Asthma, a chronic long-term lung disease, causes inflammation and narrowing of the bronchial airways.1 It affects more than 22 million people nationwide, 6 million of whom are children.1 Though the etiology is unknown, asthma is attributed to a combination of factors such as family genes, environmental factors, and airborne allergens.2 Asthma is characterized by the onset of wheezing, chest tightness, shortness of breath, and coughing, particularly at night or early in the morning.3 Asthmatic patients frequently experience severe airway constriction and inflammation, resulting from bronchial irritation after exposure to allergens and triggers.3 During an attack, the airway lining narrows from the tightening of muscles around the bronchi and from increased production of thick, viscous mucus.1 Common triggers have been identified as pollen, dust mites, pet dander, grasses, peanuts, fresh fruit, soy, tobacco smoke, and preservatives.2,3
Conventional Treatment
Traditionally, people who have asthma have been treated with a myriad of medications to overcome and prevent the inflammatory response. Products such as oral and inhaled corticosteroids, leukotriene modifiers, long- and short-acting beta-2 agonists, theophylline, antihistamines, immunotherapy, and mast cell stabilizers have all been prescribed as viable treatment options. Furthermore, many of the therapeutic modalities employ several routes of delivery, such as tablets, metered-dose and dry powder inhalers, combination products, and nebulizer solutions.4 Although these therapies have been effective in many, some patients advance to severe stages of asthma. In light of such difficult cases, in May, 2010, Asthmatx, Inc., introduced a new option for the treatment of severe, persistent asthma: the Alair Bronchial Thermoplasty System (FIGURE 1). This FDA-approved device is designed to perform a bronchoscopic procedure that can be used to reduce the mass of airway-wall smooth muscle and attenuate bronchoconstriction.5
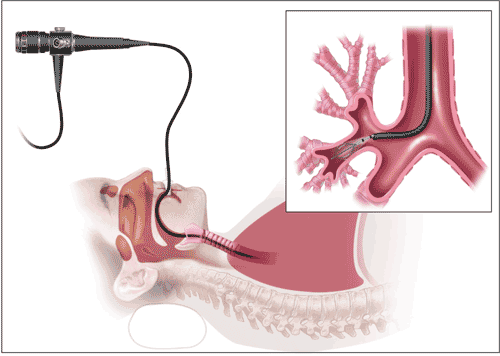
Bronchial thermoplasty (BT) is a minimally invasive outpatient procedure reserved for severe, persistent asthma in patients 18 years and older when conventional methods have been tried but are ineffective.5 Performed under moderate sedation, the procedure works by applying radio frequency heat to the bronchial airway walls, resulting in the mitigation of increased smooth-muscle mass, which is traditionally seen in the airway walls of patients with severe asthma.6,7 A complete BT treatment requires a patient to undergo 3 treatment sessions that are scheduled at least 3 weeks apart. At each session, a different area of the lungs is treated: the right lower lobe, left lower lobe, and both upper lobes are treated in the first, second, and final treatment sessions, respectively. The middle lobe is not treated because of the increased risk of mucus accumulation and atelectasis (partial collapse of the lung) in the post-treatment period.8 An outline of a typical treatment procedure can be found in TABLE 1 and a depiction of the procedure in FIGURE 2.

Following treatment, it is expected that some patients may experience the worsening of respiratory-related symptoms, such as multiple symptoms of asthma, wheezing, chest pain, shortness of breath, and chest discomfort.9 These symptoms may occur within 1 day of the procedure and may persist up to an average of 7 days with standard care. Contraindications of BT may be found in TABLE 2.

Efficacy
Castro et al conducted a randomized, double-blind, sham-controlled study designed to evaluate the safety and effectiveness of the Alair System in adults with severe asthma, known as the AIR2 trial. Participating patients were treated with high doses of standard asthma medications, yet they remained symptomatic.10
The study compared the effects of BT, delivered by the Alair System (BT group) to a sham treatment (sham group), in patients using conventional therapy. Primary end-point outcomes were measured by using the Asthma Quality of Life Questionnaire (AQLQ). The AQLQ assessed the difference between study groups as measured by the change in score from baseline to follow-up, averaged across scores obtained at 6-, 9-, and 12-month follow-up visits. Secondary endpoint outcomes included measures of pulmonary function, exacerbations, and other measures of asthma severity. Baseline demographics and clinical characteristics were well matched between groups. A total of 580 subjects were screened; of the enrolled participants, 297 were randomized to either the BT group or the sham control group, and a total of 288 received treatment (BT: n = 190; sham: n = 98). All patients had severe and inadequately controlled asthma as evidenced by their requirement for high-dose inhaled corticosteroids and long-acting beta-agonist treatment regimens. High asthma control questionnaire scores were consistent with poorly controlled asthma and a low AQLQ score. Furthermore, 86% of the BT group and 88% of the sham control group met American Thoracic Society (ATS) criteria for severe refractory asthma.
In the evaluation of the primary end-point, the study reported that 79% of Alair-treated patients responded with a clinically meaningful improvement in AQLQ score as compared to 64% in the sham control. Secondary end-point treatment outcomes included a 32% reduction in severe exacerbations, an 84% overall reduction in emergency room(ER) visits, and a 66% reduction in days lost from work. Both groups, however, experienced an increase in respiratory-related adverse events, with more events reported in the BT group when compared to the sham group (85% vs. 76%, respectively).10
Conclusion
Bronchial thermoplasty in subjects with severe asthma has been shown to improve asthma-specific quality of life with a reduction in severe exacerbations, ER visits, days lost from work or school, and other measures of asthma severity during the post-treatment period. Though BT has also been associated with increased risk of adverse events (i.e., airway irritation, wheezing, chest discomfort, and pain) in the first year of treatment, in some, the benefits may outweigh the risks.10 Accordingly, clinicians should carefully evaluate patients' risk-benefit profiles and counsel patients regarding risks and benefits prior to recommending the procedure. For more information on the Alair Bronchial Thermoplasty System, visit
www.btforasthma.com/bronchial-thermoplasty/about-bronchial-
thermoplasty.
REFERENCES
1. What is asthma? National Heart Lung and Blood Institute Web site. www.nhlbi.nih.gov/health/dci/Diseases/Asthma/Asthma_Causes.html. Accessed July 17, 2010.
2. Asthma overview. Asthma and Allergy Foundation of America Web site. www.aafa.org/display.cfm?id=8&cont=6. Accessed July 17, 2010.
3. Cox GT, Thomson NC, Rubin AS, et al. Asthma control during the year after bronchial thermoplasty. N Engl J Med. 2007; 356(13):
1327-1337.
4. How is asthma treated and controlled? National Heart Lung and Blood Institute Web site.
www.nhlbi.nih.gov/health/dci/
Diseases/Asthma/Asthma_Treatments.html. Accessed July 17, 2010.
5. Niven R, Pavord I, Laviolette M, et al. Bronchial thermoplasty in refractory asthma: interim results in a severe asthma population (Research in Severe Asthma [RISA] Trial). J Allergy Clin Immunol. 2007;119(1):S1.
6. Firszt R, Kraft M. Pharmacotherapy of severe asthma. Curr Opin Pharmacol. 2010;10:266-271.
7. Solway J, Irvin C. Airway smooth muscle as a target for asthma therapy. N Engl J Med. 2007;
356(13):1367-1369.
8. Fernandez A. Bronchial thermoplasty in the treatment of asthma. Arch Bronconeumol. 2010;46(2):85-91.
9. A new procedure for severe asthma. Sunnyvale, CA: Asthmatx, Inc. http://multivu.prnewswire.com/mnr/bronchialthermoplasty/43629/docs/43629-BT_patient-brochure.pdf. Accessed September 17, 2010.
10. Castro M, Rubin A, Laviolette M, et al. Effectiveness and safety of bronchial thermoplasty in the treatment of severe asthma: a multicenter, randomized, double-blind, sham-controlled clinical trial. Am J Respir Crit Care Med. 2010; 181:116-124.
To comment on this article, contact robdavidson@www.uspharmacist.com.






