.1-3.2 kg and SBP of 3.6-8.5 mm Hg, depending on dose and duration of use. Hypoglycemia was uncommon and often not severe with empagliflozin. An adverse effect occurring more frequently in all empagliflozin trials was genital mycotic infection. These infections occurred in 3% to 12% of patients. UTIs occurred at similar rates when compared with placebo and other antidiabetic medications.17-20
Advantages
The SGLT2 inhibitors offer many advantages.21 These medications provide effective treatment for patients in all stages of type 2 diabetes regardless of blood glucose level, insulin sensitivity, and insulin secretion.4 SGLT2 inhibitors work independently of insulin secretion, therefore lowering the risk of hypoglycemia.4 Studies comparing SGLT2 inhibitors with other second-line agents, including DPP-IV inhibitors, TZDs, and sulfonylureas showed a similar or reduced rate of hypoglycemia, with the added benefit of a modest decrease in weight when given in addition to metformin.22 Weight loss from these medications is likely secondary to increased calorie loss due to elevated glucose excretion.23 It is important to note that despite the osmotic and diuretic effects, a reduction in total fat mass as compared to placebo and other antidiabetic drugs has been observed.24
The leading cause of death associated with diabetes is atherosclerotic cardiovascular disease (ASCVD). Reduction in blood pressure is of paramount importance in assisting with ASCVD reduction.4 SGLT2 inhibitors were associated with a slight reduction in systolic and diastolic blood pressure as compared with placebo.8-14,16-20,25 Given that weight loss and blood-pressure lowering are essential to reduction of ASCVD, it can be postulated that the use of SGLT2 inhibitors may result in a reduction in cardiovascular events. In patients with ASCVD, SGLT2 inhibitor use has been associated with a reduced ASCVD event rate and mortality. The EMPA-REG OUTCOME study explored a primary outcome of death from cardiovascular causes, nonfatal myocardial infarction, or nonfatal stroke in patients assigned to receive empagliflozin 10 mg or 25 mg, or placebo. The study found no difference in the rate of myocardial infarction or stroke.24 Results indicated a lower rate of death from cardiovascular causes as compared with placebo (5.9% vs. 3.7%, respectively) when empagliflozin was added as adjunctive therapy to the standard of care.24 This finding is interesting because it is unclear why a reduction in cardiovascular death occurred. It has been proposed that the SGLT2 inhibitors may prolong survival in patients who experience a cardiovascular event rather than slowing atherosclerosis.26
A common complication of diabetes is the development of chronic kidney disease. The EMPA-REG OUTCOME study also found that changes in renal function occurred. Within the first four weeks of treatment, patients taking empagliflozin experienced a greater reduction in estimated glomerular filtration rate (eGFR). However, as the study progressed, the eGFR remained stable in the empagliflozin group, but decreased in the placebo group. These findings suggest that when used long-term, empagliflozin will slow the progression of renal impairment. The annual eGFR decrease was 0.19±0.11 mL/min/1.73 m2 with empagliflozin and 1.67±0.13 mL/min/1.73 m2 with placebo.27
Disadvantages
SGLT2 inhibitors come with some disadvantages.4-7 Increased glucosuria causes an increased incidence of genitourinary infections, occurring more frequently in women. A study comparing incidence of UTIs in patients on dapagliflozin versus placebo found an infection rate of 0.3% and 0.1%, respectively. Overall, the study noted these infections were mild to moderate and were managed clinically without discontinuing SGLT2-inhibitor therapy.28,29
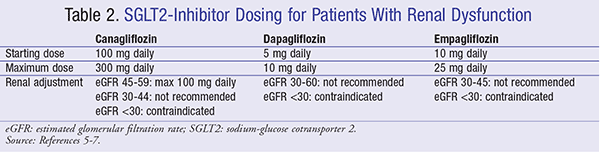
SGLT2 inhibitors can adversely affect lab values, including low-density lipoprotein (LDL) and serum creatinine (SCr). The mechanism by which SGLT2 inhibitors increase LDL is not fully understood, but research in hamsters indicates that this increase may be secondary to a shift from carbohydrate to lipid metabolism, resulting in reduction of LDL catabolism. There does appear to be a proportional increase in HDL; subsequent clinical significance has not yet been established.30 A transient increase in SCr upon SGLT2-inhibitor initiation is likely secondary to the mechanism of action. The therapeutic effect of SGLT2 inhibitors is reduced with a decline in renal function.31 Table 2 provides the dosing recommendations for patients with renal dysfunction. For patients started on SGLT2-inhibitor therapy, frequency of monitoring should be increased as eGFR drops below 60 mL/min/1.73 m2 and the medication should be discontinued if the eGFR falls below 30 mL/min/1.73 m2.5-7 A study of dapagliflozin versus placebo in moderate renal impairment (eGFR 30-60 mL/min/1.73 m2) showed a reduction in weight and blood pressure but no improvement in A1C.31
Since SGTL2 inhibitors can decrease blood pressure, hypotension may be an issue in some patients. Hypotension occurs more frequently in volume-depleted or elderly patients. The risk of volume depletion increases as the dose increases. Concomitant medications, age, and renal function should be taken into consideration when deciding to use these medications.5-7,29
In May 2015, the FDA released a drug-safety communication warning stating that use of SGLT2 inhibitors may result in diabetic ketoacidosis (DKA).32 As a result, modifications of all SGLT2 package inserts have been made. The American Association of Clinical Endocrinologists and the American College of Endocrinology also released a statement on SGLT2 inhibitors and DKA. Both entities agreed that the benefit of SGLT2 inhibitors outweighs the risk and found that DKA does not occur more frequently with these medications as compared with other antidiabetic drugs. The DKA cases reported thus far have occurred in patients with type 1 diabetes or in those patients requiring multiple insulin doses daily. The report details indicators of SGLT2-inhibitor-associated DKA, including insulin deficiency, surgery, extensive exercise, prolonged fasting, alcohol, or severe infections. All of these indicators are common in DKA, regardless of medication therapy prescribed.5-7,32
Finally, when compared with other second-line treatment options, SGLT2 inhibitors are relatively expensive (Table 3).33 Clinicians should evaluate patient access to medication, specifically regarding medication insurance coverage, prior to initiation of an SGLT2 inhibitor.
Pharmacist’s Role in Clinical Management
SGLT2 inhibitors are a reasonable addition to a patient’s diabetes regimen once metformin therapy has been optimized.4 SGLT2 inhibitors should be initiated at low doses and titrated upward to reach the A1C goal. Periodic monitoring of A1C and blood glucose is necessary to determine efficacy. Renal function and signs and symptoms of genital infections should be monitored for safety. Patients with renal dysfunction should not be started on an SGLT2 inhibitor. Because each medication has its own eGFR cutoff, package inserts should be reviewed to ensure the medication can be safely initiated. As with any diabetes medication, patients should be counseled on the importance of appropriate diet and exercise as adjunct therapy to medications.5-7
Conclusion
SGLT2 inhibitors provide a new avenue for reduction of A1C by increasing glucose excretion via the kidneys. While the type 2 diabetes epidemic continues to grow, these adjunct medications can reduce A1C regardless of disease progress. The A1C-lowering ability, low risk of hypoglycemia, and reduction in cardiovascular risk make these medications appealing to a wide range of patients.
REFERENCES
1. CDC. Diabetes Public Health Resource. Number (in Millions) of civilian, non-institutionalized adults with diagnosed diabetes, United States, 1980-2014. Updated December, 2015. www.cdc.gov/diabetes/statistics/prev/national/figadults.htm. Accessed May 26, 2016.
2. CDC. National Diabetes Statistics Report: Estimates of diabetes and its burden in the United States, 2014. Atlanta, GA: US Department of Health and Human Services; 2014. www.cdc.gov/diabetes/data/statistics/2014statisticsreport.html. Accessed June 8, 2016.
3. Fowler MJ. Microvascular and macrovascular complications of diabetes. Clin Diabetes. 2008;26(2):77-82.
4. American Diabetes Association. Standard of Medical Care in Diabetes-2016. 2016;39(Supplement 1).
5. Invokana [package insert]. Titusville, NJ: Janssen Pharmaceuticals, Inc; 2016.
6. Farxiga [package insert]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2016.
7. Jardiance [package insert]. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals, Inc; 2016.
8. Lavalle-González FJ, Januszewicz A, Davidson J, et al. Efficacy and safety of canagliflozin compared with placebo and sitagliptin in patients with type 2 diabetes on background metformin monotherapy: a randomised trial. Diabetologia. 2013;56(12):2582.
9. Stenlöf K, Cefalu WT, Kim KA, et al. Efficacy and safety of canagliflozin monotherapy in subjects with type 2 diabetes mellitus inadequately controlled with diet and exercise. Diabetes Obes Metab. 2013;15(4):372-382.
10. Wilding JP, Charpentier G, Hollander P, et al. Efficacy and safety of canagliflozin in patients with type 2 diabetes mellitus inadequately controlled with metformin and sulphonylurea: a randomised trial. Int J Clin Pract. 2013;67(12):1267-1282.
11. Cefalu WT, Leiter LA, Yoon KH, et al. Efficacy and safety of canagliflozin versus glimepiride in patients with type 2 diabetes inadequately controlled with metformin (CANTATA-SU): 52 week results from a randomised, double-blind, phase 3 non-inferiority trial. Lancet. 2013;382(9896):941-950.
12. Neal B, Perkovic V, Zeeuw D, et al. Efficacy and safety of canagliflozin, an inhibitor of sodium–glucose cotransporter 2, when used in conjunction with insulin therapy in patients with type 2 diabetes. Diabetes Care. 2015;38(3):403-411.
13. Bailey CJ, Gross JL, Pieters A, et al. Effect of dapagliflozin in patients with type 2 diabetes who have inadequate glycaemic control with metformin: a randomised, double-blind, placebo-controlled trial. Lancet. 2010;375(9733):2223-2233.
14. Wilding JP, Norwood P, T’joen C, et al. A study of dapagliflozin in patients with type 2 diabetes receiving high doses of insulin plus insulin sensitizers: applicability of a novel insulin-independent treatment. Diabetes Care. 2009;32(9):1656-1662.
15. Bolinder J, Ljunggren Ö, Kullberg J, et al. Effects of dapagliflozin on body weight, total fat mass, and regional adipose tissue distribution in patients with type 2 diabetes mellitus with inadequate glycemic control on metformin. J Clin Endocrinol Metab. 2011;97(3):1020-1031.
16. Nauck MA, Prato SD, Meier JJ, et al. Dapagliflozin versus glipizide as add-on therapy in patients with type 2 diabetes who have inadequate glycemic control with metformin. Diabetes Care. 2011;34(9):2015-2022.
17. Rosenstock J, Seman LJ, Jelaska A, et al. Efficacy and safety of empagliflozin, a sodium glucose cotransporter 2 (SGLT2) inhibitor, as add-on to metformin in type 2 diabetes with mild hyperglycaemia. Diabetes Obes Metab. 2013;15(12):1154-1160.
18. Häring HU, Merker L, Seewaldt-Becker E, et al. Empagliflozin as add-on to metformin in patients with type 2 diabetes: a 24-week, randomized, double-blind, placebo-controlled trial. Diabetes Care. 2014;37(6):1650-1659.
19. Roden M, Weng J, Eilbracht J, et al. Empagliflozin monotherapy with sitagliptin as an active comparator in patients with type 2 diabetes: a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Diabetes Endocrinol. 2013;1(3):208-219.
20. Ridderstråle M, Andersen KR, Zeller C, et al. Comparison of empagliflozin and glimepiride as add-on to metformin in patients with type 2 diabetes: a 104-week randomised, active-controlled, double-blind, phase 3 trial. Lancet Diabetes Endocrinol. 2014;2(9):691-700.
21. Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycemia in type 2 diabetes, 2015: a patient-centered approach: update to a position statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care. 2015;38(1):140-149.
22. Goring S, Hawkins N, Wygant G, et al. Dapagliflozin compared with other oral anti‐diabetes treatments when added to metformin monotherapy: a systematic review and network meta‐analysis. Diabetes Obes Metab. 2014;16(5):433-442.
23. Barnett AH. Impact of sodium glucose cotransporter 2 inhibitors on weight in patients with type 2 diabetes mellitus. Postgrad Med. 2013;125(5):92-100.
24. Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373(22):2117-2128.
25. Bolinder J, Ljunggren Ö, Kullberg J, et al. Effects of dapagliflozin on body weight, total fat mass, and regional adipose tissue distribution in patients with type 2 diabetes mellitus with inadequate glycemic control on metformin. J Clin Endocrinol Metab. 2011;97(3):1020-1031.
26. Abdul-Ghani M, Del Prato S, Chilton R, DeFronzo RA. SGLT2 inhibitors and cardiovascular risk: lessons learned from the EMPA-REG OUTCOME study. Diabetes Care. 2016;39(5):717-725.
27. Johnsson KM, Ptaszynska A, Schmitz B, et al. Urinary tract infections in patients with diabetes treated with dapagliflozin. J Diabetes Complications. 2013;27(5):473-478.
28. Scheen AJ. Pharmacodynamics, efficacy and safety of sodium–glucose co-transporter type 2 (SGLT2) inhibitors for the treatment of type 2 diabetes mellitus. Drugs. 2014;75(1):33-59.
29. Briand F, Mayoux E, Brousseau E, et al. Empagliflozin, via switching metabolism toward lipid utilization, moderately increases LDL cholesterol levels through reduced LDL catabolism. Diabetes. 2016;65(7):2032-2038.
30. Kohan DE, Fioretto P, Tang W, List JF. Long-term study of patients with type 2 diabetes and moderate renal impairment shows that dapagliflozin reduces weight and blood pressure but does not improve glycemic control. Kidney Int. 2014;85(4):962-971.
31. Handelsman Y, Henry RR, Bloomgarden ZT, et al. American Association of Clinical Endocrinologists and American College of Endocrinology position statement on the association of sglt-2 inhibitors and diabetic ketoacidosis. Endocr Pract. 2016;22(6):753-762.
32. Lopez JMS, Macomson B, Ektare V, et al. Evaluating drug cost per response with SGLT2 inhibitors in patients with type 2 diabetes mellitus. Am Health Drug Benefits. 2015;8(6):309-318.
33. Lexicomp Online, Hudson, Ohio: Lexi-Comp, Inc.; Accessed May 19, 2016.
To comment on this article, contact rdavidson@uspharmacist.com.