US Pharm. 2018;43(8):12-16.
ABSTRACT: Androgenetic alopecia (AGA), or male pattern baldness, is the most common cause of hair loss in men. There are several therapies available for the treatment of this condition, with 5-alpha reductase inhibitors and minoxidil most commonly used. Other current treatment options include laser therapy, scalp microneedling, hair mesotherapy, and hair transplantation. The development of new pharmacologic therapies has been slow; however, research is currently being conducted using Janus kinase inhibitors and autologous platelet-rich plasma injections in men with AGA.
Androgenetic alopecia (AGA), or male pattern baldness, is a common condition that causes many men to seek medical attention. It is characterized by hair follicles that gradually decrease in size and lead to baldness over time. AGA affects approximately 80% of Caucasian males. Although it is common in men of different ethnic backgrounds, Caucasian males are affected more frequently.1 Traditionally, pharmacologic treatment of AGA targets decreasing dihydrotestosterone (DHT) and stimulating hair follicles through the use of 5-alpha reductase (5AR) inhibitors or minoxidil; however, new and experimental therapies are exploring inhibition of Janus kinase (JAK) and the use of platelet-rich plasma (PRP). Other therapies include laser therapy, scalp microneedling, hair mesotherapy, and hair transplantation.1
Pathophysiology
AGA is the result of genetic predisposition; however, evidence shows that the cause is androgen related. DHT is the primary androgen involved in AGA, with DHT concentrations being higher in men with AGA.2 Circulating testosterone is converted to DHT by 5AR.
There are three types of alpha reductase receptor isoenzymes, and type I and type II are significant in the treatment of AGA. Type I is located in the skin, including the sebaceous glands and hair follicles, and type II is located in the inner root sheath of hair follicles in the scalp, face, chest, genitals, and prostate gland.3 The hair-growth cycle is also affected in AGA. Hair in the telogen, or dormant, phase is more predominant than hair in the anagen, or growth, stage. This leads to a decrease in hair on certain areas of the head. In addition, some of the hair follicles are smaller and more sensitive to androgen. The pattern in which the hair loss occurs is genetically predetermined by the distribution of these smaller hair follicles.2
5AR Inhibitors
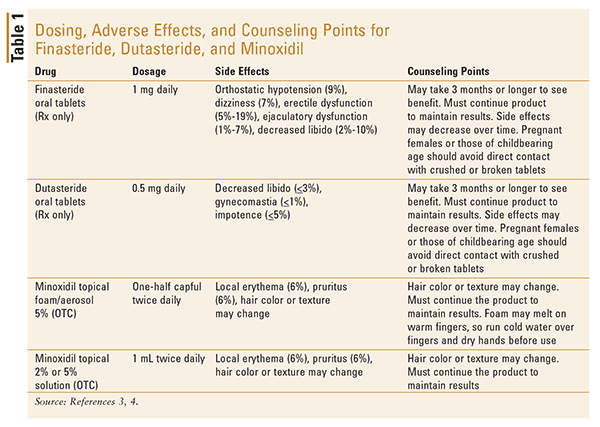
Finasteride is a 5AR inhibitor that is approved by the FDA for androgenetic alopecia. The drug works by inhibiting the conversion of testosterone to DHT by blocking type II 5AR. Dosing, side effects, and counseling points are found in TABLE 1.4 Due to unsatisfactory side effects with oral finasteride, studies have been conducted comparing finasteride 0.25% and 0.5% topical solution with oral finasteride 1 mg. The studies were small, but they evidenced a similar improvement in hair growth with fewer side effects compared with the topical-therapy groups.5 (The topical formulation is not currently available in the United States.)
Dutasteride is also used off-label for treatment of AGA and is an inhibitor of both type I and type II 5AR. In addition, dutasteride is a more potent inhibitor of both of these isoenzymes compared with finasteride.5 A multicenter, randomized, double-blinded, double-dummy, parallel-group study was conducted over 29 weeks in 917 men aged 20 to 50 years with AGA. The men were randomized to dutasteride 0.02, 0.1, or 0.5 mg/day, finasteride 1 mg/day, or matched placebo. The primary endpoint was a change from baseline in hair count within a 2.54-cm–diameter area at the scalp vertex. Dutasteride 0.5 mg was found to be superior to finasteride 1 mg at Weeks 12 and 24 in increasing hair count (both, P = .003). Secondary endpoints included hair growth and hair restoration. Dutasteride 0.5 mg significantly improved hair count and hair width when compared with finasteride at Week 24 (P = .016 and P = .004, respectively).
The tolerability of these agents was similar. This study indicates that dutasteride is an alternative to finasteride in the treatment of AGA, as it appears to offer improved results when compared with finasteride in the short term.6 Long-term trials are needed to confirm these results. TABLE 1 lists the side effects, dosing, and counseling points for dutasteride.3,4
Dutasteride has also been used in hair mesotherapy, the process of injecting small amounts of compounds into the mesoderm of the scalp just below the skin’s surface. Hair mesotherapy using dutasteride in combination with some vitamins has been studied on a limited basis. Mesotherapy is used to prevent systemic side effects and to increase local circulation of the pharmaceutical agents or vitamins. The studies are limited and the outcomes are variable.7
Minoxidil
Topical minoxidil was the first drug approved by the FDA for treatment of AGA. It is available in 2% and 5% solution and 5% foam, and the 5% solution has been shown to be more effective than the 2% solution.5,8 There are several mechanisms by which minoxidil may promote hair growth; however, the exact mechanism of action is unclear. It has been shown both in vivo and in vitro to have a direct mitogenic effect on epidermal cells, and in vitro it prolongs the survival time of keratinocytes. In addition, topical minoxidil may oppose calcium entry into the cells, which may increase epidermal growth factors to allow hair growth.8 In approximately 50% of men, hair loss is stopped, and a small percentage of men experience some hair regrowth.5 The drug must be continued indefinitely or hair growth will subside. Dosing, side effects, and counseling points are included in TABLE 1.4
Ketaconazole, Spironolactone, and Prostaglandin F2 Analogues
Ketaconazole shampoo has been considered as an agent to aid in the treatment of AGA as combination therapy with oral finasteride.1 The mechanism is unclear; however, ketaconazole inhibits the DHT pathway, conferring antiandrogenic properties. In addition, it may reduce inflammation in the skin. Clinical trials need to be conducted in order to validate its effectiveness.1,5
Spironolactone is a potassium-sparing diuretic that has antiandrogenic properties; it blocks androgen receptors and decreases testosterone levels. Most of the clinical data using 50 to 200 mg/day of spironolactone have been in women with AGA, and the results have been variable.1
Patients using the prostaglandin F2 analogue latanoprost to treat glaucoma experienced the side effect of elongation of eyelashes and eyebrows; as a result, a randomized, double-blind, placebo-controlled trial was conducted in 16 men using lantanoprost 0.1% daily versus placebo to evaluate scalp hair growth. The 24-week study resulted in a significant increase in hair density from baseline when compared with placebo. Different concentrations of bimatoprost, another prostaglandin F2 analogue, are currently being studied in men with AGA.1 Although these results appear positive, the study populations are too small to extrapolate results. Additional studies need to be conducted in a larger patient population for a longer period of time to determine efficacy of the therapy.
Low-level Laser Therapy
Low-level laser therapy has also been used for the treatment of AGA. It appears that lasers can stimulate hair growth at certain wavelengths; however, the mechanism for this action has not been determined. There are several laser therapy devices, including a comb, a hood, and a helmet that fits on the head. Among these, the HairMax LaserComb is the only device that has FDA clearance.5 A meta-analysis was conducted to assess the effectiveness of nonsurgical treatments for AGA. Low-level laser light therapy showed a mean difference in hair count of 17.66 hairs/cm2 when compared with placebo (P <.00001).9 This therapy may be used in conjunction with drug therapy to improve results. More information is needed to determine the effectiveness and best place in therapy.
Microneedling and Hair Transplantation
Microneedling has gained popularity and has been found to be beneficial in stimulating hair regrowth in alopecia. It was previously used for cosmetic purposes and is now used to improve topical drug delivery. Microneedling uses multiple fine needles, generally attached to a roller, to create tiny punctures in the skin that stimulate neovascularization, release growth factors, and promote the expression of Wnt proteins. Hair growth is a result of the release of certain growth factors and activation of the hair bulge, and Wnt proteins have been found to stimulate dermal papillae stem cells, leading to hair growth. Microneedling is generally used in conjunction with topical therapy such as minoxidil and it has been used with PRP. Studies assessing the efficacy of microneedling in conjunction with topical minoxidil are small, but some have reported statistically significant results.
A 12-week study was conducted to compare weekly microneedling in conjunction with minoxidil 5% solution with minoxidil 5% solution alone. Minoxidil was applied twice daily in both cohorts of 50 men each. In the microneedling group, minoxidil was applied 24 hours after the microneedling session since the session produced mild erythema in the area in which it was performed. One of the study endpoints was hair count in a predefined, 1 cm–diameter area at the end of 12 weeks. Patients in the treatment group showed a hair count of 91.4 hairs per cm2 compared with 22.2 hairs per cm2 in the control group (P = .039). Patients reported no adverse effects, and results were sustained for 8 months following treatment.
Results from other studies are varied; however, it appears that microneedling may increase penetration of topical drugs into the scalp, thereby promoting effectiveness.10 This may be something for providers to consider in those patients in whom traditional topical therapy has been ineffective.
Hair transplantation is a commonly used treatment option for AGA. Effectiveness of the treatment varies due to differences in techniques and ability of the surgeon. In hair transplantation, hair follicles are taken from areas of the scalp that are resistant to androgen and transplanted into areas that are androgen dependent. Results are generally evident within 6 to 8 months after the transplant, and the use of pharmacologic agents such as minoxidil may be required to prevent progression of the condition.1
Emerging Therapies
Several new therapies are emerging or being studied as treatment options for AGA, including JAKs and PRP injections. JAKs are under investigation as new drugs to treat alopecia; however, there is limited information about their use in AGA.1 One of these products has been granted Fast Track approval by the FDA for the treatment of moderate-to-severe alopecia areata (autoimmune-related hair loss). The class of drugs was found to proliferate hair regrowth in cases of alopecia universalis (hair loss over the entire body) and, topically, the class promoted hair growth in mice and follicle growth in humans.1,11 It is important to note that alopecia areata and alopecia universalis appear to be autoimmune conditions and are not androgen related. Although there are differences in etiology, companies are now investigating both topical and oral formulations of this drug class to treat AGA.11
Injecting autologous PRP into the scalp is a new therapy for the treatment of AGA. The platelets are gathered through a blood draw and then separated, concentrated, and prepared through several methods utilizing commercially available kits. Platelets release growth factors that are involved in the stimulation of endothelial and stem cells.
A systematic review evaluated 14 studies that used PRP. The studies used between 1.5 and 12 mL of PRP per treatment, treatments were given at intervals of every 2 weeks to 3 months, and the number of injection sessions ranged from one to five. The studies assessed different endpoints, including hair thickness, hair loss, and hair density. Of the 14 studies, seven showed a significant increase in the number of hairs, ranging from 12.3 to 45.9 hairs/cm2. The studies showed more variability in hair loss and hair thickness endpoint results. Overall, there were few adverse effects, with localized irritation and pain the main side effects reported. PRP injections must be continued in order to maintain the benefits. Although the studies are favorable, larger, longer-term trials are needed to determine the efficacy of this therapy.12
Conclusion
Over the past 30 years, there has been very little advancement in the treatment of AGA; however, with the investigation of JAK inhibitors, that may change in the future. As clinicians consider options for treating this condition, pharmacologic products such as finasteride, minoxidil, and dutasteride, which have been widely studied and have provided positive outcomes, are available and should be considered as first-line therapies. In addition, PRP and microneedling are less common options that provide promising outcomes.
Healthcare providers should discuss options with patients and prescribe therapy based on actual outcomes in conjunction with patient preference while taking into consideration dosing schedules and cost.
REFERENCES
1. Kelly Y, Blanco A, Tosti A. Androgenetic alopecia: an update of treatment options. Drugs. 2016;76:1349-1364.
2. Hogan DJ, Chamberlain M. Male pattern baldness. South Med J. 2000;93(7):657-662.
3. Hirshburg JM, Kelsey PA, Therrien CA, et al. Adverse effects and safety of 5-alpha reductase inhibitors (finasteride, dutasteride): a systematic review. J Clin Aesth Dermatology. 2016;9(7):56-62.
4. Lexicomp mobile. Wolters Kluwer Clinical Drug Information, Inc. 2018, version 4.0.4.
5. Rossi A, Anzalone A, Fortuna MC, et al. Multi-therapies in androgenetic alopecia: review and clinical experiences. Dermatol Ther. 2016;29:424-432.
6. Gubelin-Harcha WG, Martinez JB, Tsen-Fang T, et al. A randomized, active and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia. J Am Acad Dermatol. 2014;70(3):489-498.
7. Busanello EB, Turcatel E. Androgenic alopecia and dutasteride in hair mesotherapy: a short review. Our Dermatol Online. 2017; 9(1):75-79.
8. Bolduc C, Shapiro J. Management of androgenetic alopecia. Am J Clin Dermatol. 2000;1(3):151-158.
9. Adil A, Godwin M. The effectiveness of treatments for androgenetic alopecia: a systematic review and meta-analysis. J Am Acad Dermatol. 2017;77(1):136-141.
10. Fertig RM, Gamret AC, Tosti JC. Microneedling for the treatment of hair loss? JEADV. 2018;32:564-569.
11. Belgraviacentre.com. Oral alopecia areata JAK inhibitor treatment fast-tracked by FDA. www.belgraviacentre.com/blog/oral-alopecia-areata-jak-inhibitor-treatment-fast-tracked-by-fda. Accessed April 11, 2018.
12. Picard F, Hersant JN, Meningaud JP. Injections of platelet-rich plasma for androgenic alopecia: a systematic review. J Stomatol Oral Maxillofac Surg. 2017;118:291-297.
To comment on this article, contact rdavidson@uspharmacist.com.





