US Pharm. 2013;38(3):Epub.
ABSTRACT: Cardiac arrest is a leading cause of mortality and morbidity in the United States, the majority secondary to neurologic injury. Lowering the body temperature post–cardiac arrest has been shown to improve morbidity and mortality, and it has become the gold standard of care. Methods to induce and maintain therapeutic hypothermia include invasive and noninvasive techniques. Adverse effects during therapeutic hypothermia are shivering, fluid and electrolyte shifts, bradycardia, and infection. The pharmacokinetics and pharmacodynamics of multiple medications are altered during hypothermia. Pharmacists can help play a role in adverse-effect prevention and medication management.
Every year in the United States, an estimated 300,000 patients experience an out-of-hospital cardiac arrest.1 Ultimately, only 7.9% of those patients survive to discharge.1 The rate of survival of an in-patient cardiac arrest is also low at approximately 33%.1 Upwards of 90% of survivors experience permanent neurologic injury.2
The utilization of therapeutic hypothermia to improve patient outcomes after cardiac arrest dates back over 200 years to Russia, when patients were covered in snow to attempt to return circulation.3,4 From the 1800s to the 1960s, therapeutic hypothermia was investigated for multiple indications ranging from cancer prevention to decreasing intracranial pressure during cardiac surgery. Due to complications, the use of hypothermia was abandoned until the 1980s. Starting with successful animal trials followed by human clinical trials led to our current improved post-resuscitative efforts.5
Post–Cardiac Arrest Brain Injury
During cardiac arrest, there is a total lack of perfusion to the brain, which initiates a complex cascade of events. A key aspect of these events is that they are all temperature dependent. There are two main components of post–cardiac arrest brain injury: ischemia and reperfusion. Due to the lack of blood flow during cardiac arrest, there is a loss of adenosine triphosphate (ATP) production in the brain causing the release of glutamate, which is responsible for neuronal injury. An influx of intracellular calcium causes an increase of cell permeability that contributes to brain cell death. After return of spontaneous circulation, reperfusion injury can occur from free radicals and mitochondrial injury.6,7
There are multiple proposed mechanisms by which therapeutic hypothermia protects against neurologic injury. Hypothermia decreases neuronal metabolism, glucose and oxygen consumption, glutamate release, and blood-brain barrier breakdown. Overall, the neurologic cell death is decreased with hypothermia.8
Supporting Clinical Data
In 2002, the first two randomized, controlled trials investigating use of therapeutic hypothermia after out-of-hospital cardiac arrest were simultaneously released.9,10 These two trials were landmark studies that have revolutionized post–cardiac arrest care.
Bernard et al conducted a trial in Australia to
investigate the impact of therapeutic hypothermia on neurologic outcomes after cardiac arrest due to ventricular arrhythmias.9
There were 77 patients included in the study who were randomized to
hypothermia (33°C for 12 hours) or normothermia. In the hypothermia
group, 49% survived with a good neurologic outcome (discharged home or
to rehabilitation facility), while only 26% in the normothermia group
survived with good neurologic outcome (P = .046). There was also a decrease in mortality in the hypothermia group; however, it was not statistically significant.9
The Hypothermia After Cardiac Arrest Study Group conducted a similar trial in Europe.10 The 275 patients included in the study were randomized to hypothermia (32°C-34°C for 24 hours) or normothermia. In the hypothermia group, 55% survived with good neurologic outcome at 6 months, while only 39% in the normothermia group had a good neurologic outcome at 6 months (P = .009). There was a statistically significant (P = .02) decrease in mortality in the hypothermia group (41%) compared to the normothermia group (55%).10
Based on the clinical data, the Advanced Life Support Task Force of the International Liaison Committee on Resuscitation (ILCOR) and the American Heart Association (AHA) both strongly recommend mild therapeutic hypothermia after cardiac arrest for out-of-hospital ventricular cardiac arrest.11-13 There are limited data supporting the use of cardiac arrest in nonshockable rhythms. A recent cohort review of 437 patients with pulseless electrical activity or asystole showed no benefit with the use of therapeutic hypothermia.14 A possible benefit for therapeutic hypothermia has been seen with in-patient cardiac arrest (TABLE 1).13

Temperature Management
There are several methods that may be utilized to induce and maintain hypothermia. They can be divided into two categories: noninvasive and invasive. Noninvasive methods include exposure of skin to cold water, fans, ice packs, and gel-circulating cooling blankets or pads (FIGURE 1). Invasive methods include infusion of ice-cold fluids, intravascular catheters, and extracorporeal circulation. Each method has its own advantages and disadvantages.15 A combination of methods may be a more effective tactic to achieve goal temperature.16
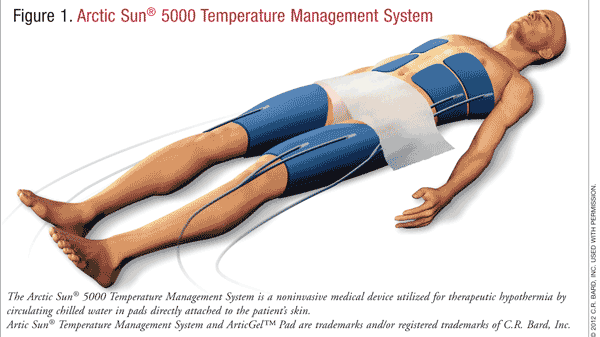
Temperature management during therapeutic hypothermia can be divided into three phases: induction, maintenance, and rewarming. In the induction phase, the goal of treatment is to decrease the patient’s temperature below 34°C as quickly as possible. During this phase, the patient is the most unstable due to hypovolemia, electrolyte abnormalities, and hemodynamic disturbances. This instability can be minimized with a rapid induction to goal temperature. During the maintenance phase, the patient becomes more stable, and the focus should be on prevention of long-term complications. The rewarming phase can be problematic, with electrolyte abnormalities, hypoxia, and cardiovascular instability. However, these effects may be mitigated with a slow, controlled warming (TABLE 2).15

Management of Adverse Effects
Shivering is the most common side effect during the induction phase. Shivering is an involuntary muscle response that increases heat production to restore normal body temperature. The response typically begins at 35.5°C and diminishes at 33.5°C. This reaction can lead to increased metabolism and oxygen consumption and a stress-like response. Shivering can be negated in several different ways. Surface counter warming, which is the warming of the face, hands, and feet, may be utilized to decrease the sensation of cold. Multiple pharmacologic agents have been used to abate shivering; however, no one agent has been shown to be superior. The most commonly used medications are opioids, sedatives, anesthetics, magnesium, and neuromuscular blockers. Paralyzing agents are the most effective agents for shivering cessation; however, their use should be minimized due to adverse effects.15,17
Fluid and electrolyte disturbances are common during hypothermia, prominently during the induction phase. Hypovolemia caused by hypothermia diuresis typically requires volume resuscitation. Hypokalemia frequently occurs due to cold diuresis and intracellular shifting. Potassium should be aggressively replaced during the cooling and maintenance phase. Due to the possibility of electrolyte shifts during the rewarming phase, conservative potassium replacement strategies should be employed.18 Hyperglycemia is also observed during the cooling and maintenance phase, as there is a decrease in insulin secretion and sensitivity. Use of an insulin drip may be required to maintain an appropriate blood glucose range.19
Bradycardia is observed in most patients undergoing therapeutic hypothermia, which decreases cardiac output. Heart rates can drop as low as 40 beats per minute (bpm); however, this typically does not require treatment. Arrhythmias are uncommon, unless the temperature drops below 30°C.15
Rarely, therapeutic hypothermia can lead to coagulation abnormalities.15 Patients who are actively bleeding may require less aggressive hypothermia, as clotting factors are not affected until the temperature is less than 35°C.20
Patients’ inflammatory response is suppressed during hypothermia. A retrospective review of 537 patients undergoing therapeutic hypothermia showed frequent infections (67%).21 The most frequent was pneumonia, followed by bacteremia and line infections. Despite an increase in infection rates, there was not an increase in mortality or favorable neurologic outcomes.21 Empiric broad-spectrum antibiotics may be considered.15
Medication Management
Therapeutic hypothermia can lead to alterations in the pharmacokinetics and pharmacodynamics of multiple medications.22 For example, there is decreased receptor response to morphine during hypothermia. During the cooling phase, the morphine dosage may need to be escalated to achieve adequate effect. However, during the rewarming phase, if the morphine dosage is not titrated downward, toxicity may occur.23 There is a lack of clinical studies describing the clinical effects of hypothermia on medications. Pharmacists should be aware of potential effects and monitor for ineffectiveness and toxicity.
Additional Indications
Based on the success of therapeutic hypothermia in cardiac arrest, its use has expanded to multiple other indications. Hypothermia has shown a reduction of morbidity and mortality in perinatal asphyxia. Several studies have evaluated the effect of hypothermia on traumatic brain injury. While no benefit in mortality was observed, hypothermia was able to decrease intracranial pressure. Therapeutic hypothermia has also been investigated to treat ischemic and hemorrhagic stroke, spinal cord injury, and hepatic encephalopathy; however, currently the data do not support or refute its use.24
Conclusion
Therapeutic hypothermia is becoming the standard of care for post–cardiac arrest care. Understanding temperature management, adverse effects from hypothermia, and potential medication alterations are key to pharmacist participation. Pharmacists can play a vital role in managing adverse effects and preventing medication errors.
REFERENCES
1. Roger VL, Go AS, Lloyd-Jones DM, et al; on behalf of
the American Heart Association Statistics Committee and Stroke
Statistics Subcommittee. Heart disease and stroke statistics—2011
update: a report from the American Heart Association. Circulation. 2011;123:e18–e209.
2. Neumar RW, Nolan JP, Adrie C, et al. Post-cardiac
arrest syndrome: epidemiology, pathophysiology, treatment, and
prognostication: a consensus statement from the International Liaison
Committee on Resuscitation (American Heart Association, Australian and
New Zealand Council on Resuscitation, European Resuscitation Council,
Heart and Stroke Foundation of Canada, InterAmerican Heart Foundation,
Resuscitation Council of Asia, and the Resuscitation Council of Southern
Africa); the American Heart Association Emergency Cardiovascular Care
Committee; the Council on Cardiovascular Surgery and Anesthesia; the
Council on Cardiopulmonary, Perioperative, and Critical Care; the
Council on Clinical Cardiology; and the Stroke Council. Circulation. 2008;118:2452-2483.
3. Varon J, Sternbach GL. Cardiopulmonary resuscitation: lessons from the past. J Emerg Med. 1991;9:503-507.
4. Liss HP. A history of resuscitation. Ann Emerg Med. 1986;15:65-72.
5. Marion DW, Leonov Y, Ginsberg M, et al. Resuscitative hypothermia. Crit Care Med. 1996;24(suppl 2):s81-s89.
6. Vaagenes P, Ginsberg M, Ebmeyer U, et al. Cerebral resuscitation from cardiac arrest: pathophysiology mechanisms. Crit Care Med. 1996;24:s57-s68.
7. Geocadin RG, Koenig MA, Jia X, et al. Management of brain injury after resuscitation from cardiac arrest. Neurol Clin. 2008;26:487-506.
8. Colbourne F, Sutherland G, Corbett D. Postischemic
hypothermia: a critical appraisal with implications for clinical
treatment. Mol Neurobiol. 1997;14:171-201.
9. Bernard SA, Gray TW, Buist MD, et al. Treatment of
comatose survivors of out-of-hospital cardiac arrest with induced
hypothermia. N Engl J Med. 2002;346:557-563.
10. Hypothermia After Cardiac Arrest Study Group. Mild
therapeutic hypothermia to improve the neurological outcome after
cardiac arrest. N Engl J Med. 2002;346:549-556.
11. Nolan JP, Morley PT, Vanden Hoek TL, et al.
Therapeutic hypothermia after cardiac arrest: an advisory statement by
the Advanced Life Support Task Force of the International Liaison
Committee on Resuscitation. Circulation. 2003;108:118-121.
12. American Heart Association. 2005 American Heart
Association guidelines for cardiopulmonary resuscitation and emergency
cardiovascular care. Circulation. 2005;112(suppl):1-211.
13. Peberdy MA, Callaway CW, Neumar RW, et al. Part 9:
post-cardiac arrest care: 2010 American Heart Association Guidelines for
Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2010;122(suppl 3):S768-S786.
14. Dumas F, Grimald D, Zuber B, et al. Is hypothermia
after cardiac arrest effective in both shockable and nonshockable
patients?: Insights from a large registry. Circulation. 2011;123:877-886.
15. Polderman KH, Herold I. Therapeutic hypothermia and
controlled normothermia in the intensive care unit: practical
considerations, side effects, and cooling methods. Crit Care Med. 2009;37:1101-1120.
16. Polderman KH, Rijnsburger ER, Peerdeman SM, et al.
Induction of hypothermia in patients with various types of neurologic
injury with use of large volumes of ice-cold intravenous fluid. Crit Care Med. 2005;33:2744-2751.
17. Presciutti M, Bader MK, Hepburn M. Shivering management during therapeutic temperature modulation: nurses’ perspective. Critical Care Nurse. 2012;32:33-42.
18. Bernard SA. Therapeutic hypothermia after cardiac arrest. Neurol Clin. 2006;24:61-71.
19. Lee R, Asare K. Therapeutic hypothermia for out-of-hospital cardiac arrest. Am J Health Syst Pharm. 2010;67:1229-1237.
20. Tokutomi T, Miyagi T, Morimoto K, et al. Effect of
hypothermia on serum electrolyte, inflammation, coagulation, ad
nutritional parameters in patients with severe traumatic brain injury. Neurocrit Care. 2004;1:171-182.
21. Mongardon N, Perbet S, Lemiale V, et al. Infectious
complications in out-of-hospital cardiac arrest patients in the
therapeutic hypothermia era. Crit Care Med. 2011;39:1359-1394.
22. Zhou J, Poloyac SM. The effect of therapeutic
hypothermia on drug metabolism and drug response: cellular mechanisms to
organ function. Expert Opin Drug Metab Toxicol. 2011;7:803-816.
23. Puig MM, Warner W, Tang CK, et al. Effects of temperature on the interaction of morphine with opioid receptors. Br J Anesth. 1987;59:1459-1464.
24. Rivera-Lara L, Zhang J, Muehlschlegel S. Therapeutic hypothermia for acute neurological injuries. Neurotherapeutics. 2012;9:73-86.
To comment on this article, contact rdavidson@uspharmacist.com.





