US Pharm. 2012;37(6):34-38.
Testosterone deficiency, also called androgen deficiency syndrome or hypogonadism, typically occurs during the postpubertal phase of a man’s life. Low testosterone concentrations affect approximately 4 million men in the United States, and testosterone replacement therapy (TRT) in aging men is widespread and on the increase.1 Up to 20% of men aged older than 70 years are estimated to have hypogonadism. According to pharmaceutical-industry estimates, more than 1.8 million prescriptions for testosterone products were written in the U.S. in 2002, a 30% increase over 5 years.2 In 2005, 2.3 million prescriptions were written for men with testosterone deficiency.3
Pathophysiology
Testosterone, a steroid hormone, plays a key role in male development by exerting anabolic and androgenic effects. Anabolic effects include growth of muscle mass, strength, increased bone density, and bone maturation. Androgenic effects include maturation of sex organs prenatally and in early infancy. The androgenic secondary sex characteristics (usually occurring at puberty) include deepening of the voice and growth of beard and axillary hair.
Testosterone is formed via the hypothalamic–pituitary–gonadal (HPG) axis. The hypothalamus releases gonadotropin-releasing hormone (GnRH), which stimulates the release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) by the anterior pituitary.4 LH stimulates the production of testosterone, but once the testosterone reaches a certain blood concentration, it inhibits the release of GnRH by the hypothalamus.4 This inhibition, in turn, limits the release of LH by the anterior pituitary, thus reducing testosterone production.
There are three forms of hypogonadism.5 Primary hypogonadism, the most common type, results when HPG-axis abnormalities exist at the testicular level and the testes fail to respond to FSH and LH.5 Primary failure involves low testosterone concentrations, elevated gonadotropin concentrations, and impaired spermatogenesis. Secondary hypogonadism is caused by central defects in the hypothalamus or pituitary in which insufficient FSH and LH are produced. Patients will have low or low-to-normal gonadotropin and low testosterone concentrations.6 A pituitary tumor or systemic illness may be the cause of this type of hypogonadism.6 The rarest form of hypogonadism, mixed, results when there are defects at the hypothalamus-pituitary level along with a defect at the testicular level of the HPG axis.5
Clinical Features and Diagnosis
Diagnosis of testosterone deficiency requires the presence of symptoms and low serum testosterone concentrations, according to the Endocrine Society’s clinical practice guidelines (TABLE 1). For most symptoms, the average testosterone threshold corresponding to the lower limit of normal (LLN) in young men is approximately 300 ng/dL, with a greater likelihood of symptoms occurring below this threshold than above it.7

Serum testosterone concentrations are higher in the morning and exhibit diurnal patterns in younger men, compared with the blunted diurnal patterns in most older men.8,9 Serum testosterone concentrations exhibit a circadian variation with peak values in the morning.7 Because of this variation and the fact that normal ranges for serum testosterone are usually established using morning blood samples, testosterone measurement for diagnosing androgen deficiency should be performed between 8 AM and 11 AM.7 It also is important to confirm low concentrations in men with an initial testosterone concentration in the mildly hypogonadal range because 30% of these patients have a normal testosterone concentration upon repeat measurement.
Free testosterone should be measured if it is suspected that the patient has alterations in sex hormone–binding globulin (SHBG). Testosterone is highly bound to SHBG and albumin; thus, when SHBG concentrations are significantly affected, circulating testosterone concentrations can be affected as well. Free testosterone concentrations should be measured when total testosterone concentrations are close to the LLN and when altered SHBG concentrations are suspected in older men and in those with obesity, diabetes mellitus (DM), chronic illness, or thyroid disease.7 The free testosterone concentration, measured by equilibrium dialysis, should be 5 to 9 pg/mL.10 The LLN should be used for healthy young men. Diagnostic strategy should aim to avoid TRT in men without unequivocally low testosterone concentrations and in men with low testosterone due to SHBG abnormalities, natural variations in testosterone concentrations, or transient disorders.
Benefits of TRT
Testosterone deficiency has varied manifestations depending upon its degree and duration. Restoring serum testosterone concentrations to normal range can produce beneficial effects in men.11 TRT can improve libido, erectile function, mood and cognition, body composition and strength, DM, and bone mineral density (BMD).12
Sexual Function: Overall sexual function in men with testosterone deficiency improves with restored concentrations of testosterone. Testosterone treatment can have a positive effect on sexual thoughts, motivation, achievement of erections, and successful intercourse. Improvements in sexual activity following testosterone replacement are not dose dependent, and sexual function returns to normal once serum testosterone is restored to lower adult male concentrations.13 TRT alone may be insufficient to improve sexual function in some men with testosterone deficiency and comorbid vascular disease.
Mood and Cognition: Results regarding the beneficial effects of TRT on mood and cognition are mixed. In a meta-analysis reviewing impact on mood, TRT was found to be beneficial.14,15 Improvements in testosterone may also benefit men with mild cognitive deficits or memory disorders. Positive mood scores were shown to improve with treatment and be sustained, whereas negative mood parameters were reduced and remained significantly lower than baseline, without further changes, after 6 months of TRT.16
Body Composition and Strength: TRT has been proven to significantly alter body mass and strength. TRT reduces fat mass and increases lean body mass, with no overall change in body weight.16 These effects have been noted in men with average baseline testosterone levels below and above 300 mg/dL. In the Hormonal Regulators of Muscle and Metabolism in Aging study, significant increases in total lean body mass and appendicular lean tissue occurred, as well as a significant reduction in total fat mass and abdominal fat. Moreover, composite upper- and lower-body muscle strength increased, as did aerobic endurance.17
DM: Low testosterone is common in men with type 2 DM and may be associated with insulin resistance.18 In addition, free testosterone concentrations have been independently correlated with indices of insulin resistance in both men with type 2 DM and those with type 1 DM.18,19 Some studies have found that TRT improves hemoglobin A1C, but whether it improves glucose control remains to be established in prospective trials.18
BMD: TRT can significantly increase BMD in men with documented hypogonadism.20 Hypogonadism is the most common secondary cause of osteoporosis in men and has been shown to result in bone loss and increased fracture risk. Testosterone androgens appear to play a significant role in bone homeostasis by stimulating bone formation and inhibiting bone breakdown.
Potential Risks of TRT
The risks and adverse effects of TRT vary from mild to serious, and can even be life-threatening. Mild risks include acne, worsened male pattern baldness, gynecomastia, and suppressed spermatogenesis. More serious effects include precipitation or worsening of sleep apnea; benign prostatic hyperplasia (BPH); prostate cancer; accelerated growth of breast cancer; erythrocytosis; dyslipidemia; and cardiovascular (CV) events.21
Acne and Male Pattern Baldness: The prevalence of acne may increase with TRT. Sebum, the oily substance secreted by the sebaceous glands to help prevent hair and skin from drying out, can cause both acne and baldness. TRT can increase sebum secretion through the conversion of testosterone to dihydrotestosterone. The excess sebum can block pores in hairless areas and build up under the skin, causing acne. This blockage also affects hairy areas such as the scalp and can damage hair follicles. Treatment with an 5-alpha reductase inhibitor can alleviate this effect.4
Gynecomastia: TRT can cause or worsen the development of mammary glands in some males.22 Usually this abnormal effect does not require discontinuation of treatment. If the breasts are sore, treatment with an antiestrogen such as tamoxifen can be benefical.23
Sleep Apnea: Low testosterone in males may be caused by sleep apnea, and treatment of sleep apnea may increase testosterone concentrations.24,25 Excessive doses of testosterone have been reported to cause or worsen sleep apnea, but evidence that replacement doses of testosterone cause sleep apnea is lacking.26 Patients with symptoms suggestive of sleep apnea should be evaluated before testosterone treatment is initiated.26
BPH, Prostate Cancer, and Breast Cancer: Testosterone treatment can stimulate BPH; it also can seriously affect metastatic prostate cancer and breast cancer, since both of these cancers are hormone dependent.7 Testosterone should not be administered to men with prostate cancer or breast cancer.25,27 TRT can significantly increase the risk of symptomatic BPH and may increase serum prostate-specific antigen (PSA).27
CV Effects: Men have a higher incidence of CV disease than women, which is the basis for the theory that testosterone is a predisposing factor for CV disease.28 However, men with lower testosterone concentrations are at increased risk for CV events compared with men with high concentrations.29 Evidence is insufficient as to whether TRT would increase, decrease, or have no effect on the incidence of CV disease. Elevated testosterone concentrations stimulate erythropoiesis. Erythrocytosis has been associated with an increase in CV events such as myocardial infarction, coronary insufficiency, and death from coronary heart disease. Recently, high hematocrit and blood viscosity have been associated with carotid atherosclerosis, ischemic disease, and stroke.
Treatment Options
The use of TRT in males has been well documented. The principal goal of TRT is to restore serum testosterone to the normal range. Treatment with testosterone gel, the transdermal patch, or intramuscular (IM) injection is indicated for men with low total serum testosterone concentrations and hypogonadal symptoms such as loss of sexual interest and activity, decreased muscle mass, and reduced BMD. Regardless of the route of administration, improvements in libido and sexual function have been shown in hypogonadal men.
It is recommended that men receiving TRT be monitored according to a standard plan during the course of treatment. Baseline testosterone concentrations should be measured and monitored 3 to 6 months after initiation of therapy to ensure that they are within normal range (TABLE 2).30 A digital rectal examination and PSA test should be performed in most men prior to and during therapy. Hematocrit should be checked periodically for evidence of polycythemia, which may occur in patients receiving androgen therapy.
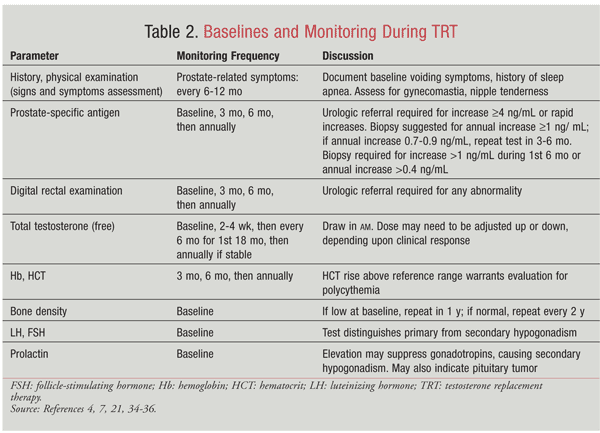
TRT products, particularly the oral form, may cause liver and lipid abnormalities. Liver-function tests and a lipid panel should be performed at baseline and periodically during therapy. Testosterone products are available in a variety of forms, including buccal, oral, parenteral, transdermal, and pellets (TABLE 3). The optimal delivery method for testosterone has not been ascertained.30,31 Determining factors for the selection of an appropriate testosterone delivery method include pharmacokinetic properties, treatment burden, and therapy cost. Testosterone is well absorbed from the intestine, but it is heavily metabolized by liver enzymes, causing an overall reduction in bioavailability. Since oral testosterone is so easily and rapidly metabolized, researchers modified the molecule, ultimately changing the method of delivery. Alkylating androgens such as methyltestosterone are available for oral use; however, they are not widely used because of a lack of virilization and the increased potential for hepatotoxicity.32
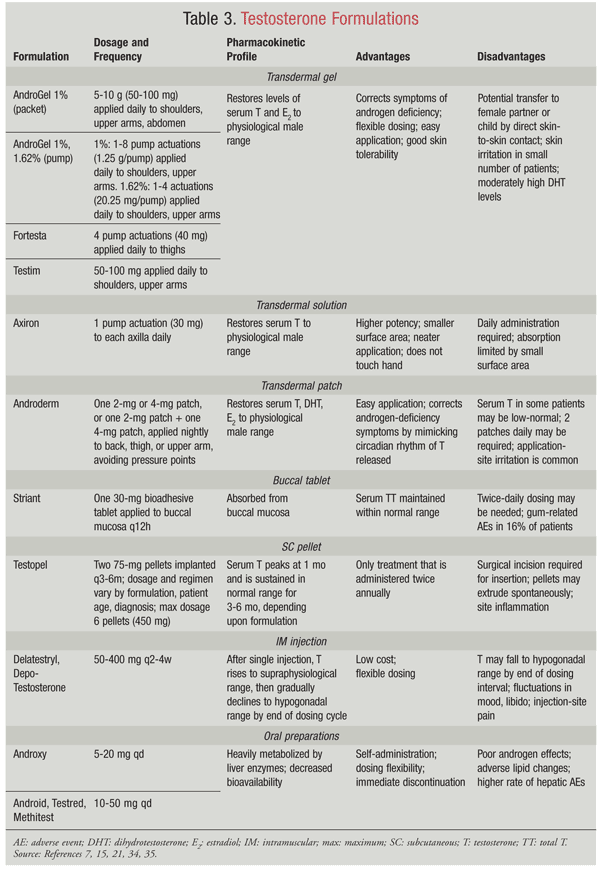
Three testosterone gels are currently available: Androgel, Testim, and Fortesta. These products provide longer-lasting elevations of serum testosterone compared with the transdermal patch delivery system.33 Axiron, another testosterone product, is available as a solution.33
Testosterone is also available in a buccal formulation. Testosterone administered via the transbuccal route is not subject to first-pass hepatic metabolism.33 Pharmacokinetically, the buccal testosterone delivery system releases testosterone in a pulsatile manner, mimicking endogenous testosterone secretion with rapid peak testosterone levels, and steady state is achieved by the second dose.
Another testosterone formulation is esterified testosterone, which is used primarily as an IM injection; it is suggested that this method allows the testosterone to become more lipophilic, enabling optimal storage and release from the oil-based vehicle. Proper administration technique is crucial with IM injections. These oily, highly viscous preparations may be difficult to withdraw from the vial without the proper needle size. The 23-gauge, 1-inch and 25-gauge, 1-inch needles are standard for testosterone injections. The smaller-gauge needle (higher number) will reduce tissue scarring after years of TRT. Needle sizes above 25-gauge may be used, but it may take longer to withdraw and administer the solution. Testosterone is also formulated as fused, cylindrical, crystalline, subcutaneous testosterone pellet implants. The pellets maintain normal serum levels of testosterone for months and provide a long duration of biological activity.33
Conclusion
Pharmacists play an important role in the treatment of men with testosterone deficiency. Testosterone deficiency has been associated with a wide range of potentially distressing signs and symptoms, but TRT can be beneficial. Counseling and medication therapy management are essential for proper administration of TRT. Depending upon the product being used, pharmacists must advise patients about skin irritation at the application or injection site, as well as caution them about the potential transfer of the product to a female partner or child by direct skin-to-skin contact.
REFERENCES
1. Rhoden EL, Morgentaler A. Risks of testosterone-replacement therapy and recommendations for monitoring. N Engl J Med. 2004;350:482-492.
2. Liverman CT, Blazen DG, eds. Testosterone and Aging: Clinical Research Directions. Washington, DC: National Academies Press; 2004.
3. The extent and nature of testosterone use [news release]. Fairfield, CT: IMS Health, Inc; September 2006.
4. Cawley MH. Testosterone replacement therapy: what to look for, when to treat. Nurse Pract. 2009;34:47-52.
5. Kelleher S, Conway AJ, Handelsman DJ. Blood testosterone threshold for androgen deficiency symptoms. J Clin Endocrinol Metab. 2004;89:3813-3817.
6. Bhasin S, Cunningham GR, Hayes FJ, et al. Testosterone therapy in
adult men with androgen deficiency syndromes: an Endocrine Society
clinical practice guideline. J Clin Endocrinol Metab. 2006;91:1995-2010.
7. Bhasin S, Cunningham GR, Hayes FJ, et al. Testosterone therapy in
men with androgen deficiency syndromes: an Endocrine Society clinical
practice guideline. J Clin Endocrinol Metab. 2010;95:2536-2559.
8. Brambilla DJ, Matsumoto AM, Araujo AB, McKinlay JB. The effect of
diurnal variation on clinical measurement of serum testosterone and
other sex hormone levels in men. J Clin Endocrinol Metab. 2009;94:907-913.
9. Bremner WJ, Vitiello MV, Prinz PN. Loss of circadian rhythmicity in blood testosterone levels with aging in normal men. J Clin Endocrinol Metab. 1983;56:1278-1281.
10. Harman SM, Metter EJ, Tobin JD, et al. Longitudinal effects of
aging on serum total and free testosterone levels in healthy men. J Clin Endocrinol Metab. 2001;86:724-731.
11. Marks LS, Mazer NA, Mostaghel E, et al. Effect of testosterone
replacement therapy on prostate tissue in men with late-onset
hypogonadism: a randomized controlled trial. JAMA. 2006;296:2351-2361.
12. Wang C, Cunningham G, Dobs A, et al. Long-term testosterone gel
(AndroGel) treatment maintains beneficial effects on sexual function and
mood, lean and fat mass, and bone mineral density in hypogonadal men. J Clin Endocrinol Metab. 2004;89:2085-2098.
13. Bhasin S, Woodhouse L, Casaburi R, et al. Testosterone dose-response relationships in healthy young men. Am J Physiol Endocrinol Metab. 2001;281:E1172-E1181.
14. Zarrouf FA, Artz S, Griffith J, et al. Testosterone and depression: systemic review and meta-analysis. J Psychiatr Pract. 2009;15:289-305.
15. Shores MM, Kivlahan DR, Sadak TI, et al. A randomized,
double-blind, placebo-controlled study of testosterone treatment in
hypogonadal older men with subthreshold depression (dysthymia or minor
depression). J Clin Psychiatry. 2009;70:1009-1016.
16. Isidori AM, Giannetta E, Greco EA, et al. Effects of testosterone
on body composition, bone metabolism and serum lipid profile in middle
aged men: a meta-analysis. Clin Endocrinol (Oxf). 2005;63:280-293.
17. Knezevich EL, Knezevich JT. Testosterone deficiency in men: more common than you think. US Pharm. 2011;37(12)(suppl).
18. Grossmann M, Thomas MC, Panagiotopoulos S, et al. Low
testosterone levels are common and associated with insulin resistance in
men with diabetes. J Clin Endocrinol Metab. 2008;93:1834-1840.
19. Selvin E, Feinleib M, Zhang L, et al. Androgen and diabetes in
men: results from the Third National Health and Nutrition Examination
Survey (NHANES III). Diabetes Care. 2007;30:234-238.
20. Behre HM, Kliesch S, Leifke E, et al. Long-term effect of testosterone therapy on bone mineral density in hypogonadal men. J Clin Endocrinol Metab. 1997;82:2386-2390.
21. Vondracek SF, Hansen LB. Current approaches to the management of osteoporosis in men. Am J Health Syst Pharm. 2004;61:1801-1811.
22. Braunstein GD. Gynecomastia. N Engl J Med. 2007;357:1229-1237.
23. Deplewski D, Rosenfield RL. Role of hormones in pilosebaceous unit development. Endocr Rev. 2000;21:363-392.
24. Grunstein RR, Handelsman DJ, Lawrence SJ, et al. Neuroendocrine
dysfunction in sleep apnea: reversal by continuous positive airways
pressure therapy. J Clin Endocrinol Metab. 1989;68:352-358.
25. Santamaria JD, Prior JC, Fleetham JA. Reversible reproductive dysfunction in men with obstructive sleep apnoea. Clin Endocrinol (Oxf). 1988;28:461-470.
26. Cunningham GR, Toma SM. Why is androgen replacement in males controversial? J Clin Endocrinol Metab. 2011;96:38-52.
27. Advisory Panel on Testosterone Replacement in Men. Report of
National Institute on Aging Advisory Panel on Testosterone Replacement
in Men. J Clin Endocrinol Metab. 2001;86:4611-4614.
28. Wu FC, von Eckardstein A. Androgens and coronary artery disease. Endocr Rev. 2003;24:183-217.
29. Liu PY, Death AK, Handelsman DJ. Androgens and cardiovascular disease. Endocr Rev. 2003;24:313-340.
30. Comparison of testosterone products. Pharmacist’s Letter. 2011;27:270-301.
31. Margo K, Winn R. Testosterone: why, when, and how? Am Fam Physician. 2006;73:1591-1598.
32. Emmelot-Vonk MH, Verhaar HJ, Nakhai Pour HR, et al. Effect of
testosterone supplementation on functional mobility, cognition, and
other parameters in older men: a randomized controlled Trial. JAMA. 2008;299:39-52.
33. Snyder PJ. Clinical features and diagnosis of male hypogonadism.
www.uptodate.com/contents/clinical-features-and-diagnosis-of-male-hypogonadism?source=see_link.
Accessed February 4, 2012.
34. AndroGel 1% (testosterone gel) product information. North Chicago, IL: Abbott Laboratories; November 2011.
35. AndroGel 1.62% (testosterone gel) product information. North Chicago, IL: Abbott Laboratories; April 2011.
36. Seftel A. Testosterone replacement therapy for male hypogonadism;
part III. Pharmacologic and clinical profiles, monitoring, safety
issues, and potential future agents. Int J Impot Res. 2007;19:2-24.
To comment on this article, contact rdavidson@uspharmacist.com.





