US Pharm. 2021;46(7):30-35.
ABSTRACT: Chronic obstructive pulmonary disease (COPD) is a complex disease that requires differential diagnosis and proper classification to identify appropriate therapy. Nonpharmacologic therapy and preventative measures should be utilized to prevent worsening of disease, symptoms, and complications. The Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines classify a patient’s COPD and provide recommendations for first-line treatment. The 2021 update included information regarding the use of e-cigarettes as nicotine replacement, triple therapy, and how the SARS-CoV-2 virus impacted patients with COPD. This article will focus on the diagnosis, classification, and treatment of COPD, as well as recently updated information regarding the use of e-cigarettes and the impact of COVID-19.
Chronic obstructive pulmonary disease (COPD) is a common, preventable, and treatable disease characterized by persistent respiratory symptoms and limitations to airflow due to abnormalities within the airway and/or alveolar. Common symptoms of COPD include chronic or progressive dyspnea, cough with sputum production, and recurrent lower respiratory tract infections. A patient may be at an increased risk for COPD due to exposure to noxious particles via cigarette smoking, occupational exposures, or air pollution, as well as due to genetics, socioeconomic status, airway hyperreactivity, and other factors.
Pharmacologic and nonpharmacologic therapy for the management of COPD is used to reduce the frequency and severity of symptoms and exacerbations, increase exercise tolerance, and improve overall quality of life. Smoking cessation is important in preventing progression of COPD and is an area where pharmacists can assist. A proper treatment regimen is determined based on severity of airflow limitation, symptom burden, and risk of exacerbation, as defined in the GOLD guidelines.
The SARS-CoV-2 virus has posed unique obstacles to the diagnosis and management of COPD. This may include difficulties differentiating between COVID-19 infection and COPD, increased use of telemedicine, inability to properly assess inhaler technique, infection risks of using certain inhaler types, and increased risk of severe complications. This review will summarize the updated 2021 GOLD guidelines for the diagnosis, management, and prevention of COPD.1
Diagnosis, Assessment, and Classification
COPD is diagnosed through a combination of spirometry and medical history, including symptom history and presence of risk factors. Spirometry is a noninvasive, readily available, objective measure of airflow limitation. Spirometry is required to make an official diagnosis of COPD with a postbronchodilator forced expiratory volume/forced vital capacity (FEV1/FVC) <0.70 confirming persistent airflow limitation. Key risk factors include smoking status and occupational or environmental exposures. Common symptoms of COPD include dyspnea, cough, sputum production, wheezing, and chest tightness. When assessing symptoms, it is important to consider differential diagnosis, especially during the COVID-19 pandemic, as noted by the updated Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines.
The 2021 GOLD guidelines recommend that the use of spirometry be limited to only patients who require urgent or essential testing for diagnosis and/or to assess lung function for interventional procedures or surgery, as such testing may lead to viral transmission. A safer alternative may be the use of home measurement of peak expiratory volume and the use of validated patient questionnaires. These questionnaires are completed via an interview to determine respiratory health, symptoms, comorbidities, and risk factors for developing COPD.2
Symptoms of COPD and COVID-19 infection may overlap. The two main overlapping symptoms are cough and shortness of breath. Some of the symptoms that favor COVID-19 include fever, hypoxia, loss of smell or taste, headaches, and lymphopenia. The SARS-CoV-2 virus may also cause pathophysiologic changes such as vascular injury, pneumonitis associated with hypoxemia, coagulopathy, high levels of systemic inflammation, and multiorgan involvement.
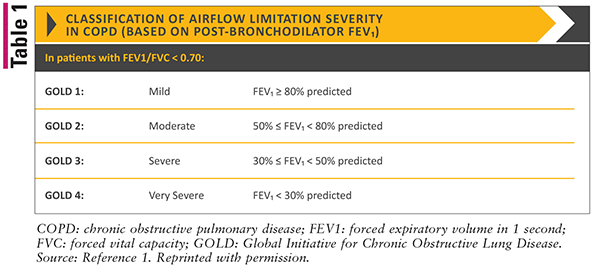
The GOLD guidelines classify patients into four different categories: GOLD 1 (mild), GOLD 2 (moderate), GOLD 3 (severe), or GOLD 4 (very severe) based on their level of airflow limitation. This is assessed by evaluating a postbronchodilator FEV1/FVC. Refer to TABLE 1 for more information regarding FEV1 values and GOLD classification.
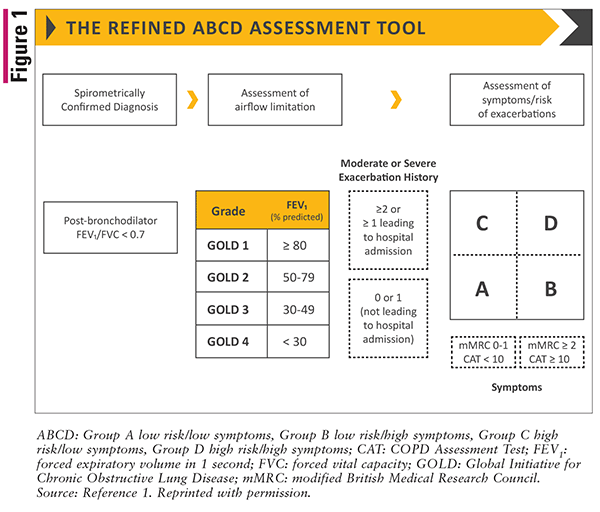
The Modified British Medical Research Council (mMRC) Questionnaire and COPD Assessment Test (CAT) are the two most widely used measures to assess symptoms of COPD. The mMRC is used to assess breathlessness of patients with COPD. It consists of grades 0 to 4, with grade 0 indicating breathlessness only with strenuous exercise and grade 4 indicating breathlessness that affects activities of daily living as well as the ability to leave the house. The CAT is an eight-item measure of impairment experienced from COPD, with scores ranging from 0 to 40. Higher scores on the CAT indicate a larger impact of COPD on the patient’s life.
Symptom burden and risk of exacerbation are also further classified into GOLD groups A through D, which is used to guide therapy. Classification is patient-specific, and each patient’s treatment regimen should be tailored specifically to their needs. Refer to FIGURE 1 for the GOLD group classification algorithm.
Nonpharmacologic and Preventative Therapy
Nonpharmacologic therapy for COPD includes smoking cessation, physical activity, and vaccination. Smoking cessation is important to prevent disease progression. Patients with COPD who are current smokers should be assessed for willingness to quit smoking and be provided with education on how to quit.
According to the 2021 GOLD guidelines, counseling by healthcare professionals, even for brief periods of time, significantly increases quit rates over self-initiated strategies. The recommended treatment for smoking cessation is nicotine-replacement therapy (NRT) with agents such as nicotine gum, inhalers, nasal spray, transdermal patches, sublingual tablets, or lozenges. Other pharmacologic agents such as varenicline, bupropion, and nortriptyline have been shown to increase long-term quit rates, but these should only be used as part of a supportive intervention program rather than monotherapy for smoking cessation.
The use of e-cigarettes for smoking cessation has been gaining popularity over recent years. These products provide a vaporized nicotine to be inhaled. While e-cigarettes have been available for over 15 years, the safety and efficacy data of e-cigarettes as a smoking cessation tool are still uncertain. In addition to nicotine, vape products are made with chemicals such as vegetable glycine, flavoring agents, volatile carbonyls, diacetyl, reactive oxygen species, furones, metals, and others. Severe acute lung injury, eosinophilic pneumonia, alveolar hemorrhage, respiratory bronchiolitis, other lung abnormalities, and even death have been linked to e-cigarette use.
One randomized, controlled trial by Bullen et al investigated the efficacy of e-cigarettes compared with nicotine patches in helping people to quit smoking.3 A total of 657 participants were randomized in a 4:4:1 ratio to 16-mg nicotine e-cigarettes, 21-mg once-daily nicotine patches, or placebo e-cigarettes. The primary outcome measure was biochemically verified continuous abstinence from smoking after 6 months. At this 6-month mark, 7.3% participants in the e-cigarette group had verified abstinence, 5.8% in the nicotine patch group, and 4.1% in the placebo group. However, there was insufficient statistical power to conclude the superiority of nicotine e-cigarettes over patches.
Another randomized, controlled trial by Hajek et al compared the efficacy of e-cigarettes to NRT of the patient’s choice.4 Randomization included 446 patients in the NRT group and 438 in the e-cigarette group. The primary outcome measure was sustained abstinence rates at 52 weeks. The NRT group had a 1-year quit rate of 9.9%, while the e-cigarette group had a rate of 18%. These results were deemed statistically significant with P <.001. This trial also found that participants in the e-cigarette group showed significantly better adherence and experienced fewer urges to smoke in the initial 4 weeks of quitting. Due to possible risks and a lack of further data, the 2021 GOLD guidelines do not make a recommendation for the use of e-cigarettes for smoking cessation.
Patients with COPD are also at higher risk for serious complications from certain vaccine-preventable illnesses.5 Pneumococcal vaccination, PPSV23, may be recommended for patients aged 19 to 64 years with COPD if they have significant comorbidities, including chronic lung or heart disease, to reduce the incidence of community-acquired pneumonia. One of the key changes to the 2021 update to the GOLD guidelines is the inclusion of the Tdap vaccination.
Kandeil et al conducted a systematic review across multiple countries, including North America, to investigate the potential value of a booster vaccination against pertussis in older adults.6 The review discussed that pertussis can lead to complications such as pneumonia as well as exacerbation in patients with COPD. The burden of pertussis is underestimated in older adults, and vaccination is therefore underutilized. A pertussis booster was found to be immunogenic, cost-effective, and well-tolerated. This review concluded that the pertussis vaccine can lower the disease burden and prevent transmission in more vulnerable populations.
One major role a pharmacist can take in supporting patients with COPD is providing education on self-management, medication adherence, and proper inhaler technique. A personalized written action plan that includes avoidance of aggravating factors, how to monitor or manage worsening symptoms, and breathlessness and energy-conservation techniques may be beneficial. Monitoring should include worsening of COPD symptoms, decline in spirometry results, smoking status, and number and frequency exacerbations. With an increased use of telemedicine and lack of face-to-face appointments during the COVID-19 pandemic, community pharmacists can have a larger impact on patient self-management and counseling.
Maintenance Treatment of COPD
GOLD classifications are used to determine initial treatment options for patients with COPD. FIGURE 2 shows the initial pharmacologic treatment for each GOLD group classification. Refer to TABLE 1 and FIGURE 2 for classifications. Importantly, all patients with COPD should be prescribed a rescue short-acting bronchodilator for immediate symptom relief.
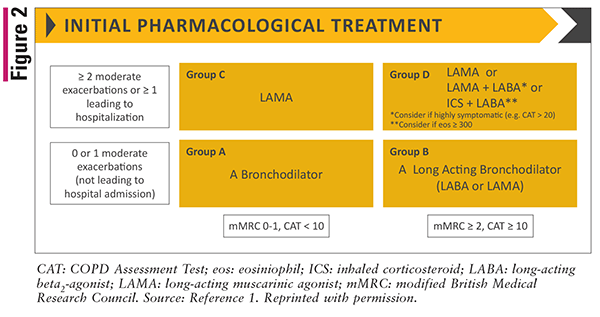
Patients in Group A are recommended to be treated with either a short- or long-acting bronchodilator to address breathlessness. Initial therapy for patients in Group B consists of a long-acting bronchodilator, either a long-acting beta2-agonist (LABA) or a long-acting muscarinic antagonist (LAMA). There is no evidence to support a preference between the two classes of inhaled long-acting bronchodilators, so the choice may be patient-specific.
For patients in Group C, a LAMA is recommended. LAMAs were found to be superior to LABAs in terms of prevention of exacerbations.7,8
Vogelmeier et al conducted a randomized, double-blind, double-dummy, parallel-group study over 1 year to investigate superiority of tiotropium (LAMA) compared with salmeterol (LABA) in preventing COPD exacerbations in patients with COPD aged 40 years or older with at least a 10 pack-year smoking history and a history of at least one exacerbation requiring systemic glucocorticoids or hospitalization in the previous 12 months.7 Enrollment included 7,376 patients randomly assigned to the tiotropium 18 µg once-daily intervention (3,707 patients) and the salmeterol 50 µg twice-daily intervention (3,669 patients). The primary endpoint was the time to first exacerbation of COPD. The results of the study show that tiotropium increased the time to first exacerbation (187 vs. 145 days) with a 17% reduction in risk of exacerbation (hazard ratio 0.83; confidence interval 0.77-0.90; P <.001). Tiotropium was also shown to reduce the annual number of moderate-to-severe exacerbations. The results of this study showed that in patients with COPD, tiotropium was more effective in preventing exacerbations.
Decramer et al also performed a randomized, blind, double-dummy, parallel-group study to compare the safety and efficacy of indacaterol (LABA) and tiotropium (LAMA) in patients aged 40 years or older with severe COPD and a history of at least one moderate-to-severe exacerbation in the previous year.8 Patients were randomized 1:1, stratified by baseline to either indacaterol 150 µg or tiotropium 18 µg once daily for 52 weeks. Enrollment included 3,444 patients, with 1,723 patients in the indacaterol arm and 1,721 patients in the tiotropium arm. The primary endpoint was whether indacaterol was noninferior to tiotropium in terms of trough FEV1 at Week 12. The secondary endpoint was the rate of exacerbations at Week 52. The results showed that indacaterol was noninferior to tiotropium in terms of FEV1 improvement; however, indacaterol did not show noninferiority in terms of annual exacerbation rates. This study suggests that tiotropium offers more protection by reducing annual exacerbations.
Patients in Group D may be treated with a LAMA due to its effects on both breathlessness and prevention of exacerbations. If a patient is highly symptomatic, with greater dyspnea and/or exercise limitation, they may be treated with a combination of a LAMA and a LABA. The preferred treatment option for patients in Group D that present with eosinophils ≥300 cells per microliter or have a history of asthma is an inhaled corticosteroid (ICS) plus a LABA. Another update to the guidelines states that the level of efficacy and benefit on lung function with the use of an ICS is lower in heavy or current smokers in comparison to light or ex-smokers; however, this is not a reason to avoid ICS use in these patients.
When it comes to choosing an inhaler formulation, one may want to take into consideration the current COVID-19 pandemic. The SARS-CoV-2 virus has been shown to be viable in aerosols for up to 3 hours. For this reason, aerosol therapy, such as the use of a nebulizer, should be avoided if possible until the pandemic resolves. The use of pressurized metered-dose ihalers, dry powder inhalers, and soft mist inhalers should be used for drug delivery as an alternative to avoid the spread of infection. If a nebulizer must be used, it should be done so in the absence of other people and in an area with increased air circulation.
In terms of managing patient follow-up, the guidelines recommend modifying therapy based on two treatable targets: dyspnea or exacerbations. If a patient presents with both dyspnea and exacerbations, the guidelines recommend following the exacerbation treatment pathway as shown in FIGURE 3.
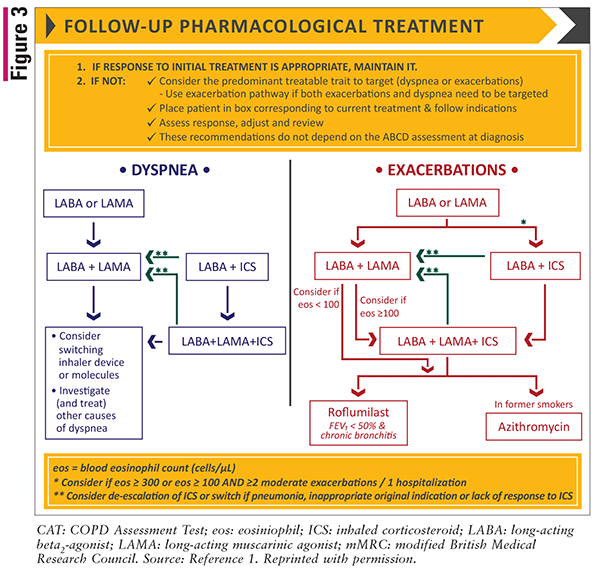
The 2021 update to the GOLD guidelines included new findings on mortality reduction with triple therapy compared with dual bronchodilation based on the IMPACT and ETHOS randomized, controlled trials. Both trials showed similar results, finding that triple therapy significantly lowers mortality compared with dual bronchodilation. However, there were no differences between triple therapy and LABA/ICS.
A randomized, controlled trial by Lipson et al assessed the once-daily use of triple therapy (ICS/LAMA/LABA) compared with dual therapy (ICS/LABA or LAMA/LABA) in terms of exacerbations and hospitalizations.9 A total of 10,355 patients with COPD were randomized into groups in which 4,151 received triple therapy with 100 µg fluticasone furoate, 62.5 µg umeclidinium, and 25 µg vilanterol; 4,134 participants received 100 µg fluticasone furoate/25 µg vilanterol; and 2,070 received 62.5 µg umeclidinium/25 µg vilanterol. The rates of moderate or severe exacerbations were 0.91 per year in the triple-therapy group, 1.07 per year in the fluticasone furoate/vilanterol group, and 1.21 per year in the umeclidinium/vilanterol group. The annual rates of hospitalization due to severe exacerbation were 0.13 with triple therapy and 0.19 in the umeclidinium/vilanterol group. These results were statistically significant with P <.001. The authors of this trial concluded that the use of triple therapy resulted in a lower rate in both moderate-severe COPD exacerbations and hospitalization.
COPD Exacerbations
Exacerbations may be triggered by several factors, most commonly infections of the respiratory tract. Conditions with nonspecific symptoms similar to a COPD exacerbation include pneumonia, pneumothorax, pleural effusion, pulmonary embolism, pulmonary edema, and/or cardiac arrhythmias. The goal of therapy when treating a COPD exacerbation is to minimize the negative impact of the current exacerbation and prevent future events. The recommended initial treatment for mild exacerbations is the use of short-acting beta2-agonists (SABA), with or without short-acting anticholinergics. In moderate-to-severe exacerbations, in addition to the SABA, systemic corticosteroids and antibiotics may also be used for no more than 5 to 7 days to improve FEV1, shorten recovery time, reduce the risk of early relapse, and shorten duration of hospitalization. The guidelines recommend continuing the use of long-acting bronchodilators throughout the exacerbation or to start these medications as soon as possible before discharge.
Infection with the SARS-CoV-2 virus may put a patient with COPD at increased risk of complications, including exacerbation and hospitalization. One systematic review and meta-analysis looked at the prevalence, severity, and mortality associated with COPD and smoking in patients with COVID-19. This analysis looked at 15 different studies and a total of 2,473 confirmed COVID-19 patients. While only 2% of the patients also had COPD, they concluded that patients with COPD, without differentiation of COPD severity, were at a higher risk of COVID-19 severity (63%) and mortality compared with patients without (33.4%).10 While patients with COPD may not be at an increased risk of infection, they may be at an increased risk of complications and hospitalization due to infection.
If a patient with COPD contracts the SARS-CoV-2 virus, there is no evidence that COPD medications should be altered, and treatment should be the same standard of care that other COVID-19 patients receive. As this is a respiratory virus, it may trigger and aggravate COPD exacerbation. Post COVID-19 infection, all patients with COPD should undergo rehabilitation and follow-up.
Conclusions
First-line management of COPD is dependent upon patient-specific factors such as GOLD classification and patient preference; however, nonpharmacologic and preventative methods should always be considered. Preventative methods include vaccination, including the new recommendation for a Tdap booster in patients with COPD. All patients with COPD should be prescribed a short-acting bronchodilator for immediate relief. New findings regarding triple therapy suggest that there is a significant decrease in mortality in comparison to dual bronchodilation therapy and a decrease in exacerbations and hospitalization. The management of patients at follow-up is dependent on two treatable targets: dyspnea and exacerbations.
Pharmacists can assist patients with smoking cessation and other recommendations, and tailoring therapy to patient-specific needs. A recommendation for the use of e-cigarettes over typical NRT cannot be made at this time. While studies point to conclude that its use may be effective, there is lack of data proving its safety and true efficacy. The update to the guidelines also states that the level of efficacy and benefit on lung function with the use of an ICS is lower in heavy or current smokers in comparison with light or ex-smokers.
While patients with COPD may not be at an increased risk of becoming infected with COVID-19, they are at risk for greater severity, complications, and mortality associated with infection. If a patient with COPD does become infected with COVID-19, it is recommended that they are treated with the same standard of care as other patients but may be monitored more closely with frequent rehabilitation and follow-up visits. The overall treatment for COPD should be individualized to the patient based on their specific needs and preferences.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
REFERENCES
1. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. 2021 report.
2. Jithoo A, Enright P, Burney P, et al. Case-finding options for COPD: results from the Burden of Obstructive Lung Disease study. Eur Respir. 2013;41(3):548-555.
3. Bullen C, Howe C, Laugesen M, et al. Electronic cigarettes for smoking cessation: a randomised controlled trial. Lancet. 2013;382:1629-1637.
4. Hajek P, Phillips-Walter A, Przulij D, et al. E-cigarettes compared with nicotine replacement therapy within the UK Stop Smoking Services: the TEC RCT. Health Technol Assess. 2019;23:1-82.
5. CDC. Lung disease including asthma and adult vaccination. Updated February 23, 2021. Accessed June 23, 2021.
6. Kandeil W, Atanasov P, Avramioti D, et al. The burden of pertussis in older adults: what is the role of vaccination? A systematic literature review. Expert Rev Vaccines. 2019;18:439-455.
7. Vogelmeier C, Hederer B, Glaab T, et al. Tiotropium versus salmeterol for the prevention of exacerbations of COPD. N Engl J Med. 2011;364:1093-1103.
8. Decramer ML, Chapman KR, Dahl R, et al. Once-daily indacaterol versus tiotropium for patients with severe chronic obstructive pulmonary disease (INVIGORATE): a randomised, blinded, parallel-group study. Lancet Respir Med. 2013;1:524-533.
9. Lipson DA, Barnhart F, Brealey N, et. al. Once-daily single-inhaler triple versus dual therapy in patients with COPD. N Engl J Med. 2018;378:1671-1680.
10. Alqahtani J, Oyelade T, Aldhahir AM, et al. Prevalence, severity and mortality associated with COPD and smoking in patients with COVID-19: a rapid systematic review and meta-analysis. PLoS One. 2020;15:e0233147.





