US Pharm. 2014;39(4):69-74.
ABSTRACT: Although for decades warfarin has been the gold standard for prevention of thromboembolic events, management is complicated by the drug’s relatively narrow therapeutic window and the high incidence of interactions. Target-specific oral anticoagulants (TSOAs) are an attractive alternative to warfarin because they do not require routine monitoring and are associated with a low number of clinically relevant drug interactions. TSOAs include dabigatran, apixaban, and rivaroxaban. Pharmacology, dosing, clinical efficacy, and special considerations for these TSOAs are reviewed.
For decades, warfarin (Coumadin) has been the gold standard for long-term anticoagulation in patients at high risk for thromboembolic events (TEEs). The two most common indications for warfarin are prevention of embolic or ischemic stroke in patients with atrial fibrillation (AF) and primary treatment and secondary prevention of TEEs, including deep venous thrombosis (DVT) and pulmonary embolism (PE). Without proper anticoagulation, the risk of TEEs in these patients is high: Those with AF and a CHADS2 (Congestive heart failure; Hypertension; Age ≥75 years; Diabetes; prior Stroke, transient ischemic attack, or thromboembolism) score ≥2 have an estimated risk of 4.5% to 9.6% per year, and those with a history of unprovoked DVT have a 10% risk of recurrence at 1 year and a 30% risk of recurrence at 5 years.1,2 These risks are significant, since a TEE can result in death or permanent disability. Multiple trials have demonstrated that, compared with placebo, warfarin can reduce the risk of death by 25% and that of nonfatal stroke by up to 67% in AF patients.1 In addition, extended therapy (>3 months) has been shown to reduce the risk of recurrent TEEs by 82% in patients with a history of unprovoked DVT or PE.2
Despite overwhelming evidence demonstrating the benefits of warfarin therapy, management is complicated by drug-drug, drug-food, and drug-disease interactions, all of which necessitate frequent international normalized ratio (INR) monitoring, dosage adjustments, and patient education. Even when therapy is managed by a specialized anticoagulation clinic with a high level of provider and patient vigilance, time in therapeutic range (TTR) averages only 63%. Furthermore, TTR is reduced by 11% to 12% when therapy is monitored by the community practitioner,3 and at least one study examining patients with AF found that the risk of bleeding and stroke increases significantly when TTR drops below 54%.4 The challenges associated with maintaining warfarin within its relatively narrow therapeutic range render target-specific oral anticoagulants (TSOAs) an attractive option because these agents do not require routine monitoring and are associated with a low number of clinically relevant drug interactions.
This article will review the pharmacology, dosing, and precautions for the factor Xa (FXa) inhibitors apixaban and rivaroxaban and the direct thrombin (factor IIa, FIIa) inhibitor dabigatran. Additionally, the clinical efficacy and safety of these agents used in nonvalvular AF or for the treatment of DVT or PE will be summarized.
Pharmacology, Dosing, and Interactions
Apixaban: Apixaban (Eliquis) binds selectively and reversibly to FXa.5 This agent currently is FDA-approved only for TEE prophylaxis in patients with nonvalvular AF (NVAF), but there is mounting evidence of the drug’s efficacy and safety for the treatment and prophylaxis of recurrent DVT or PE. The most severe adverse effects (AEs) associated with apixaban use involve bleeding; accordingly, patients should be counseled regarding signs and symptoms of severe bleeding and about medications with antiplatelet activity (e.g., nonsteroidal anti-inflammatory drugs [NSAIDs]). In rare cases, hypersensitivity and anaphylactic-type reactions have occurred (<1%). Apixaban may be taken with or without food, and the recommended dosage for most patients is 5 mg orally twice daily (see TABLE 1 for dosage adjustments). Apixaban has a prolonged absorption period and, as a result, has a half-life of approximately 12 hours with repeat dosing despite the shorter elimination half-life. This allows for twice-daily dosing, but also means that the anticoagulant effect lasts for approximately 24 hours when the agent is stopped.

Apixaban is partially metabolized by the liver and then undergoes renal and biliary elimination.5 Major inhibitors of CYP3A4 and the efflux transporter P-glycoprotein (Pgp) (e.g., ketoconazole, itraconazole, clarithromycin, ritonavir) can reduce apixaban elimination, thereby increasing the risk of bleeding. Dosage adjustments for apixaban in combination with one of these agents have been recommended (TABLE 1). Conversely, major inducers of these enzymes (e.g., rifampin, carbamazepine, phenytoin, St. John’s wort) can reduce exposure to apixaban and increase the risk of TEEs; therefore, concomitant use should be avoided. Owing to its inhibition of FXa, apixaban interferes with laboratory tests such as prothrombin time (PT), INR, and activated partial thromboplastin time (aPTT); the magnitude of this effect is highly variable between patients, so these tests cannot be used to monitor apixaban’s efficacy.
Rivaroxaban: Rivaroxaban (Xarelto) is approved not only for NVAF and for treatment and long-term prevention of recurrent DVT or PE, but also for prophylaxis of TEEs associated with hip- or knee-replacement surgery.6 Like apixaban, rivaroxaban selectively inhibits the active site on FXa. The most severe AEs, as expected, are related to bleeding. Other AEs reported in clinical trials include abdominal discomfort, musculoskeletal pain, and pruritus. Food enhances the absorption of rivaroxaban 15 mg or 20 mg, and patients should be counseled to take each dose consistently at the same time every day with a meal; the tablet may be crushed and taken with applesauce or via a feeding tube. Because the oral bioavailability of the 10-mg dose is nearly 100%, food does not affect absorption. Dosing depends upon the indication (TABLE 2), and there are several recommended adjustments.
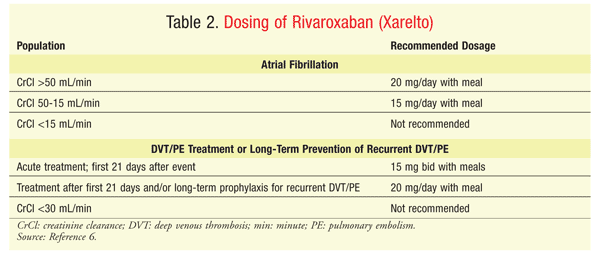
Approximately 50% of the rivaroxaban dose is metabolized by the liver, with metabolites eliminated via urine and feces.6 Nearly one-third of the dose is removed unchanged in the urine via Pgp. It is not recommended to use rivaroxaban with strong combined inhibitors of CYP3A4 and Pgp (e.g., ketoconazole, itraconazole, ritonavir) or strong combined inducers of these enzymes (e.g., rifamycin, carbamazepine, phenytoin, St. John’s wort). Coadministration with short courses of erythromycin or clarithromycin was not found to increase the risk of bleeding and therefore is not contraindicated.6
Dabigatran: Dabigatran (Pradaxa) is FDA-approved for the prevention of TEE in patients with NVAF.7 Dabigatran and its active metabolites bind directly to thrombin, thereby preventing conversion of fibrinogen to fibrin during the coagulation cascade. Like the other TSOAs, dabigatran affects PT, aPTT, and INR, but the effects are unpredictable and small in magnitude; therefore, these tests are not reliable for monitoring the drug’s efficacy. AEs that led to discontinuation of dabigatran in clinical trials include bleeding, gastrointestinal (GI) hemorrhage, gastritis-like symptoms, dyspepsia, nausea, and diarrhea. The recommended dosage in patients with NVAF is 150 mg twice daily, but adjustments are recommended in patients with renal impairment and in those taking certain interacting medications (TABLE 3).
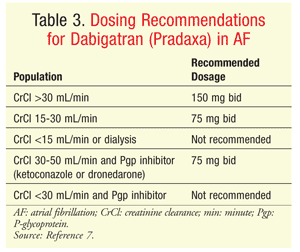
Dabigatran is converted to four active glucuronide metabolites, and the drug and metabolites are nearly 80% renally cleared; significant renal impairment requires dosage adjustment.7 Strong inducers of Pgp reduce dabigatran concentrations by up to 70%, so concomitant use should be avoided. Pgp inhibitors have variable effects on dabigatran concentration and elimination; ketoconazole and dronedarone require dosage reductions of dabigatran, but amiodarone, verapamil, quinidine, and clarithromycin do not alter the pharmacokinetics of dabigatran in a manner expected to be clinically significant.
Efficacy and Safety
AF: The 2012 guidelines developed by the American College of Chest Physicians (ACCP) recommend that providers assess the risk of stroke or TEE in AF patients on an individualized basis to determine whether antiplatelet therapy or anticoagulant therapy is more appropriate.1 For patients with a CHADS2 score ≥1, anticoagulation is recommended over antiplatelet therapy; dabigatran is recommended over dose-adjusted warfarin for patients without valvular heart disease (e.g., mitral stenosis, mechanical heart valve) who are candidates for anticoagulation therapy.1 These guidelines do not specifically address apixaban or rivaroxaban for TEE prevention in patients with NVAF because at that time they had been inadequately studied in this population. All three agents now carry FDA approval for this indication.
Each agent has been compared with warfarin in a major double-blind, controlled trial (TABLE 4). Rivaroxaban was noninferior to warfarin, and apixaban and dabigatran were superior to warfarin, for prevention of stroke and TEEs in patients with NVAF.8-10 Compared with warfarin, apixaban significantly reduces the risk of all-cause mortality. Apixaban has a lower risk of both major bleeding and intracranial bleeding compared with warfarin, and it does not increase the risk of GI bleeding. The risk of major bleeding with dabigatran and rivaroxaban appears to be similar to that for warfarin, but both agents have a slightly higher risk of GI bleeding events. Despite this, dabigatran and rivaroxaban have a lower risk of intracranial bleeding compared with warfarin, and rivaroxaban has a lower risk of fatal bleeds.

DVT/PE Treatment and Long-Term Prophylaxis for Recurrent DVT/PE: The ACCP recommends ≥3 months of anticoagulation therapy for patients who experience an acute DVT or PE.2 For patients with a coagulopathy (e.g., protein C deficiency) or recurrent TEEs, or for those with an unprovoked TEE who have a low-to-moderate risk of bleeding, the guidelines advise indefinite (lifetime) prophylactic therapy with anticoagulants. Low-molecular-weight heparin (LMWH) and warfarin are weakly favored over agents such as apixaban, rivaroxaban, and dabigatran owing to the lack of postmarketing safety data available at that time. TABLE 5 summarizes the clinical trials of TSOAs for treatment of acute venous thromboembolism (VTE) or prophylaxis for recurrent VTE.
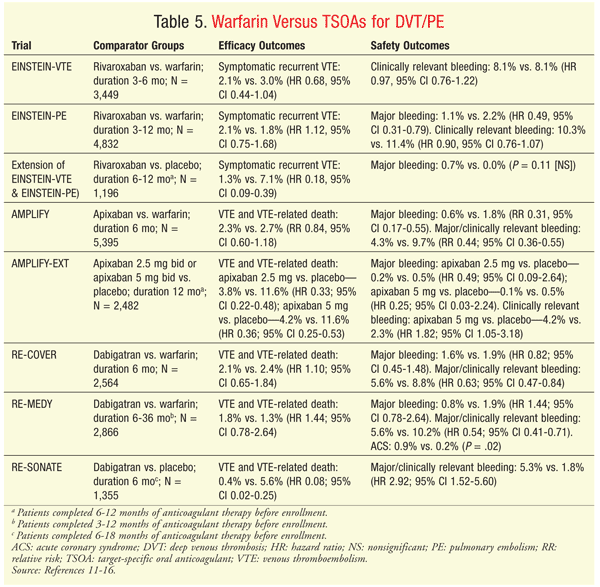
Rivaroxaban is the only TSOA FDA-approved for treatment of acute DVT or PE and prophylaxis for recurrent TEE. In clinical trials, rivaroxaban 15 mg twice daily was administered for the first 3 weeks, followed by 20 mg daily for the remainder of the treatment period.11,12 Compared with standard anticoagulation with LMWH and warfarin, rivaroxaban was noninferior for treating acute DVT or PE.11,12 In both trials, the bleeding risk was similar between the two comparator groups; however, there was a significantly lower risk of major bleeding in rivaroxaban patients versus warfarin patients in the treatment of acute PE. In an extension of the original acute DVT and PE trials, patients who had completed 6 to 12 months of anticoagulant therapy and for whom there was clinical equipoise about whether to continue anticoagulation were randomized to rivaroxaban 20 mg daily or placebo. Rivaroxaban significantly reduced the risk of recurrent VTE without increasing the risk of bleeding.11
When used for the acute treatment of DVT or PE, apixaban 10 mg twice daily for 7 days followed by 5 mg twice daily was noninferior to standard anticoagulation with LMWH and warfarin, but it had a significantly lower risk of major and clinically relevant bleeding.13 When apixaban was continued for an additional 12 months after initial treatment, both 5 mg twice daily and 2.5 mg twice daily were superior to placebo for preventing recurrent TEEs.14 Although neither regimen increased the risk of major bleeding compared with placebo, the 5 mg twice daily regimen increased the risk of clinically relevant bleeding.14
Dabigatran 150 mg twice daily was noninferior to warfarin for both acute treatment of VTE and extended prophylaxis of recurrent VTE, and it carried a lower risk of major and clinically relevant bleeding.15,16 Dabigatran was associated with a higher rate of acute coronary syndrome (ACS) versus warfarin when therapy was extended.16 In addition, unlike patients in the trials investigating the other TSOAs, dabigatran patients in this trial received at least 5 days of parenteral anticoagulation (e.g., LMWH) upon initiation of therapy for acute TEEs.15
Special Populations and Considerations
Older patients are more likely than younger patients to require oral anticoagulation; however, elderly patients are more likely to experience AEs as a result. Although warfarin has been used successfully for years, it remains one of the primary reasons for AEs in elderly patients in the emergency department.17 Most data supporting the use of TSOAs in AF are based on patients younger than 80 years. Only 17% of patients in the RE-LY trial and 13% of those in ARISTOTLE were 80 years or older.9,10 The ROCKET-AF trial included no patients 80 years and older.8 The 2012 Beers criteria warn against the use of dabigatran in patients aged 75 years and older, since dabigatran confers a higher extracranial bleeding risk compared with warfarin.18,19 The increase in major GI bleeds associated with rivaroxaban causes some concern with the use of this agent in older patients.8 Apixaban may be a safer alternative, as it has been associated with a lower rate of major and GI bleeding versus warfarin.9
Renal dysfunction requires a careful analysis of the anticoagulant options. With all three TSOAs, reduced dosing is recommended for patients with renal dysfunction; however, not all adjustments have been evaluated for safety and efficacy. The lower dosage of apixaban—2.5 mg twice daily—has been assessed in clinical trials; however, patients with a creatinine clearance (CrCl) <25 mL/min were excluded. The dabigatran dosage in AF patients with a CrCl <30 mL/min is a 50% reduction from the original dosage and was determined by pharmacokinetic modeling, not clinical trials.20 In trials evaluating the efficacy and safety of dabigatran in patients with AF or VTE, patients with a CrCl <30 mL/min were excluded.10,15,16 The reduced dosage of rivaroxaban—15 mg daily—has been evaluated in patients with a CrCl of 15 to 49 mL/min and NVAF8; however, for TEE treatment or prevention, rivaroxaban is not recommended in patients with a CrCl <30 mL/min.6
In pregnancy, oral anticoagulants are not favored, as data supporting their safety and efficacy are not available. LMWH remains the standard in pregnant patients requiring anticoagulant therapy.21
As noted in the RE-MEDY trial, compared with warfarin, dabigatran may increase the risk of ACS. A meta-analysis of seven noninferiority trials comparing dabigatran with warfarin revealed a slightly higher risk of myocardial infarction or ACS in patients taking dabigatran.22 It is not known whether this is because dabigatran actually increases the risk or because it is less beneficial than warfarin for lowering risk. However, it may be reasonable to select either warfarin or another TSOA over dabigatran in patients with a high risk or past history of coronary artery disease.
Conclusion
In NVAF, apixaban and dabigatran are superior to warfarin for preventing TEEs; apixaban may be the appropriate agent if the patient is at high risk for GI bleeding. Rivaroxaban, which is noninferior to warfarin, may be an option in patients who would benefit from once-daily dosing.
In addition, TSOAs may be reasonable alternatives to warfarin for the acute treatment and prophylaxis of recurrent DVT or PE in patients with a CrCl >30 mL/min. Rivaroxaban, which is FDA-approved for this indication, may be an option for patients who require once-daily dosing but have difficulty with the monitoring requirements for warfarin. Apixaban is an attractive alternative because of its potentially lower bleeding risk compared with warfarin; however, it requires twice-daily dosing. While dabigatran also has shown a lower bleeding risk versus warfarin, the extended trial demonstrated a potential risk of ACS, so this agent may not be the best choice for patients with recent ACS or with a high risk of cardiovascular disease.
REFERENCES
1. You JJ, Singer DE, Howard PA, et al. Antithrombotic therapy for
atrial fibrillation: Antithrombotic Therapy and Prevention of
Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based
Clinical Practice Guidelines. Chest. 2012;141(suppl 2);e531S-e575S.
2. Kearon C, Aki EA, Comerota AJ, et al. Antithrombotic therapy for
VTE disease: Antithrombotic Therapy and Prevention of Thrombosis, 9th
ed: American College of Chest Physicians Evidence-Based Clinical
Practice Guidelines. Chest. 2012;141(suppl 2):e419S-494S.
3. Baker WL, Cios DA, Sander SD, Coleman CI. Meta-analysis to assess
the quality of warfarin control in atrial fibrillation patients in the
United States. J Manag Care Pharm. 2009;15:244-252.
4. Nieuwlaat R, Connolly BJ, Hubers LM, et al. Quality of individual
INR control and the risk of stroke and bleeding events in atrial
fibrillation patients: a nested case control analysis of the ACTIVE W
study. Thromb Res. 2012;129:715-719.
5. Eliquis (apixaban) product information. Princeton, NJ: Bristol-Myers Squibb Co; December 2012.
6. Xarelto (rivaroxaban) product information. Titusville, NJ: Janssen Pharmaceuticals, Inc; August 2013.
7. Pradaxa (dabigatran) product information. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals, Inc; December 2013.
8. Patel MR, Mahaffey KW, Garg J, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365:1557-1559.
9. Granger CB, Alexander JH, McMurray JJ, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011;365:981-992.
10. Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361:1139-1151.
11. EINSTEIN Investigators. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med. 2010;363:2499-2510.
12. EINSTEIN-PE Investigators. Oral rivaroxaban for the treatment of symptomatic pulmonary embolism. N Engl J Med. 2012;366:1287-1297.
13. Agnelli G, Buller HR, Cohen A, et al. Oral apixaban for the treatment of acute venous thromboembolism. N Engl J Med. 2013;369:799-808.
14. Agnelli G, Buller HR, Cohen A, et al. Apixaban for extended treatment of venous thromboembolism. N Engl J Med. 2013;368:699-708.
15. Schulman S, Kearon C, Kakkar AK, et al. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med. 2009;361:2342-2352.
16. Schulman S, Kearon C, Kakkar AK, et al. Extended use of dabigatran, warfarin, or placebo in venous thromboembolism. N Engl J Med. 2013;368:709-718.
17. Budnitz DS, Pollock DA, Weidenbach KN, et al. National
surveillance of emergency department visits for outpatient adverse drug
events. JAMA. 2006;296:1858-1866.
18. American Geriatrics Society 2012 Beers Criteria Update Expert
Panel. American Geriatrics Society updated Beers Criteria for
potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2012;60:616-631.
19. Eikelboom JW, Wallentin L, Connolly SJ, et al. Risk of bleeding
with 2 doses of dabigatran compared with warfarin in older and younger
patients with atrial fibrillation: an analysis of the randomized
evaluation of long-term anticoagulant therapy (RE-LY) trial. Circulation. 2011;123:2363-2372.
20. Varughese CJ, Halperin JL. Prevention of stroke in patients with
atrial fibrillation: anticoagulant and antiplatelet options. J Interv Card Electrophysiol. 2012;35:19-27.
21. Bates SM, Greer IA, Middeldorp S, et al. VTE, thrombophilia,
antithrombotic therapy, and pregnancy: Antithrombotic Therapy and
Prevention of Thrombosis, 9th ed: American College of Chest Physicians
Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(suppl 2):e691S-736S.
22. Uchino K, Hernandez AV. Dabigatran association with higher risk
of acute coronary events: meta-analysis of noninferiority randomized
controlled trials. Arch Intern Med. 2012;172:397-402.
To comment on this article, contactrdavidson@uspharmacist.com.





