US Pharm. 2015;40(9):HS5-HS9.
ABSTRACT: Cervical cancer is the third most common cancer diagnosed in women worldwide. Most cases have been linked to an HPV infection, particularly the HPV 16 and 18 types. Immunization against HPV has been developed in recent years and is thought to reduce the number of cases encountered. Routine screening procedures ranging from cytology to biopsy can help identify cervical cancer in its early stages. If identified, the cancer is staged and managed appropriately. Pharmacists can educate patients on the importance of vaccination and routine screening for prevention as well as counsel them on any therapeutic measures they may be undergoing.
Cervical cancer is a malignant tumor that originates in the cervix. It is the third most commonly diagnosed cancer worldwide and the fourth most common cancer in women, with higher rates being observed in less developed countries.1,2 In 2012, 528,000 new cases of cervical cancer were diagnosed worldwide, accounting for nearly 8% of all cancers (excluding nonmelanoma skin cancer).1 Even though rates in the United States have dropped since the introduction of the Pap test, approximately 12,000 women are diagnosed each year, with about 4,000 deaths annually.3 The median age of diagnosis is 45 years, and infection with high-risk human papilloma virus (HPV) genotype has been identified as the most important etiological risk factor for the development of cervical cancer.4,5
Etiology and Pathogenesis
A latent HPV infection has been identified as the leading cause of cervical cancer.6 HPV infection is the most common sexually transmitted disease, and more than 80% of the population are infected with HPV at some point in their lives.6 Cervical cancer has been shown to eventually develop in about 1% of infected individuals.5 Oncogenic HPV 16 and 18 account for about 65% of cervical carcinomas, while HPV 31, 33, 35, 45, 52, and 58 play a lesser role.5
There are three histologic types of cervical tumors as defined by the World Health Organization (WHO): squamous, glandular, and other epithelial tumors.6 Squamous cell carcinomas account for about 85% of all cases, while about 15% of cases are adenocarcinomas, including adenosquamous cancers. The number of cases of squamous cell carcinomas, however, is on the decline, while adenocarcinomas are on the rise in young women, possibly because screening methods in cytology are less effective in detecting adenocarcinoma.6
Since only a small proportion of HPV infections progress to cancer, other factors that may be involved in the process of carcinogenesis have been proposed. These include7:
- The type and duration of the viral infection
- Low host immunity such as poor nutritional status, immunocompromising conditions, and HIV infection
- Coexisting sexually transmitted diseases
- Family history
- High number of sexual partners
- Smoking
- Vitamin and nutritional deficiencies
- Lack of access to routine cytology screening.
Diagnosis
The diagnosis of cervical cancer includes a medical history, physical examination, pelvic examination, cervical cytology, HPV test, endocervical curettage, colposcopy, and biopsy.8 Physical symptoms such as abnormal vaginal bleeding, vaginal discomfort, malodorous discharge, and dysuria may be observed in some patients.
Cervical cytology, commonly referred to as a Pap smear, has been the mainstay of initial cervical cancer screening, but more recent molecular techniques used in the identification of HPV DNA are highly sensitive and specific.8 In some cases, both tests are performed, a process known as cotesting.
Since most women are asymptomatic in the early stages of the condition, regular screening and diagnosis are crucial. The American Cancer Society (ACS), the American Society for Colposcopy and Cervical Pathology (ASCCP), the American Society for Clinical Pathology (ASCP), the U.S. Preventive Services Task Force (USPSTF), and the American College of Obstetricians and Gynecologists (ACOG) have jointly developed guidelines for screening specific age groups. These are summarized in TABLE 1.9-13
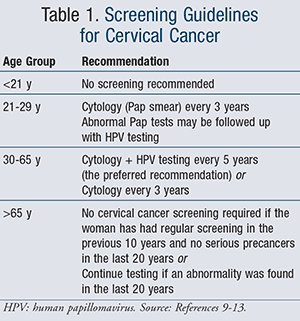
These recommendations apply to women who have had a hysterectomy without removal of the cervix as well as women who have been vaccinated against HPV.9-13 Those who have had a total hysterectomy should stop screening unless the hysterectomy was performed as part of cervical precancer treatment. Annual testing using any of the above methods is not recommended.
Prevention Through Immunization
Studies have shown that the use of HPV vaccines is effective at preventing HPV infection.14 Currently, there are three FDA-approved vaccines in the U.S. market: Gardasil, Gardasil 9 (both Merck & Co., Inc.), and Cervarix (GlaxoSmithKline).15-19
The Advisory Committee on Immunization Practices (ACIP) recommends that routine HPV vaccination be initiated at 11 or 12 years of age, and can be started as early as 9 years of age. HPV vaccination is also recommended for females aged 13 to 26 years and for males aged 13 to 21 years who have not been previously vaccinated.15
Gardasil is a quadrivalent vaccine that works against HPV types 6, 11, 16, and 18. It is FDA-approved for use in girls and women 9 to 26 years of age to prevent cervical cancer, genital warts, and anal cancer, as well as for males 9 to 26 years of age.16 The vaccine is administered as three injections of 0.5 mL each at 0, 2, and 6 months. The most common adverse effects observed with the use of Gardasil are headache, fever, nausea, dizziness, and injection-site pain, swelling, erythema, pruritus, and bruising. Its use is contraindicated in patients with hypersensitivity to yeast or previous doses of Gardasil. It is useful to note that the safety and efficacy of Gardasil have not been tested in pregnant women, children aged <9 years, and immunocompromised individuals.16
Gardasil 9, which was FDA-approved in December 2014, works against nine HPV types (6, 11, 16, 18, 31, 33, 45, 52, 58) and is indicated for females (aged 9-26 years) and males (aged 9-15 years).17 It protects against five additional HPV types that are not covered by other HPV vaccines. Like Gardasil, the vaccine is administered in three doses by IM injection at 0, 2, and 6 months. The most common local and systemic adverse reactions are headache and injection-site pain, swelling, and erythema.17
Cervarix is a bivalent vaccine that prevents cervical cancer caused by HPV types 16 and 18.18 It is FDA-approved for use in girls and women 9 to 25 years of age. It is administered as an IM injection as three doses (0.5 mL each) given at 0, 1, and 6 months. The most common adverse effects noted with its use include pain, redness, swelling at the site of injection, fatigue, headache, myalgia, gastrointestinal (GI) symptoms, and arthralgia. The safety of Cervarix has not been established in pregnant women. Immunocompromised individuals may have a reduced immune response to Cervarix. Its use is contraindicated in patients with severe allergic reactions to any component of the vaccine.19
Since vaccines can lead to syncope that may result in falling with injury, the recommendation is to observe the patient for 15 minutes after vaccination.17-19
Stage-Based Treatment
Various procedures are used for staging the cancer and guiding treatment, including colposcopy, biopsy, conization of the cervix, cystoscopy, proctosigmoidoscopy, CT and MRI imaging, and surgical staging.20
The treatment of cervical cancer varies with the stage of the disease, the patient’s age, and the size of the tumor as well as the patient’s wish to maintain fertility and/or sexual activity.20 The primary treatment of local disease consists of surgery, radiotherapy, or a combination of radiotherapy and chemotherapy. Both the European Society for Medical Oncology (ESMO) and the National Comprehensive Cancer Network (NCCN) have released guidelines for the management of the different stages of cancer. The stage-based application of these techniques is summarized in TABLE 2.5,20
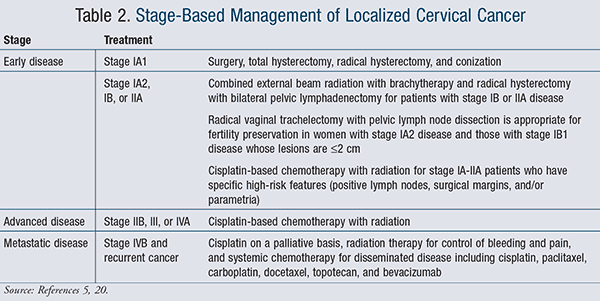
Stage I: Surgical procedures including laser surgery, cryosurgery, and the loop electrosurgical excision procedure (LEEP) are the preferred treatment for stage I cancers.20,21 If the cancer has spread to the uterus, a hysterectomy, in which the uterus and cervix are surgically removed, may be required. A trachelectomy leaves the uterus intact, whereas a radical hysterectomy involves the removal of the upper part of the vagina as well. The 5-year survival rates for appropriately managed stage 1A tumors exceed 95%.22
Stage II: Radiotherapy is generally initiated in stage II malignancies or in patients who have had a hysterectomy if other risk factors such as large tumor size, deep stromal invasion, or lymphovascular space invasion are present.21 Even though both surgery and radiotherapy are associated with infertility, radiation causes premature menopause, which can negatively affect the patient’s sex drive.
Stage IIB-IVA: Tumors that have progressed to stage IIB-IVA are managed with a combination of cisplatin chemotherapy and radiation. There are a number of options for cisplatin-based chemotherapy with radiation for the management of locally advanced cervical cancer, including20:
- Cisplatin: 40 mg/m2 IV once weekly (not to exceed 70 mg/wk); or
- Cisplatin + 5-fluorouracil (5-FU): 50-75 mg/m2 IV cisplatin on day 1 plus 5-FU 1,000 mg/m2 continuous IV infusion on days 2 to 5 and days 30 to 33 (total dose 4,000 mg/m2 each course); or
- Cisplatin + 5-FU: 50-75 mg/m2 IV cisplatin on day 1 plus 5-FU 1,000 mg/m2 continuous IV infusion over 24 hours on days 1 to 4 (total dose 4,000 mg/m2 each cycle) every 3 weeks for a total of three to four cycles.
Stage IVB: In metastatic disease, the NCCN recommends cisplatin-based chemotherapy as palliative therapy, radiation to manage bleeding and pain, and second-line agents, including docetaxel, gemcitabine, ifosfamide, 5-FU, mitomycin, irinotecan, and topotecan together with bevacizumab.20
Chemotherapeutic agents are associated with toxicities that may limit their use. Nausea and vomiting can be managed with concomitant use of antiemetics. Patients should also be monitored for bone marrow suppression, hypersensitivity reactions, allergic reactions, and neuropathies.20-22
Since cisplatin in particular results in cumulative nephrotoxicity, the patient’s serum creatinine, blood urea nitrogen, creatinine clearance, and electrolyte levels should be measured before initiating therapy and before each subsequent course. Adequate hydration throughout treatment will reduce the chances of renal toxicity and avoid treatment delays. Each dose of cisplatin should be administered 3 to 4 weeks apart. The use of greater dose frequencies may lead to irreversible neuropathies. Elderly patients are more susceptible to nephrotoxicity and neuropathy. Ototoxicity, also cumulative, can result from the use of cisplatin; therefore, patients should undergo audiometric testing before the initiation of therapy. Marked nausea and vomiting have been noted in patients treated with cisplatin. Other GI effects include diarrhea and/or anorexia. Myelosuppression is seen in 25% to 30% of patients being treated with cisplatin, with the elderly being more susceptible to this adverse effect. Other toxicities associated with the use of cisplatin include vascular toxicities, serum electrolyte disturbances, hyperuricemia, ocular toxicity, anaphylactic reactions, and hepatotoxicity.23
Bevacizumab: Bevacizumab (Avastin) was FDA-approved for the management of persistent, recurrent, or late-stage (metastatic) carcinoma of the cervix in August 2014.24,25 Bevacizumab is a recombinant humanized monoclonal immunoglobulin G1 (IgG1) antibody that binds to and inhibits the activity of human vascular endothelial growth factor (VEGF). The interaction of VEGF with its receptors leads to endothelial cell proliferation and new blood vessel formation.26 The use of bevacizumab plus chemotherapy showed a statistically significant improvement in overall survival and an increase in the rate of tumor shrinkage compared with chemotherapy alone. However, hypertension, thromboembolic events, and GI fistulas were higher in the bevacizumab group.24,27 The agent is approved for combination chemotherapy with paclitaxel and cisplatin or with paclitaxel and topotecan.24,25 Bevacizumab is administered as a dose of 15 mg/kg IV given every 3 weeks with paclitaxel/cisplatin or paclitaxel/topotecan. It is available as a 100 mg/4 mL or 400 mg/16 mL single-use vial.26
The most common adverse reactions associated with bevacizumab are epistaxis, headache, hypertension, rhinitis, proteinuria, taste alteration, dry skin, rectal hemorrhage, lacrimation disorder, back pain, and exfoliative dermatitis.26 GI perforation has been observed in 3.2% of treated patients. Bevacizumab should be discontinued in patients with GI perforation as well as wound dehiscence. Its use should be discontinued at least 28 days prior to elective surgery and should not be reinitiated for 28 days after surgery or until the surgical wound is fully healed. Since bevacizumab increases a patient’s probability of developing a severe or fatal hemorrhage, hemoptysis, GI bleeding, CNS hemorrhage, or vaginal bleeding, this solution should not be used in patients with serious hemorrhage or recent hemoptysis. Bevacizumab is also not recommended in breastfeeding mothers.26
Current Research
Early identification of and therapeutic intervention in cervical cancer have shown numerous beneficial effects. However, advanced-stage and metastatic disease is currently managed with chemotherapeutic agents that present with significant side effects and limited activity. Therefore, there is a need for more effective and less toxic agents. Targeted therapies that interfere with molecular targets expressed by the tumor are an area of significant investigation, with a focus on either epidermal growth factor receptor (EGFR) or VEGF. The only agent currently approved as a targeted therapy for cervical cancer is bevacizumab, as previously discussed.28
Role of the Pharmacist
Pharmacists can play a vital role in the prevention and management of cervical cancer by educating women on the importance of regular screening and vaccinations. Some pharmacists may also be involved in counseling patients on their cancer therapy, while others might be involved in reconstituting chemotherapeutic agents correctly in hospital pharmacies.
REFERENCES
1. World Cancer Research Fund International. Cervical cancer statistics. www.wcrf.org/int/cancer-facts-figures/data-specific-cancers/cervical-cancer-statistics. Accessed May 3, 2015.
2. Duenas-Gonzalez A, Serrano-Olvera A, Cetina L, et al. New molecular targets against cervical cancer. Int J Womens Health. 2014;6:1023-1031.
3. CDC. Cervical cancer statistics. www.cdc.gov/cancer/cervical/statistics. Accessed July 28, 2015.
4. Hoste G, Vossaert K, Poppe WA. The clinical role of HPV testing in primary and secondary cervical cancer screening. Obstet Gynecol Int. 2013;2013:610373.
5. Colombo N, Carinelli S, Colombo A, et al; ESMO Guidelines Working Group. Cervical cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012;23(suppl 7):vii28-vii32.
6. Tjalma WA, van Waes TR, van den Eeden LE, Bogers JJ. Role of human papillomavirus in the carcinogenesis of squamous cell carcinoma and adenocarcinoma of the cervix. Best Pract Res Clin Obstet Gynaecol. 2005;19(4):469-483.
7. Boardman C. Cervical cancer. Medscape. http://emedicine.medscape.com/article/253513-overview. Accessed May 13, 2015.
8. National Cancer Institute. Cervical cancer treatment for health professionals (PDQ). www.cancer.gov/types/cervical/hp/cervical-treatment-pdq. Accessed May 13, 2015.
9. American Cancer Society. The American Cancer Society guidelines for the prevention and early detection of cervical cancer. Revised December 11, 2014. www.cancer.org/cancer/cervicalcancer/moreinformation/cervicalcancerpreventionandearlydetection/cervical-cancer-prevention-and-early-detection-cervical-cancer-screening-guidelines. Accessed May 22, 2015.
10. Broder J. Pap tests less frequent under new guidelines. Medscape Medical News. October 25, 2012. www.medscape.com/viewarticle/773282. Accessed May 12, 2015.
11. American College of Obstetricians and Gynecologists. Screening for cervical cancer. Obstet Gynecol. 2012;120(5):1222-1238.
12. Saslow D, Solomon D, Lawson HW, et al. American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. CA Cancer J Clin. 2012;62(3):147-172.
13. U.S. Preventive Services Task Force. Screening for cervical cancer. AHRQ: Agency for Healthcare Research and Quality. www.uspreventiveservicestaskforce.org/uspstf/uspscerv.htm. Accessed May 5, 2015.
14. Muñoz N, Kjaer SK, Sigurdsson K, et al. Impact of human papillomavirus (HPV)6/11/16/18 vaccine on all HPV associated genital diseases in young women. J Natl Cancer Inst. 2010;102(5):325-339.
15. Petrosky E, Bocchini JA Jr, Hariri S, et al; CDC. Use of 9-valent human papillomavirus (HPV) vaccine: updated HPV vaccination recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2015;64(11):300-304. www.cdc.gov/mmwr/preview/mmwrhtml/mm6411a3.htm. Accessed May 19, 2015.
16. FDA. Gardasil. www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/UCM094042. Accessed April 28, 2015.
17. Gardasil 9 (human papillomavirus 9-valent vaccine, recombinant) prescribing information. Whitehouse Station, NJ: Merck & Co., Inc; February 2015. www.merck.com/product/usa/pi_circulars/g/gardasil_9/gardasil_9_pi.pdf. Accessed August 3, 2015.
18. CDC. FDA licensure of bivalent human papillomavirus vaccine (HPV2, Cervarix) for use in females and updated HPV vaccination recommendations from the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep. 2010;59(20):626-629.
19. Cervarix (human papillomavirus bivalent [types 16 and 18] vaccine, recombinant) prescribing information. Research Triangle Park, NC: GlaxoSmithKline; February 2015. https://www.gsksource.com/pharma/content/dam/GlaxoSmithKline/US/en/Prescribing_Information/Cervarix/pdf/CERVARIX-PI-PIL.PDF. Accessed May 2, 2015.
20. National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology: cervical cancer version 2.2015. www.nccn.org/professionals/physician_gls/PDF/cervical.pdf. Accessed May 6, 2015.
21. Carter JS. Cervical cancer guidelines. Medscape. http://emedicine.medscape.com/article/2267535. Accessed May 20, 2015.
22. Canavan T, Doshi N. Cervical cancer. Am Fam Physician. 2000;61(5): 1369-1376.
23. FDA. Platinol. http://www.accessdata.fda.gov/drugsatfda_docs/label/2011/018057s080lbl.pdf. Accessed August 3, 2015.
24. FDA approves Avastin to treat patients with aggressive and late-stage cervical cancer. FDA news release. August 14, 2014. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm410121.htm. Accessed May 15, 2015.
25. Lowes R. FDA OKs bevacizumab (Avastin) for late stage cervical cancer. Medscape Medical News. www.medscape.com/viewarticle/829985. Accessed May 21, 2015.
26. Avastin (bevacizumab) prescribing information. South San Francisco, CA: Genentech, Inc; May 2015. www.gene.com/download/pdf/avastin_prescribing.pdf. Accessed May 23, 2015.
27. Tewari KS, Sill MW, Long HJ III, et al. Improved survival with bevacizumab in advanced cervical cancer. N Engl J Med. 2014;370(8):734-743.
28. Tomao F, Di Tucci C, Imperiale L, et al. Cervical cancer: are there potential new targets? An update on preclinical and clinical results. Curr Drug Targets. 2014;15(12):1107-1120.
To comment on this article, contact rdavidson@uspharmacist.com.






