US Pharm. 2016;41(12):24-29.
ABSTRACT: Gastroesophageal reflux disease (GERD) is a diagnosis common in the community pharmacy setting. With pharmacists being one of the most accessible and trusted health care providers, patients seeking information at the pharmacy have confidence that they are being guided in the direction of achieving their treatment goals. Knowledge of the risk factors, presentation, complications, and treatment of GERD allows pharmacists to make individualized, appropriate recommendations to patients, whether those are lifestyle modifications, pharmacologic treatments, or referral to a patient’s primary care provider.
Gastroesophageal reflux disease (GERD) is the most common gastrointestinal (GI) diagnosis given by primary care providers during office visits, and it is arguably the most common diagnosis encountered in a community pharmacy setting.1 Because medications to treat GERD are both readily available OTC and frequently prescribed, knowledge of the pathophysiology, presentation, complications, and treatment options regarding this disease state is necessary when providing patients with information and making recommendations. This article will discuss GERD and its pharmacologic management with both OTC and prescription options.
INCIDENCE
GERD takes a significant toll on quality of life (QOL). Disruptive GERD, defined as GERD with symptoms occurring more than once weekly to on a daily basis, has been shown to decrease work productivity and increase time off from work. Patients with nocturnal GERD see an even bigger impact on QOL due to greater sleep disturbances.2
The estimated incidence of GERD in the United States is between 18% and 28%.3 Estimates vary based on the numerous ways GERD can be defined as well as on patients self-medicating without an appropriate diagnosis or self-reporting the disease based on symptoms. This incidence does not seem to increase with age. Symptoms actually decrease in severity after the age of 50 years, other than with erosive esophagitis, which has been linked to aging.4 Male and female prevalence of GERD is about equal, but females more commonly have nonerosive reflux disease (NERD), a subset of GERD, and males are more likely to have erosive esophagitis and develop Barrett’s esophagus and esophageal adenocarcinoma.3
Obesity is associated with an increase in GERD symptoms and complications.5 When comparing obese patients with those of a normal body mass index (BMI), a twofold increase in the development of GERD and erosive esophagitis and a 2.5-fold increase in the risk of esophageal adenocarcinoma have been observed.6
PATHOPHYSIOLOGY
Reflux occurs naturally in patients as a normal physiological occurrence. In typical conditions, reflux is diluted with saliva and cleared from the esophagus through peristaltic action.7 In addition, transient relaxation of the lower esophageal sphincter (LES), combined with normal swallowing, assists in protecting the esophagus from stomach acid reflux. In patients with GERD, transient relaxations of the LES occur more frequently than normal, allowing for an increase in episodes of reflux of large amounts of acidic GI fluids.7,8
The ability of the LES to sustain a tone higher than normal is a result of an increase in calcium mediated by excitatory cholinergic neurons. Higher levels of intracellular calcium are present in the resting LES compared with nonsphinchteric esophageal muscle.8 In addition to decreases in intracellular calcium levels and increases in transient LES relaxations, GERD is also noted to be due to deficiencies throughout the GI tract, including a delay in gastric emptying, ineffective clearance of reflux from the esophagus, and impaired mucosal defense mechanisms within the esophagus.
The mechanisms that allow incidents of reflux to become bothersome in patients with GERD are complex. As previously described, there are several defects thought to contribute to the pathophysiology of GERD. A consensus definition of differing levels of acidity was established in 2004 and defined reflux contents as either “acidic” (pH <4), “weakly acidic” (pH 4-7), or “weakly alkaline” (pH >7).9 Further studies evaluated the response of the conventional treatment of proton pump inhibitors (PPIs) on GERD-identified patients with both acidic and weakly acidic reflux contents that generated symptoms of heartburn. This evaluation further concluded that patients with weakly acidic reflux who were unresponsive to conventional treatments had esophageal damage from a gas that is present in this reflux, leading to distention of the proximal esophagus and dilation of the intercellular spaces, a known mechanism of esophagitis.10
RISK FACTORS & COMPLICATIONS
Risk factors for GERD include agents or physical states that decrease the pressure within the LES and increase transient relaxations. Some of the more commonly used medications known to do this include beta-adrenergic agonists, anticholinergics, nitrates, phosphodiesterase type 5 (PDE-5) inhibitors (e.g., sildenafil, tadalafil), theophylline, calcium channel blockers, and benzodi-azepines.8 While consumption of chocolate and carbonated beverages as well as tobacco use has been found to decrease LES pressure, cessation of these agents does not necessarily raise the pressure, decrease transient relaxations, or improve GERD symptoms.11 As discussed later in “Lifestyle Modifications,” patients have individual responses to caffeine, citrus, spicy foods, fatty foods, and alcohol, and general statements can be made about whether or not these cause GERD or put someone at a higher risk. Obesity has a strong correlation with the development of GERD. Although obesity has not been established as a cause of GERD, increasing BMI decreases LES pressure and increases reflux exposure because of more frequent sphincter relaxation.12
Prevalence of GERD and severity of symptoms increase as gestational age progresses, but symptom manifestation can occur at any time during pregnancy. In most cases, GERD in pregnant patients resolves after delivery in those who deny having symptoms before pregnancy.13 There has been controversy over whether eradication of Helicobacter pylori (H pylori) increases the risk of developing GERD. Evidence demonstrates that this is not the case in most patients, except for those with underlying peptic ulcer disease.14
Controlling GERD symptoms is crucial for patients, as this is a chronic disease. While symptoms are generally bothersome, left untreated they go beyond being a nuisance and can progress to cause serious complications. Esophageal strictures, Barrett’s esophagus, and esophageal adenocarcinoma are some of the more common complications. When left untreated, acid exposure in the esophagus leads to erosive esophagitis that can result in scarring, fibrosis, and ultimately strictures.15 Barrett’s esophagus can occur with refractory GERD due to histopathological changes in the lower esophageal epithelium. Because of the risk of developing into a malignant dysplasia, Barrett’s esophagus is a risk factor for esophageal adenocarcinoma.16
PRESENTATION & DIAGNOSIS
Although heartburn, regurgitation, difficulty swallowing, and dyspepsia tend to be the most common GERD-related complaints, GERD can display a range of various symptoms (TABLE 1).7 Recognizing symptoms that are common among patients with GERD will assist in differentiating diagnoses such as asthma, cough, laryngitis, and pneumonia that tend to also present with symptoms similar to those of GERD.

While there is no gold standard for the diagnosis of GERD, the diagnosis is generally made with a combination of symptom presentation, objective testing with endoscopy, ambulatory reflux monitoring, and response to pharmacologic management.5 Prior to utilization of invasive diagnostic tools, the patient’s symptoms are classified as either complicated or uncomplicated.7 Uncomplicated symptoms, including heartburn and/or regurgitation, are treated empirically with a single daily dose of a PPI (i.e., a PPI trial). For patients in whom no relief is obtained or for those with complicated symptoms, the dosage of the PPI is doubled. A response to therapy would ideally confirm the diagnosis; however, this method is met with limited specificity (54%) and sensitivity (78%).5 Current guidelines for the treatment of GERD recommend treatment without invasive diagnostic workup unless dysphagia, weight loss, anemia, or GI blood loss is present.5,7 Lack of response to a doubled dose of a PPI demands further objective evaluation.
An objective diagnostic tool with both an acceptable sensitivity and specificity currently remains nonexistent. Endoscopy, considered the primary tool to evaluate esophageal mucosa in patients with GERD symptoms, is positive in only about 40% of cases.8 Although endoscopy has great specificity when erosive esophagitis is present, patients with uncomplicated symptoms likely will not have erosions, therefore limiting the usefulness of endoscopy as an initial diagnostic tool.5 Esophageal biopsy, performed during an endoscopy, is currently not recommended as routine practice to diagnose GERD.5
Various other objective tools, such as barium radiographs, esophageal manometry, and ambulatory pH reflux monitoring are available but currently not recommended to be performed in the routine diagnosis of GERD.5,8
TREATMENT
In patients diagnosed with GERD, the goals of treatment include relief of symptoms, healing of erosive esophagitis, avoidance of recurrence, and prevention of complications. As with many diseases, lifestyle modifications (TABLE 2) are imperative in achieving treatment goals. In conjunction with lifestyle modifications, pharmacologic management includes agents aimed at controlling gastric acid secretion (TABLE 3), including antacids and acid-suppressive therapy with histamine2-receptor antagonists (H2RAs) and PPIs. Lastly, surgical interventions can be considered for refractory GERD with recurrent symptoms.
Lifestyle Modifications
Before making pharmacologic recommendations to patients asking for advice on treating their GERD symptoms, it is important to consider lifestyle modifications (TABLE 2).5,17,18 Patients may be under the impression that pharmacologic treatment is the only option for GERD, or some patients may be more interested in nonmedication options to help control symptoms. In any case, lifestyle modifications can improve symptoms considerably when combined with other options. Most commonly, it is recommended to avoid foods that lower the esophageal sphincter pressure and minimize behaviors that predispose one to increased esophageal acid exposure.
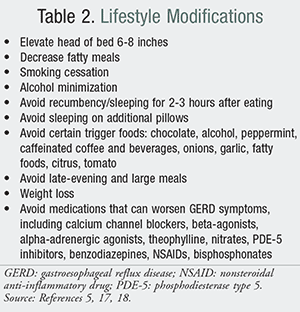
The strongest evidence for lifestyle modifications supports weight loss and head-of-bed elevation. Weight loss is associated with improvement of GERD symptoms, especially for patients who have a BMI >25 or have experienced recent weight gain.5,17 Patients may also find relief for nocturnal GERD symptoms with head-of-bed elevation using blocks or foam wedges and with avoidance of meals 2 to 3 hours before bedtime, especially foods with high fat content.5,18 Tobacco and alcohol use are often correlated with exacerbating GERD symptoms, but the evidence is not strong enough to recommend total elimination for symptom improvement. In addition, although recommended, there are no trials demonstrating that the avoidance of chocolate, citrus, mint, carbonated beverages, spicy foods, and caffeine improves GERD symptoms. Patients will vary in their responses to the cessation of some or all of these agents; therefore, recommendations can be personalized based on their reported triggers.5 Medical options for patients who fail lifestyle changes are described below.
Pharmacologic Options
Antacids: Antacids, such as Tums, Rolaids, and Maalox (aluminum hydroxide and magnesium hydroxide), are popular options for treating occasional mild episodes of reflux, but they should not be a first-line recommendation for the treatment of GERD.5 Patient education about the differences between occasional heartburn or indigestion and GERD is important so that patients do not try to self-treat and subsequently fail to achieve relief and/or succumb to medication side effects. Proper treatment of GERD should be preventive in nature rather than responsive; antacids only provide quick, short relief lasting about 30 to 60 minutes. While not a first-line recommendation, antacids can be recommended for patients with triggers known to aggravate their condition; for those with GERD symptoms occurring less than once a week; and for treatment of breakthrough symptoms not effectively treated by other medications. Despite providing symptomatic relief, antacids have not been shown to contribute to the healing of erosive esophagitis.7 If utilized, antacids should be taken after each meal and at bedtime.8
H2RAs: Currently, four H2RA agents are FDA-approved for the treatment of GERD and are also available OTC (TABLE 3).5,7,8 H2RAs are known to decrease gastric acid secretion in a reversible fashion by blocking the action of histamine on the H2 receptors of the gastric parietal cells.5 OTC formulations are available at a dose that is typically one-half of the lowest standard prescription dosage. H2RAs can be utilized as a maintenance option in patients who have symptoms of GERD but do not suffer from erosive esophagitis; in patients with milder forms of GERD; or in those who need on-demand therapy (e.g., H2RAs can be added at bedtime to PPI therapy).5,8
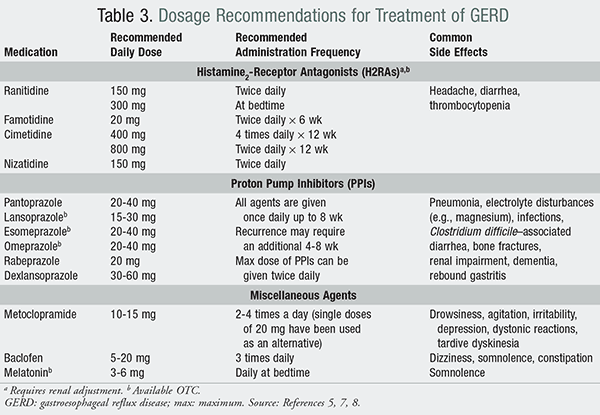
While H2RAs are more efficacious than antacids in the resolution of GERD symptoms, there are also disadvantages associated with long-term use of H2RAs. Some patients may develop tolerance with a decreased efficacy observed after 3 to 4 weeks of treatment. Other adverse effects noted with H2RAs include rare abnor-malities in CBC results, liver function test irregularities, and headache.5
PPIs: The most potent acid suppressive agents available are PPIs, and they tend to be the standard of care for patients with GERD. Irreversibly binding the gastric acid pump (i.e., the H+K+ATPase pump) of the parietal cells in the stomach (the final step in gastric secretion), PPIs have demonstrated in several trials their superiority over H2RAs for the treatment of reflux and erosive esophagitis.5,8 When administered, all PPIs should be taken once daily approximately 30 to 60 minutes prior to the first meal of the day to assure maximum efficacy; if possible, PPIs should be avoided at bedtime, as they are generally less effective.5 PPIs provide faster relief than antacids, H2RAs, or prokinetic agents with regard to symptom relief and long-term healing of erosive esophagitis.7 For those who do not find relief with once-daily administration of a PPI, adjustment of the timing of dose administration or twice-daily dosing is recommended. Nonresponders of maximal PPI therapy should be referred to their primary care physician for possible surgical evaluation.
Mechanisms of failure of PPIs include compliance, improper dosing timing, weakly acidic reflux, delayed gastric emptying, nocturnal reflux, residual acid reflux, esophageal hypersensitivity, and reduced PPI bioavail-ability.8 For most patients, PPI therapy is well tolerated, but adverse effects are associated with PPIs and include Clostridium-difficile–associated diarrhea, pneumonia, bone fractures, rebound hypersecretion, hypomagnesemia, vitamin B12 deficiency, and possible drug interactions.5,8
Miscellaneous Agents: Pharmacologic options beyond those discussed for patients with GERD with an incomplete response to PPI therapy are limited.5,8 Several alternative agents have been considered in patients with refractory GERD in which symptomatic reflux occurs despite optimal PPI therapy. Prokinetic therapy with metoclopramide, in conjunction with a PPI, has been shown to increase LES pressure, enhance esophageal peristalsis, and augment gastric emptying. Randomized controlled trials provide little evidence that prokinetic therapy improves symptoms in patients with reflux esophagitis; therefore, there is no clear role for metoclopramide in the primary treatment of GERD.5
Studies have also examined the role of gamma-aminobutyric acid type B (GABAB )–receptor agonists, such as baclofen, in patients with refractory GERD. Baclofen has shown to be effective in reducing transient LES relaxations, increasing LES tone, and decreasing reflux episodes. In addition, studies have demonstrated the effects of baclofen in reducing the number of postprandial and non–acid reflux events, nocturnal reflux activity, and belching episodes. Despite these demonstrations, there are no long-term data regarding the efficacy of baclofen in GERD, nor is it FDA-approved for the treatment of GERD.8
Although human trials for the use of melatonin for GERD are limited, results that have been published are significant in such symptoms as heartburn and epigastric pain and for improvements in LES function.7 Production of melatonin has been identified in the stomach, small intestine, and distal colon, with some evidence of production in the mouth and esophagus. Once produced, melatonin has been shown to have various mechanisms in the control of GERD symptoms, including stimulation of the production of nitric oxide and prostaglandin E2, inhibition of gastric acid secretion, reduction of inflammatory cytokines, and prevention of acid-pepsin–induced esophagitis.7 Although not conventional therapy, these agents provide more alternatives to explore in the event other pharmacologic agents do not demonstrate benefit for GERD symptom control.
Surgery
There are surgical options available for select patients who are seeking long-term treatment for their GERD, and for these eligible patients, surgical treatment is as effective as drug therapy. Surgical options include laparoscopic fundoplication (also referred to as laparoscopic antireflux surgery or LARS) and bariatric surgery. Not all patients meet the criteria that make them a good candidate for surgical GERD therapy, and identifying those patients who are eligible in the community setting can help appropriately refer them. Compliance issues, intolerable adverse effects secondary to GERD medications, hiatal hernia, refractory esophagitis, and refractory GERD symptoms all warrant referral.5
Laparoscopic fundoplication is a minimally invasive surgery that involves reducing the hiatal hernia and reinforcing the LES with reconstruction to reduce acid reflux. The best long-term outcomes to LARS are seen in patients who have responded to PPI therapy and whose main symptoms consist of heartburn and regurgitation.19 Patients nonresponsive to PPIs are generally not considered candidates for surgery, as it has been shown to be ineffective in this population, but a referral to their primary care provider can help determine other nonsurgical options to improve symptoms.20 As the incidence of obesity is on the rise, so is the incidence of bariatric surgery, another therapy shown to improve GERD symptoms. When comparing gastric bypass surgery to gastric banding in regard to improving reflux symptoms, gastric bypass is superior, with little evidence demonstrating that gastric banding made a difference in patients.21
CONCLUSION
GERD is a chronic disorder of the upper GI tract that is prevalent in many countries. Aside from the economic burden of the disease and its impact on patient QOL, it is the primary predisposing factor associated with the development of esophageal adenocarcinoma. Lifestyle modifications and acid-suppressive therapy through the use of antacids, H2RAs, or PPIs remain first-line treatment options for the management of GERD despite limited supporting data.
REFERENCES
1. Shaheen NJ, Hansen RA, Morgan DR, et al. The burden of gastrointestinal and liver diseases. Am J Gastroenterol. 2006;101:2128-2138.
2. Gerson LB, Fass R. A systematic review of the definitions, prevalence, and response to treatment of nocturnal gastroesophageal reflux disease. Clin Gastroenterol Hepatol. 2009;7:372-378.
3. El-Serag HB, Sweet S, Winchester CC, Dent J. Update on the epidemiology of gastro-oesophageal reflux disease: a systematic review. Gut. 2014;63(6):871-880.
4. Becher A, Dent J. Systematic review: aging and gastro-oesophageal reflux disease symptoms, oesophageal function and reflux oesophagitis. Aliment Pharmacol Ther. 2011;33:442-454.
5. Katz PO, Gerson LB, Vela MF. Guidelines for the diagnosis and management of gastroesophageal reflux disease. Am J Gastroenterol. 2013;108(3):308-328.
6. El-Serag HB. The association between obesity and GERD: a review of the epidemiological evidence. Dig Dis Sci. 2008;53(9):2307-2312.
7. Patrick L. Gastroesophageal reflux disease (GERD): a review of conventional and alternative treatments. Altern Med Rev. 2011;16(2):116-133.
8. Nwokediuko SC. Current trends in the management of gastroesophageal reflux disease: a review. ISRN Gastroenterol. 2012;2012:391631.
9. Sifrim D, Castell D, Dent J, Kahrilas PJ. Gastro-oesophageal reflux monitoring: review and consensus report on detection and definitions of acid, non-acid, and gas reflux. Gut. 2004;53:1024-1031.
10. Mainie I, Tutuian R, Shay S, et al. Acid and non-acid reflux in patients with persistent symptoms despite acid suppressive therapy: a multicenter study using combined ambulatory impedance-pH monitoring. Gut. 2006;55:1398-1402.
11. Tutuian R. Adverse effects of drugs on the esophagus. Best Pract Res Clin Gastroenterol. 2010;24(2):91-97.
12. Hampel H, Abraham NS, El-Serag HB. Meta-analysis: obesity and the risk for gastroesophageal reflux disease and its complications. Ann Intern Med. 2005;143:199-211.
13. Marrero JM, Goggin PM, de Caestecker JS, et al. Determinants of pregnancy heartburn. Br J Obstet Gynaecol. 1992;99:731-734.
14. Yaghoobi M, Farrokhyar F, Yuan Y, et al. Is there an increased risk of GERD after Helicobacter pylori eradication?: a meta-analysis. Am J Gastroenterol. 2010;105:1007-1013.
15. Scholten T. Long-term management of gastroesophageal reflux disease with pantoprazole. Ther Clin Risk Manag. 2007;3(2):231-243.
16. Taylor JB, Rubenstein JH. Meta-analyses of the effect of symptoms of gastroesophageal reflux on the risk of Barrett’s esophagus. Am J Gastroenterol. 2010;105:1729-1737.
17. Fraser-Moodie CA, Norton B, Gornall C, et al. Weight loss has an independent beneficial effect on symptoms of gastro-oesophageal reflux in patients who are overweight. Scand J Gastroenterol. 1999;34:337-340.
18. Pollmann H, Zillessen E, Pohl J, et al. Effect of elevated head position in bed in therapy of gastroesophageal reflux. Z Gastroenterol. 1996;34(suppl 2):93-99.
19. Oelschlager BK, Quiroga E, Parra JD, et al. Long-term outcomes after laparoscopic antireflux surgery. Am J Gastroenterol. 2008;103:280-287.
20. Swoger J, Ponsky J, Hicks DM, et al. Surgical fundoplication in laryngopharyngeal reflux unresponsive to aggressive acid suppression: a controlled study. Clin Gastroenterol Hepatol. 2006;4:433-441.
21. De Groot NL, Burgerhart JS, Van De Meeberg PC, et al. Systematic review: the effects of conservative and surgical treatment for obesity on gastro-oesophageal reflux disease. Aliment Pharmacol Ther. 2009;30:1091-1102.
22. Lacy BE, Weiser K, Chertoff J, et al. The diagnosis of gastroesophageal reflux disease. Am J Med. 2010;123(7):583-595.
23. Pilotto A, Franceschi M, Leandro G, et al. Clinical features of reflux esophagitis in older people: a study of 840 consecutive patients. J Am Geriatr Soc. 2006;54:1537-1542.
To comment on this article, contact rdavidson@uspharmacist.com.





