US Pharm. 2015;40(2):50-53.
ABSTRACT: The role of inflammatory markers in cardiovascular disease progression and risk assessment has been a topic of discussion for nearly two decades. This subject was most recently addressed by guidelines from the American College of Cardiology and the American Heart Association, as well as the National Lipid Association. Evidence supports the association of high-sensitivity C-reactive protein (hs-CRP) with cardiovascular disease. Hs-CRP testing may improve risk stratification, particularly among intermediate cardiovascular risk patients, although further research is warranted.
The role of inflammatory markers in cardiovascular risk assessment and the development of cardiovascular disease has been a topic of discussion for nearly two decades. In 1998, the American Heart Association (AHA) Prevention Conference V examined strategies for the identification of patients requiring primary cardiovascular prevention.1 The conference concluded that inflammatory markers, including high-sensitivity C-reactive protein (hs-CRP), are not yet considered applicable for routine assessment due to lack of measurement standardization, consistency in epidemiological findings, and evidence for additional risk prediction.1 In 2001, the National Cholesterol Education Program Adult Treatment Panel III Guidelines categorized hs-CRP as an emerging risk factor, which could be used as an optional risk-factor measurement to adjust absolute estimates.2 With an increase in the number of peer-reviewed reports and publications came an increase in the availability of C-reactive protein (CRP) assays and the number of inflammatory marker tests ordered by providers; however, a consensus from professional societies or governmental agencies on applicability to clinical practice had not yet been reached.3
In 2003, the CDC and the AHA released a scientific statement regarding the application of inflammatory markers to clinical and public health practice.3 This joint effort concluded that in patients with stable coronary disease or acute coronary syndromes, hs-CRP measurement may be useful as an independent marker for assessing the likelihood of recurrent events; however, secondary preventive interventions with proven efficacy should not be dependent on these concentrations or levels.3 The 2013 American College of Cardiology (ACC)/AHA Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Risk in Adults stated that hs-CRP may be considered to guide treatment decision making if, after quantitative risk assessment, a risk-based treatment decision is uncertain.4 Most recently, the National Lipid Association recommendations for patient-centered management of dyslipidemia included hs-CRP as a risk indicator that might be considered for risk refinement.5 This article will provide an overview of hs-CRP and its role in cardiovascular disease progression and risk assessment; review the most updated clinical evidence; discuss application to clinical practice; and identify needs for future research.
Background
CRP was discovered nearly 80 years ago by scientists exploring the human inflammatory response.6 CRP is produced by every single human being and plays a crucial role in the immune system as a sensitive and dynamic marker of inflammation.7 CRP can be increased 10,000- to 50,000-fold during acute responses to serious infection or major tissue damage, and usually peaks within 48 hours.7,8 Acute rises in CRP may be attributed to exercise, burns, trauma, or acute bacterial or viral infections. Situations known to cause chronic elevations in CRP levels include elevated blood pressure, elevated body mass index, cigarette smoking, cancer, metabolic syndrome, diabetes mellitus, decreased high-density lipoprotein (HDL), elevated triglycerides, estrogen and progesterone hormone use, chronic bacterial or viral infections, autoimmune conditions, and chronic inflammatory conditions.3
Evidence suggests that chronic CRP elevations may have biologic effects on endothelial function, coagulation, fibrinolysis, oxidation of low-density lipoprotein (LDL), and atherosclerotic plaque stability.9 In fact, from a pathologic viewpoint, all stages of the atherosclerotic plaque might be considered to be an inflammatory response to injury, including plaque rupture and resultant thrombosis; this is supported by consistent clinical findings that hs-CRP predicts new coronary events in patients with unstable angina and acute myocardial infarction.3 Limited evidence also suggests an association between elevated hs-CRP and sudden death and peripheral arterial disease.3
Ultimately, hs-CRP retains an independent association with incidental coronary events even after stratification and multivariable statistical adjustment; however, hs-CRP has not been shown to predict the extent of atherosclerotic disease.3 In addition, the collaborative 2003 CDC/AHA scientific statement noted some evidence to implicate a possible role for hs-CRP in the causation of atherosclerosis.3 More recently, the May 2014 issue of Life Extension magazine featured an article stating that “CRP is more than just a marker of inflammation—it is also a cause of inflammation.”10
Defining hs-CRP
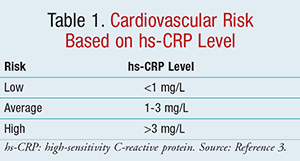
To illustrate the difference between CRP and hs-CRP, traditional testing measures CRP within the range of 10 to 1,000 mg/L, whereas hs-CRP values range from 0.5 to 10 mg/L. In simpler terms, hs-CRP measures trace amounts of CRP in the blood. Hs-CRP is the analyte of choice for cardiovascular risk assessment, due to superior assay precision, accuracy, availability, and the existence of standards for proper calibration when compared to other acute-phase reactants.3 According to the CDC and AHA, as referenced in TABLE 1, low risk for cardiovascular disease is defined as hs-CRP <1 mg/L, average risk as 1 to 3 mg/L, and high risk as >3 mg/L.3 An hs-CRP level >10 mg/L has been observed in acute plaque rupture, which may lead to thrombosis.11 As a measure of control, an hs-CRP level of this caliber should prompt identification of an obvious source of infection or inflammation, which could obscure any prediction of coronary risk that might be attributed to that level.
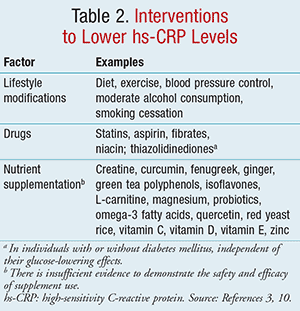
Although intrapersonal variability is common and genetic influences may occur, the coefficient of variation of hs-CRP assays is generally <10% from the 0.3 to 10 mg/L range; and the only known factor to interfere with CRP production is liver failure.6,12 To reduce variability, the hs-CRP assay should be performed in a metabolically stable person without obvious inflammatory or infectious conditions.3,5 It is recommended to check two separate levels approximately 2 weeks apart and use the average of the readings for cardiovascular risk assessment and screening purposes.3 In contrast, interventions known to reduce hs-CRP levels are listed in TABLE 2.3,10
Guidelines
A recently published consensus on the role of hs-CRP in cardiovascular risk assessment and disease progression can be found in the 2013 ACC/AHA Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Risk in Adults.4 The highly anticipated guideline expressed interest regarding biomarkers and noninvasive testing that may indicate elevated atherosclerotic cardiovascular disease (ASCVD) risk, but did not include these factors in its pooled cohort equations for predicting 10-year ASCVD risk. Four major statin benefit groups were identified for whom the ASCVD risk reduction clearly outweighs the risk of adverse events. These groups included those: 1) with clinical ASCVD; 2) with primary elevations of LDL-C (calculated low-density lipoprotein) >190 mg/dL; 3) aged 40 to 75 years with diabetes and LDL-C 70 to 189 mg/dL and without clinical ASCVD; or 4) without clinical ASCVD or diabetes with LDL-C 70 to 189 mg/dL and estimated 10-year ASCVD risk >7.5%.4 These individuals will be referred to as high cardiovascular risk patients.
The final work group report concluded that in selected individuals who are not in one of the four previously mentioned statin benefit groups, and for whom a decision to initiate statin therapy is otherwise unclear, additional factors may be considered to inform treatment decision making.4 Hs-CRP >2 mg/L is included among these risk factors. Individuals falling within these criteria will be further referred to as intermediate cardiovascular risk patients.
The 2013 ACC/AHA Assessment of Cardiovascular Risk Full Work Group examined several systematic reviews and studies: a 2009 review by Buckley et al for the U.S. Preventive Services Task Force (USPSTF) that focused on the potential risk related to CRP >3 mg/L versus CRP <1 mg/L; a 2009 USPSTF report on CRP and eight other risk factors by Helfand et al; a 2010 meta-analysis by Kaptoge et al through the Emerging Risk Factor Collaboration; a 2010 systematic review by Schnell-Inderst et al evaluating the utility of hs-CRP screening in asymptomatic adults; and the JUPITER trial (Justification for the Use of Statins in Primary Prevention: an Intervention Trial Evaluating Rosuvastatin) for CRP levels >2 mg/L.7,13-16
Overall, there is strong evidence indicating that hs-CRP is associated with cardiovascular disease, although the magnitude of these associations may be confounded by established cardiovascular risk factors. Moderate evidence suggests that hs-CRP testing may improve risk stratification, particularly among intermediate cardiovascular risk patients, where the decision to initiate statin therapy is unclear; however, the clinical relevance and cost-effectiveness remains uncertain. For this reason, hs-CRP was not included in the risk prediction model development process for the 2013 ACC/AHA guidelines.4
Of note, the newly published National Lipid Association recommendations for patient-centered management of dyslipidemia identify an hs-CRP level ³2 mg/L as a risk indicator, in conjunction with major ASCVD risk factors, that might be considered for risk refinement.5 The organization is careful to note that “except in the case of evidence of subclinical disease defining the presence of ASCVD, reclassification to a higher risk category is a matter of clinical judgment.”5 The guidelines reference the JUPITER trial, as described above, in their consideration of additional cardiovascular risk factors, but do not provide further evaluation of the evidence.16 A summary of major guideline statements on hs-CRP and cardiovascular risk assessment is provided in TABLE 3.2,4,5

Role of hs-CRP
The above stated compilation of evidence can be used to discuss applicability in clinical practice. Individuals at high risk for cardiovascular events, including secondary prevention candidates, will likely already be on or qualify for therapies known to reduce hs-CRP in addition to improving overall cardiovascular outcomes, such as statin or aspirin therapy. It has been suggested that hs-CRP may be useful as an independent marker of prognosis for recurrent events, including death, myocardial infarction, and restenosis in patients with stable coronary disease or acute coronary syndromes or those who have undergone percutaneous coronary intervention.3 This information may be especially useful when counseling patients on the importance of compliance with secondary preventive interventions.3 Hs-CRP may also be useful in individuals without risk factors for cardiovascular disease, known as low cardiovascular risk patients, who would not otherwise be identified as potential candidates for primary prevention therapy. However, at this time, evidence does not exist to support the clinical relevance or cost-effectiveness of using an hs-CRP >2 mg/L to independently predict cardiovascular risk. In fact, the AHA specifically recommends against screening hs-CRP levels for the entire adult population for purposes of cardiovascular risk assessment.3 Therefore, hs-CRP will have the most significant impact on patients at an intermediate risk for cardiovascular events, where the decision to initiate primary preventive therapy is unclear.
Individuals presenting with an hs-CRP level >2 mg/L with additional cardiovascular risk factors, otherwise classified as intermediate cardiovascular risk patients, should undergo repeat testing in 2 weeks to rule out an acute inflammatory response. Patients with a persistently unexplained, marked elevation of hs-CRP >10 mg/L after repeat testing should be evaluated for noncardiovascular etiologies, such as infection, active arthritis, or concurrent illness.3,5 If the hs-CRP level remains elevated, the patient may be reclassified as being at high cardiovascular risk based on clinical judgment, which would justify initiating therapy for primary cardiovascular prevention. It is important to note that the best ways to lower CRP are already known to lower cardiovascular risk, including diet, exercise, blood pressure control, and smoking cessation.6
Specific areas requiring further investigation include establishing independence of hs-CRP from other cardiovascular risk factors, determining the association with cardiovascular endpoints, establishing the cost-effectiveness of screening hs-CRP levels, and the potentially earlier initiation of primary preventive therapy. The benefit of including hs-CRP in pooled cohort equations to classify cardiovascular risk also requires further investigation, although this has been examined through the Reynolds Risk Score for both men and women.17,18 Specifically in women, the Reynolds Risk Score reclassified 40% to 50% of women at intermediate risk into higher or lower cardiovascular risk categories, and in men the prediction model incorporated hs-CRP, parental history, and significantly improved global cardiovascular risk prediction.17,18 In addition, although this strategy remains uncertain and has not been tested, hs-CRP levels may be useful in motivating patients to improve lifestyle behaviors and comply with drug therapies.3
Conclusion
Strong evidence supports the association of hs-CRP with cardiovascular disease, although the magnitude of these associations may be confounded by established cardiovascular risk factors. Moderate evidence suggests that hs-CRP testing may improve risk stratification, particularly among intermediate cardiovascular risk patients where the decision to initiate primary preventive therapy is unclear. Future research must be conducted to justify a more concrete recommendation for the application of hs-CRP levels in clinical practice. However, with regard to clinical judgment, evidence does exist to support the reclassification of intermediate cardiovascular risk patients based on elevated hs-CRP levels, which would justify initiating therapy for primary cardiovascular prevention. In contrast, evidence does not exist to support the clinical relevance or cost-effectiveness of using elevated hs-CRP levels to independently predict cardiovascular risk in low cardiovascular risk patients. Lifestyle modifications, which are known to lower both CRP and cardiovascular risk, should remain the mainstay of all treatment strategies for those at risk for cardiovascular disease.
REFERENCES
1. Grundy SM, Bazzarre T, Cleeman J, et al. Prevention Conference V: Beyond secondary prevention: identifying the high-risk patient for primary prevention: medical office assessment. Circulation. 2000;101:e3-e11.
2. Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA. 2001;285:2486-2497.
3. Pearson TA, Mensah GA, Alexander RW, et al. Markers of inflammation and cardiovascular disease: application to clinical and public health practice: a statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. Circulation. 2003;107(3): 499-511.
4. Stone NJ, Robinson JG, Lichtenstein AH, et al. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;63:2889-2934.
5. Jacobson TA, Ito MK, Maki KC, et al. National Lipid Association recommendations for patient-centered management of dyslipidemia: part 1—executive summary. J Clin Lipidol. 2014;8(5):473-488.
6. Ridker PM. Cardiology Patient Page. C-reactive protein: a simple test to help predict risk of heart attack and stroke. Circulation. 2003;108(12):e81-e85.
7. Kaptoge S, Di Angelantonio E, Lowe G, et al. C-reactive protein concentration and risk of coronary heart disease, stroke, and mortality: an individual participant meta-analysis. Lancet. 2010;375:132-140.
8. Mcpherson M. C-reactive protein usage in medical treatment. Ezine Articles. June 3, 2010. http://ezinearticles.com/?C-Reactive-Protein-Usage-in-Medical-Treatment&id=4411095 Accessed June 12, 2014.
9. Boekholdt SM, Kastelein JJ. C-reactive protein and cardiovascular risk: more fuel to the fire. Lancet. 2010;375:95-96.
10. Simmonds S. Testing for C-reactive protein may save your life. Life Extension. 2014;20(5):37-47.
11. Hong YJ, Mintz GS, Kim SW, et al. Impact of plaque rupture and elevated C-reactive protein on clinical outcome in patients with acute myocardial infarction: an intravascular ultrasound study. J Invasive Cardiol. 2008;20(9):428-435.
12. Roberts WL, Moulton L, Law TC, et al. Evaluation of nine automated high-sensitivity C-reactive protein methods: implications for clinical and epidemiological applications: part 2. Clin Chem. 1997;43:52-58.
13. Buckley DI, Fu R, Freeman M, et al. C-reactive protein as a risk factor for coronary heart disease: a systematic review and meta-analyses for the U.S. Preventive Services Task Force. Ann Intern Med. 2009;151:483-495.
14. Helfand M, Buckley DI, Freeman M, et al. Emerging risk factors for coronary heart disease: a summary of systematic reviews conducted for the U.S. Preventive Services Task Force. Ann Intern Med. 2009;151:496-507.
15. Schnell-Inderst P, Schwarzer R, Göhler A, et al. Prognostic value, clinical effectiveness, and cost-effectiveness of high-sensitivity C-reactive protein as a marker for major cardiac events in asymptomatic individuals: a health technology assessment report. Intl J Tech Assess Health Care. 2010;26:30-39.
16. Ridker PM, Danielson E, Fonseca FA, et al; JUPITER Study Group. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med. 2008;359:2195-2207.
17. Ridker PM, Buring JE, Rifai N, Cook NR. Development and validation of improved algorithms for the assessment of global cardiovascular risk in women: the Reynolds Risk Score. JAMA. 2007;297:611-619.
18. Ridker PM, Paynter NP, Rifai N, et al. C-reactive protein and parental history improve global cardiovascular risk prediction: the Reynolds Risk Score for men. Circulation. 2008;118:2243-2251.
To comment on this article, contact rdavidson@uspharmacist.com.





