US Pharm. 2010;35(9):41-48.
Lyme disease, also known as Lyme borreliosis, is the most common vector-transmitted illness in the United States.1,2 The disease was first recognized in 1975 in a number of children in Lyme, Connecticut, who were initially diagnosed with juvenile rheumatoid arthritis because of overlapping signs and symptoms.1 In 1982, Willy Burgdorfer, PhD, discovered that a spirochete isolated from Ixodes scapularis, a deer tick, was the same spirochete found in patients with Lyme disease.
Confirmed cases of Lyme disease are defined as erythema migrans (EM) greater than 5 cm in diameter or one or more objective late manifestations of Lyme disease with laboratory confirmation in patients with possible exposure to ticks. Healthy People 2010 set a goal of a 44% decrease in the incidence of Lyme disease by 2010. Since 2000, the CDC has contributed more than $3.5 million per year for new research on Lyme disease.2
The white-footed mouse and the white-tailed deer are the primary reservoirs for Lyme disease transmission in the U.S.2,3 In the U.S., the two insect vectors that carry the gram-negative pathogen Borrelia burgdorferi are I scapularis (black-legged or deer tick) and Ixodes pacificus (Western black-legged tick).1 Northeastern, Midwestern, and mid-Atlantic states experience pathogen transmission primarily by the black-legged tick, whereas the Western black-legged tick is the cause of infection on the Pacific Coast (FIGURE 1).1,3
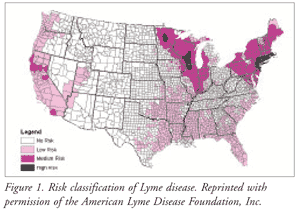
Infection rates are highest during the spring to early fall, with most cases occurring from June to August.1-3 The Northeastern Coast, the Pacific Coast, and the Midwestern states (Great Lakes region) have the highest rates of infection in the U.S.2,3 Infections predominate in heavily forested areas.
Pathogenesis
Both the black-legged tick and the Western deer tick have three stages of development that affect the rate of pathogen transmission to humans (FIGURE 2).1,3 After hatching from the egg, the larva (stage 1) latches onto and feeds off the blood of the white-tailed mouse (reservoir), which may or may not contain the B burgdorferi pathogen.1,3 Next (stage 2), during late spring, the fed larva falls off the mouse reservoir and transforms into a dormant nymph. The following spring, nymphal activity begins. The tick attaches to a new host, possibly a human. This stage is responsible for most cases of Lyme disease because the nymph tick, which is the size of a poppy seed, often goes undetected until it is fully engorged with blood.
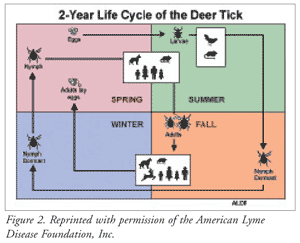
Once fully fed, the tick detaches itself onto taller foliage to ensure attachment to its preferred host, the deer, although any mammal (e.g., human, dog, horse) may be targeted. At this time, it also transforms into an adult (stage 3) and becomes significantly larger; it is now more readily discovered and less likely to spread disease. Transmission of B burgdorferi can occur within 24 hours of attachment, but most transmissions occur after 36 to 48 hours of consistent feeding by the tick. Once winter begins and fewer hosts are available, the adult is dormant until the spring, when it looks for another host. Once a host is found and the adult feeds, it mates and lays eggs, perpetuating the cycle.
Prevention and Prophylaxis
There are measures people can take to prevent infection with Lyme borreliosis.3 The best method is to avoid tick-infested areas. Other measures include frequently checking skin and clothing, wearing light-colored clothing that easily shows ticks, wearing protective clothing (e.g., long-sleeved shirt tucked into pants, long pants tucked into socks), spraying skin and clothing with tick and insect repellents (containing N,N-diethyl-3-methylbenzamide [DEET]), and removing an attached tick with a fine-tip tweezer or forceps.3 DEET should be reapplied frequently; although there have been cases of neurologic symptoms from overuse in children, the Infectious Diseases Society of America (IDSA) recommends its use in children as young as 2 months if used as directed.3 An available permethrin clothing spray kills ticks upon contact; it should not be used on the skin owing to inactivation caused by skin oils.3 A previously available vaccine (LYMErix) was discontinued by the manufacturer (SmithKline Beecham Pharmaceuticals) in 2002 because of low sales.4 Currently, no Lyme disease vaccine is available.
Studies indicate that transmission of B burgdorferi requires at least 36 hours of attachment.5 After a recognized tick bite, routine prophylaxis with a single dose of doxycycline 200 mg is not recommended unless the following four conditions are met: “(a) the attached tick can be reliably identified as an adult or nymphal I. scapularis tick that is estimated to have been attached for ≥36 h on the basis of the degree of engorgement of the tick with blood or of certainty about the time of exposure to the tick; (b) prophylaxis can be started within 72 h of the time that the tick was removed; (c) ecologic information indicates that the local rate of infection of these ticks with B. burgdorferi is ≥20%; and (d) doxycycline treatment is not contraindicated.”3 Prophylaxis after an I pacificus tick bite usually is not needed because the rate of infection is low.3
Presentation and Treatment
Lyme disease has three stages: early localized, early disseminated, and late persistent (TABLE 1).2 The stage the patient presents with determines the choice of antibiotic and duration of treatment. Early localized disease frequently begins with the classic bull's-eye lesion--erythema migrans--at the site of the bite. This characteristic skin lesion is regarded as the strongest diagnostic sign of Lyme disease, manifesting in 90% of cases and usually appearing within a month of the bite. Early disease may be characterized by cardiac and neurologic involvement. Laboratory data are not often used owing to slow multiplication of the cultured organism, low sensitivity of polymerase chain reaction, and confounding of past infections versus active infections in serologic testing.6 The pathogen disseminates over time; a patient may present with multiple lesions, and constitutional symptoms may be more pronounced. In late persistent Lyme disease, the pathogen has further infiltrated various organ systems; notably, it causes arthritis in large joints, particularly the knee.
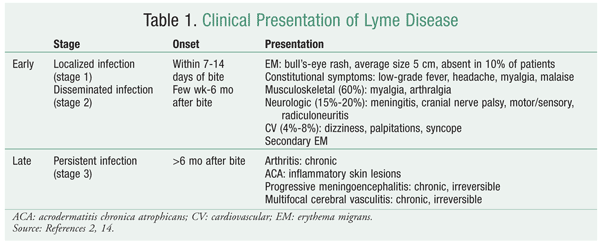
Antimicrobials are an effective therapy for Lyme disease. The IDSA has developed evidence-based guidelines to aid in the management of Lyme borreliosis. Multiple studies have shown that B burgdorferi is sensitive to tetracyclines, penicillins, and second- and third-generation cephalosporins.7,8 The use of macrolides as first-line therapy is not recommended, as clinical trials found them to be less effective than other antibiotics.9-11 Macrolide therapy should be reserved for patients who are intolerant to or cannot take any of the aforementioned antibiotics. First-generation cephalosporins should never be used to treat Lyme disease.
During the early localized stage, which involves EM, adults and children may be treated with doxycycline, amoxicillin, or cefuroxime axetil. Doxycycline should not be given to pregnant or breastfeeding women or to children under 8 years of age owing to adverse effects such as increased risk of teratogenicity in the former and tooth discoloration in the latter. In the absence of these contraindications, doxycycline is the drug of choice because it also is active against common co-infecting organisms like Anaplasma phagocytophilum, the cause of human granulocytic anaplasmosis, and has fairly good central nervous system penetration, which the other agents do not. Because EM may be similar in appearance to community-acquired cellulitis, it is best to use antibiotics--such as cefuroxime axetil and amoxicillin/clavulanic acid--that can cover both types of infection when it is difficult to make the distinction.12 See TABLE 2.
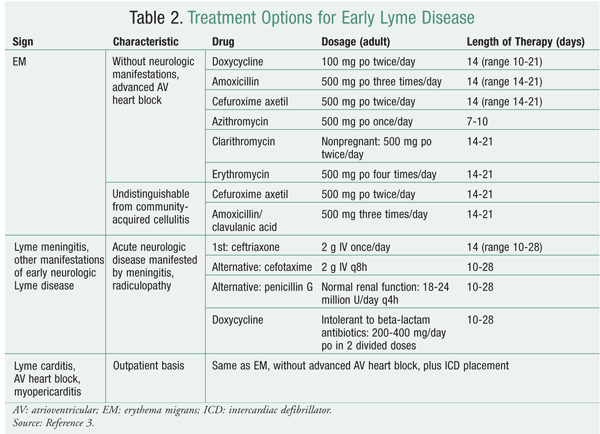
When neurologic manifestations such as meningitis or radiculopathy present, IV ceftriaxone may be used. Alternatives to ceftriaxone include IV cefuroxime and penicillin G. If the patient is allergic to beta-lactam antibiotics, doxycycline may be used as a substitute.3 See TABLE 2.
Patients with atrioventricular heart block and/or myopericarditis during early Lyme disease usually are treated for an average of 14 days. Hospitalized adults and children should receive IV ceftriaxone as an initial recommendation, in the same regimen as for Lyme meningitis. A patient who presents with advanced atrioventricular heart block may receive a temporary pacemaker on the advice of a cardiologist. For outpatients, the regimen is the same as for EM without carditis.3
Late-stage Lyme disease involves infection that persists months to years after the initial infection. Symptoms include arthritis, neurologic disease, and acrodermatitis chronica atrophicans (ACA; a dermatologic condition that leads to skin atrophy).13 Treatment, as per IDSA guidelines, is similar to that for early Lyme disease, with the difference that the length of therapy is longer for late-stage disease. The manifestations of arthritis and ACA are treated with doxycycline, amoxicillin, or cefuroxime axetil, as for early-stage EM.3 The length of therapy is 28 days for arthritis and 21 days for ACA, whereas the average length for EM is 14 days.3 Therapy for late-stage neurologic disease consists of ceftriaxone, cefotaxime, or penicillin G for 14 to 28 days, which is similar to the treatment for meningitis, which can occur during early-stage Lyme disease.3
Adverse Effects
Doxycycline: Some common gastrointestinal (GI) disturbances are dysphagia, anorexia, diarrhea, pseudomembranous colitis, and vomiting.14 Dermatologic manifestations may include photosensitivity, rash, and skin hyperpigmentation. Some rare adverse events are esophageal ulceration, esophagitis, exfoliative dermatitis, and hepatotoxicity. Because doxycycline may cause fetal harm (pregnancy category D), it is preferable not to use it during pregnancy.
Amoxicillin, Amoxicillin/Clavulanic Acid, Penicillin G: Nausea, vomiting, diarrhea, and pseudomembranous colitis are common GI side effects.14 A rash may develop, including life-threatening types such as Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), which should be reported to a physician immediately.
Cefuroxime Axetil: GI disturbances such as nausea, vomiting, and diarrhea may occur.14 More uncommon adverse effects include pseudomembranous colitis, SJS, TEN, and stomach cramps.
Ceftriaxone: Since ceftriaxone is a parenteral medication that may be given IV in the setting of Lyme disease, injection-site pain and tenderness may occur.14 Rash and diarrhea may also occur. Rare adverse events are similar to those for cefuroxime axetil: pseudomembranous colitis, SJS, and TEN.
Macrolides (Azithromycin, Clarithromycin, Erythromycin): Nausea, vomiting, diarrhea, and rash may occur.14 Rarely, a patient may have SJS, TEN, hearing loss, or pseudomembranous colitis, or may experience arrhythmias due to QTc prolongation. Azithromycin and erythromycin are pregnancy category B; clarithromycin is category C because some studies have documented adverse fetal effects. As a result, ISDA guidelines recommend the use of clarithromycin in nonpregnant patients only.3
Duration of Therapy
The choice of antimicrobial agent for treating Lyme disease has been well studied, but the duration of treatment continues to be subject to scrutiny.3,15 Current IDSA guidelines recommend 10 to 14 days for early Lyme disease. A recent retrospective study found that results were similar in patients receiving less than 10 days of therapy versus those receiving longer therapy.15 This supports prior studies, which concluded that treatment for 10 days was as effective as longer treatment regimens. Another point of interest from this recent study is the data regarding overprescription by physicians. At the researchers' institution, fewer than 16% of patients received less than 10 days of therapy, while 44% received more than 16 days. Often, additional treatment leads to more adverse effects. Therefore, if patients continue to have constitutional symptoms, they should be reevaluated for other conditions discussed in the following section.
Monitoring Response
Treatment failure is rare and frequently is mislabeled for a number of reasons.3,16 Most patients have a full recovery after taking the appropriate antibiotics, but a small number of patients continue to experience constitutional symptoms such as headache, fatigue, and muscle aches. Although this state is referred to as Post-Lyme disease syndrome or chronic Lyme disease, it is not caused by persistent infection with B burgdorferi, nor is it fully understood. To date, there have been no reports of resistant B burgdorferi.
When a patient continues to complain of symptoms after completing a course of appropriate antibiotics, a number of factors should be investigated. The patient may not have been compliant with therapy; the initial diagnosis of Lyme disease may have been incorrect; reinfection with a new tick or co-infection with another organism transmitted by ticks may have occurred; or another condition, like fibromyalgia or chronic fatigue syndrome, may be present. Although there is no benefit from additional antibiotics after initial treatment, prescribers continue to represcribe antibiotics without thorough reevaluation. As research continues, awareness of the ineffectiveness of retreatment likely will spread.
Conclusion
It is imperative to have a good understanding of Lyme disease, the most common tickborne disease in the U.S. The number of reported cases has increased by 101% from 1996 (9,908 cases) to 2002 (19,931 cases).17 Even more alarmingly, the number continues to rise, with 28,921 confirmed cases of Lyme disease in 2008.18 Patients should be educated about preventive measures they can take when traveling to certain areas, and they should be counseled on specific medications to enhance their compliance and clear the infection. In April 2010, the IDSA reevaluated its 2006 guidelines and upheld its recommendations.
REFERENCES
1. Bratton RL, Whiteside JW, Hovan MJ, et al. Diagnosis and treatment of Lyme disease. Mayo Clin Proc. 2008;83:566-571.
2. CDC. Learn about Lyme disease.
www.cdc.gov/ncidod/dvbid/lyme. Accessed April 17, 2010.
3. Wormser GP, Dattwyler RJ, Shapiro ED, et al. The clinical assessment, treatment, and prevention of Lyme disease, human granulocytic anaplasmosis, and babesiosis: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2006;43:1089-1134.
4. National Institute of Allergy and Infectious Diseases. Lyme disease. Vaccine production.
www.niaid.nih.gov/topics/
5. Steere AC, Klitz W, Drouin EE, et al. Antibiotic-refractory Lyme arthritis is associated with HLA-DR molecules that bind a Borrelia burgdorferi peptide. J Exp Med. 2006;203:961-971.
6. Marques AR. Lyme disease: a review. Curr Allergy Asthma Rep. 2010;10:13-20.
7. Johnson SE, Klein GC, Schmid GP, Feeley JC. Susceptibility of the Lyme disease spirochete to seven antimicrobial agents. Yale J Biol Med. 1984;57:549-553.
8. Agger WA, Callister SM, Jobe DA. In vitro susceptibilities of Borrelia burgdorferi to five oral cephalosporins and ceftriaxone. Antimicrob Agents Chemother. 1992;36:1788-1790.
9. Luft BJ, Dattwyler RJ, Johnson RC, et al. Azithromycin compared with amoxicillin in the treatment of erythema migrans. A double-blind, randomized, controlled trial. Ann Intern Med. 1996;124:785-791.
10. Terekhova D, Sartakova ML, Wormser GP, et al. Erythromycin resistance in Borrelia burgdorferi. Antimicrob Agents Chemother. 2002;46:3637-3640.
11. Steere AC, Hutchinson GJ, Rahn DW, et al. Treatment of the early manifestations of Lyme disease. Ann Intern Med. 1983;99:22-26.
12. Eppes SC, Childs JA. Comparative study of cefuroxime axetil versus amoxicillin in children with early Lyme disease. Pediatrics. 2002;109:1173-1177.
13. Feder H, Johnson B, O'Connell S, et al. A critical appraisal of “chronic Lyme disease.” N Engl J Med. 2007;357:1422-1430.
14. Lexi-Comp Online [subscription required]. Hudson, OH: Lexi-Comp, Inc; 2010.
15. Kowalski TJ, Tata S, Berth W, et al. Antibiotic treatment duration and long-term outcomes of patients with early Lyme disease from a Lyme disease-hyperendemic area. Clin Infect Dis. 2010;50:512-520.
16. Dattwyler RJ. A commentary on the treatment of early Lyme disease. Clin Infect Dis.
17. Bacon RM, Kugeler KJ, Mead PS. Surveillance for Lyme disease--United States, 1992-2006. MMWR Surveill Summ. 2008;57:1-9.
18. CDC. Reported cases of Lyme disease by year, United States, 1994-2008.
www.cdc.gov/ncidod/dvbid/lyme/ 2010;50:521-522.
To comment on this article, contact rdavidson@uspharmacist.com.





