US Pharm. 2014;39(6)31-33.
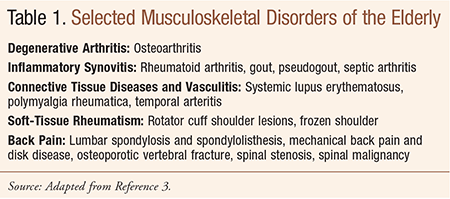
The prevalence of disabling joint diseases increases with age. Osteoarthritis (OA), considered a musculoskeletal disorder (Table 1), is the most prevalent articular disease in the elderly.1 Musculoskeletal disease is the most common cause of chronic disability in individuals older than 65 years, and arthritis, currently a growing medical problem, is the most common cause of disability in people over 75 years of age.2,3 Further, according to a North American survey, adults over age 65 years accounted for 37% of arthritis cases in 2005 and will account for 50% by 2030.4
Today, arthritis and foot pain are both major public health problems.5 The prevalence of foot pain among community-dwelling older adults ranges from approximately 36% to 70%, depending on definitions and populations studied.6 According to the Framingham Heart Study, prevalence data indicate that 29% of women and 19% of men reported foot pain on most days of the month.7
Shoes and other footwear may be an important modifiable factor in foot pain, particularly as it relates to rheumatic disorders; according to Riskowski et al, it is surprising that this topic has received little attention in the rheumatology community.5 Interestingly, Dufour et al examined factors associated with foot pain, including current and past shoe wear; their findings revealed that women who wore “poor” shoes (e.g. high heels, sandals, slippers) on a regular basis in their past were more likely to experience foot pain in their later years compared with those who wore “average” shoes.7
First Metatarsophalangeal Joint OA
OA of the first metatarsophalangeal joint (MPJ) of the big toe/great toe is a common condition (hallux limitus/rigidus) marked by pain, stiffness, and impaired ambulation.8 Furthermore, reduced flexibility and mobility in seniors can result in limitations affecting not only the movements and locomotion required for everyday functioning and responsibilities, but also leisure activities, vacations, and caregiving. Pharmacists need to consider that instability can arise secondary to pain, stiffness, and impaired ambulation when OA affects the MPJ; furthermore, falls occurring in the elderly need to be assessed using a multidimensional approach that considers such factors as medications, functional disability, and neurologic impairment.
OA Pathophysiology and Risk Factors
Differences between normally aging cartilage and osteoarthritic cartilage have been described, suggesting that OA is truly a disease and not just a natural consequence of aging.9 For example, the water content and the ratio of chondroitin sulfate to keratin sulfate constituents differ in normally aging and osteoarthritic cartilage. Furthermore, while found in both normally aging and osteoarthritic cartilage, denatured type II collagen is more prevalent in OA.9 Another important distinction is that elevated degradative enzyme activity is found in OA, but not in cartilage undergoing normal aging. The primary enzymes responsible for the degradation of cartilage are the matrix metalloproteinases (MMPs); together with pro-inflammatory cytokines (e.g., interleukin [IL]-1), MMPs appear to be important mediators of cartilage destruction.9 Following destruction, progressive cartilage degradation and OA ensue.
Age is the most consistently identified risk factor for OA, regardless of the joint being studied.10 Prevalence rates for both radiographic OA and, to a lesser extent, symptomatic OA rise steeply after age 40 years in women and age 50 years in men. OA is rarely found in persons under 35 years of age; in this population, secondary causes or other types of arthritis (Table 1) should be strongly considered.10 Additional risk factors for OA include:
• Gender, with women more likely to report pain in all affected joints
• Obesity, which is one of the most important modifiable risk factors and requires appropriate counseling
• Joint stress (e.g., occupation-related repetitive injury and physical trauma)
• Genetics
Clinical Manifestations
Pain is the predominant symptom in degenerative arthritis.11,12 Early in the course of OA, patients experience localized joint pain that worsens with activity, is relieved by rest, and is generally worse at the end of the day than it is at the beginning of the day; as the disease progresses, patients may also experience pain at rest. Stiffness, occurring in the morning or following a period of rest or inactivity is referred to as the gel phenomenon, but rarely lasts more than 30 minutes.10 Joint pain and tenderness are a result of changes in structure (e.g., bony enlargement) and function that produce decreased range of motion, muscle weakness, joint instability, and disability; in advanced disease, weight bearing joints may lock or give way.10,13 Other physical findings in OA joints include crepitus (an audible grinding produced by joint motion) and cool effusions (joint swelling without warmth secondary to intra-articular fluid).10 Absent in OA is the synovitis seen in inflammatory arthritis (e.g., rheumatoid arthritis) or the intense, hot inflammation seen in crystalline arthropathies (e.g., gout).10
Patients with OA of the first MPJ experience pain in the joint during activity, especially when pushing off on the toes while walking; they also experience stiffness in the great toe and find it difficult or impossible to dorsiflex or plantarflex the first MPJ.14 An enlargement forms and overlies the joint, and swelling develops around the joint. In MPJ OA, a change in the ability to ambulate with ease, coupled with stiffness and pain, accentuates the risk of falls in senior patients.
Diagnosis and Diagnostic Testing
Symptoms, physical findings, and radiographic changes constitute the diagnostic triad of OA. The American College of Rheumatology has set forth classification criteria to aid in the identification of patients with symptomatic OA that include, but do not rely solely on, radiographic findings.10
When cases are less straightforward, differential diagnoses should be considered, such as: 10,15
• Inflammatory arthritis if there is prolonged stiffness of greater than 1 hour (e.g., rheumatoid arthritis) or marked warmth and erythema in a joint (e.g., crystalline etiology)
• Systemic illness or other inflammatory condition if weight loss, fatigue, fever, and loss of appetite are noted or reported or when multiple joints are involved (e.g., polymyalgia rheumatica, rheumatoid arthritis, lupus, viral-related arthritis, enteropathic arthritis, sepsis, malignancy). A systemic cause is investigated with a rheumatic disease assessment (e.g., rheumatoid factor, erythrocyte sedimentation rate [ESR], antinuclear antibodies). Of note, the presence of monarticular heat, redness, and swelling indicates infection until proven otherwise, although gout is more likely.
• Periarticular structural derangement (e.g., bursitis, tendinitis, periostitis)
• MPJ pain can usually be differentiated from neuralgia or neuroma of the interdigital nerves when there is a lack of burning, numbness, tingling, and interspace pain (although these symptoms may result from joint inflammation); palpation may help with differentiation.
Treatment
Current treatment for OA is limited to control of symptoms (see
Resources
); analgesics, orthotics, and other modalities for MPJ OA, occasionally including surgery, are outlined in
Table 2
.
At this time, there are no pharmacologic agents capable of retarding the progression of OA or preventing OA. This is a fundamental and important area of current research. New therapies focused on reducing MMP activity and on stimulating matrix synthesis are in development.
Treatment goals are focused on relieving pain and stiffness; maintaining and improving joint mobility; preserving functional independence; educating the patient, family members, and caregivers, and maintaining quality of life.16-20
Emerging research suggests that there may be a significant role for foot orthotics and footwear in the treatment of the hip, knee, and foot; footwear is an important treatment strategy for reducing pain and improving function in populations with arthritis.5 Improved function better enables seniors to exercise (Table 2), and exercise (e.g., resistive strengthening) improves gait, strength, and overall function. Studies of community-dwelling and nursing home elderly clearly demonstrate that an important benefit of exercise is a reduction in the number of falls.1 According to a study by Riskowski et al, clinical trials and prospective studies will be required to unravel the links between foot pain, foot conditions, and interventions that lessen the impact of rheumatic diseases.5
Conclusion
Both arthritis and foot pain are major public health problems. Pharmacists can guide patients, healthcare providers, and caregivers toward modalities to relieve MPJ pain and stiffness for the maintenance and improvement of joint mobility, joint functionality, and quality of life.
REFERENCES
1. Bathon JM. Osteoarthritis: treatment. HopkinsArthritis.org. Updated December 2011. www.hopkinsarthritis.org/arthritis-info/osteoarthritis/oa-treatments. Accessed May 18, 2014.
2. Loeser RF, Jr. Delbono O. Aging of the muscles and joints. In: Halter JB, Ouslander JG, Tinetti ME, et al, eds.
Hazzard's Geriatric Medicine and Gerontology. 6th ed. New York, NY: McGraw-Hill; 2009:1355-1368.
3. Scott DL. Arthritis in the elderly. In: Fillit HM, Rockwood K, Woodhouse K, eds. Brocklehurst's Textbook of
Geriatric Medicine and Gerontology. 7th ed. Philadelphia, PA: Saunders Elsevier; 2010:566-576.
4. Hootman JM, Helmick CG. Projections of US prevalence of arthritis and associated activity limitations. Arthritis Rheumatol. 2006;54:226-229.
5. Riskowski J, Dufour AB, Hannan MT. Current musculoskeletal research on feet. Curr Opin Rheumatol. 2011;23:148-155.
6. Menz HB, Tiedemann A, Kwan MM, et al. Foot pain in community-dwelling older people: evaluation of the Manchester Foot Pain and Disability Index.
Rheumatol (Oxford). 2006:45:863-867.
7. Dufour AB, Broe KE, Nguyen US, et al. Foot pain: is current or past shoewear a factor?
Arthritis Rheum. 2009;61:1352-1358.
8. Menz HB, Levinger P, Tan JM, et al. Rocker-sole footwear versus prefabricated foot orthoses for the treatment of pain associated with first metatarsophalangeal joint osteoarthritis: study protocol for a randomised trial. BMC Musculoskelet Disord. 2014;15:86.
9. Ling SM, Bathon JM. Osteoarthritis: pathophysiology. HopkinsArthritis.org. www.hopkinsarthritis.org/arthritis-info/osteoarthritis/oa-pathophysiology.
Accessed May 18, 2014.
10. Manno R. Osteoarthritis: signs and symptoms. HopkinsArthritis.org. Updated: March 27, 2012. www.hopkinsarthritis.org/arthritis-info/osteoarthritis/signs-and-symptoms. Accessed May 17, 2014.
11. Heiberg T, Kvien TK. Preferences for improved health examined in 1,024 patients with rheumatoid arthritis: pain has highest priority. Arthritis Rheum. 2002;47:391-397.
12. Kidd BL. Osteoarthritis and joint pain. Pain. 2006;123:6-9.
13. Buys LM, Elliott ME. Osteoarthritis. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 8th ed. New York, NY: McGraw-Hill Inc; 2011:1599-1619.
14. Arthritis of the 1st Metatarso-phalangeal joint (hallux limitus/rigidus). www.podiatryaffiliates.com/referring-hallux.htm. Accessed May 18, 2014.
15. Metatarsophalangeal joint pain. MerckManuals.com. Reviewed December 2012, Modified May 2013. www.merckmanuals.com/professional/musculoskeletal_and_connective_tissue_disorders/foot_and_ankle_disorders/metatarsalgia.html. Accessed May 19, 2014.
16. Lane NE. Clinical practice. Osteoarthritis of the hip. N Engl J Med. 2007;57:1413-1421.
17. Felson DT. Clinical practice. Osteoarthritis of the knee. N Engl J Med. 2006;354:841-848.
18. Roddy E, Zhang W, Doherty M, et al. Evidence-based recommendations for the role of exercise in the management of osteoarthritis of the hip or knee–The MOVE consensus. Rheumatol (Oxford). 2005;44:67-73.
19. Hunter DJ, Felson DT. Osteoarthritis. BMJ. 2006;332:639-642.
20. Zhang W, Moskowitz RW, Nuki G, et al. OARSI recommendations for the management of hip and knee osteoarthritis, part II: OARSI evidence-based, expert consensus guidelines. Osteoarthritis Cartilage. 2008;16:137-162.
21. Clark MA, Finkel R, Rey JA. eds. Pharmacology. 5th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2012.
22. Semla TP, Beizer JL, Higbee MD. Geriatric Dosage Handbook. 19th ed. Hudson, OH: Lexicomp; 2014.
To comment on this article, contact rdavidson@uspharmacist.com.