US Pharm. 2008;33(12):30-37.
Ulcerative colitis (UC) is a chronic inflammatory bowel disorder (IBD) characterized by diffuse mucosal inflammation of the colon. It affects between 250,000 and 500,000 Americans.1 Unlike Crohn's disease, which may affect the entire gastrointestinal (GI) tract, UC is limited to the colon. UC almost always involves the rectum, but may extend proximally in a contiguous fashion to involve portions of, or the entire, colon (TABLE 1). In the majority of new cases diagnosed, disease is limited to the distal or left side of the colon, and there is some evidence that aggressive pharmacotherapy early in the disease may prevent progression and extension of inflammation.2 This article will review the initial treatment options for UC and their place in therapy for both acute attacks and maintenance of remission.

Overview of UC
The hallmark symptom of UC is bloody diarrhea. Other symptoms patients may commonly experience include abdominal pain, rectal urgency, and tenesmus. Severity of symptoms will depend on the severity and extent of inflammation. The clinical course is marked by exacerbations and remissions, and about half of patients with UC will experience an exacerbation in any given year. About 15% of patients will have severe attacks involving the entire colon, and colectomy will be required in 30% of these patients.3 Patients with UC have an increased risk of cancer, depending mainly on both the duration and extent of disease. The American College of Gastroenterology recommends a colonoscopy eight to 10 years after diagnosis and then every one to two years thereafter.4
Onset of UC is usually between 15 and 40 years of age, with another peak in incidence after age 60. Men and women are equally affected.5 The etiology of UC is unknown, but is most likely the result of interactions between genetic, environmental, and microbial factors, as well as the intestinal immune system.
A complete history and examination should be done when UC is suspected. Recent antibiotic use may suggest pseudomembranous colitis; therefore, testing for Clostridium difficile (C. diff) toxin in the stool should always be done to rule out C. diff colitis. Lab tests such as C-reactive protein and erythrocyte sedimentation rate that suggest inflammation may be elevated. Abdominal x-rays are essential to detect major complications that can occur, such as colonic distention that can lead to toxic megacolon or intestinal perforation. They can also help to assess the extent of disease. Colonoscopy or proctosigmoidoscopy and biopsy are the gold standards for diagnosis of UC. Diffuse, continuous changes in the vascular pattern of the colonic mucosa as well as ulcerations and exudates are commonly seen.3,6
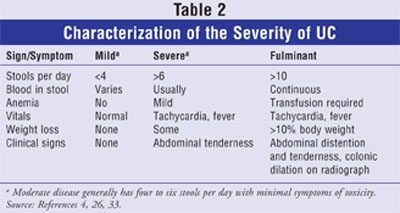
Once the diagnosis of UC is made, both the extent and severity of disease should be assessed in order to guide treatment (TABLE 2). The goals of treatment are directed at inducing and then maintaining remission of symptoms and inflammation in order to improve quality of life. About 65% of patients will achieve clinical remission with medical treatment, and 80% of patients who are compliant with treatment will maintain remission.7 Therefore, it is important to educate patients on the benefits of complying with treatment regimens.
Aminosalicylates
5-aminosalicylic acid (5-ASA), also known as mesalamine, is effective for both induction and maintenance of remission in patients with UC.8,9 Sulfasalazine, which is a sulfapyridine molecule linked to 5-ASA, was originally used to treat UC. It is associated with many intolerable and life-threatening adverse reactions (e.g., exanthema, fever, angioedema, liver damage). When studies found that the active portion of sulfasalazine was 5-ASA and that many of the adverse effects were a result of the sulfapyridine moiety, efforts became directed toward developing pure 5-ASA formulations, which have now become the mainstay of therapy.10,11
5-ASA exerts its therapeutic effects topically on the inflamed mucosa of the colon by interacting with damaged epithelium. The exact mechanism of action is unknown, but 5-ASA is a known scavenger of oxygen-free radicals, and it blocks production and chemotaxis of leukotrienes, in addition to many other actions aimed at modifying the immune response and inflammation.12 When given orally, 5-ASA is readily absorbed prior to reaching the colon. Since the efficacy is dependent on 5-ASA reaching its site of action, delivery systems designed specifically to release 5-ASA in the colon have been developed, including both rectal and oral preparations (TABLE 3).
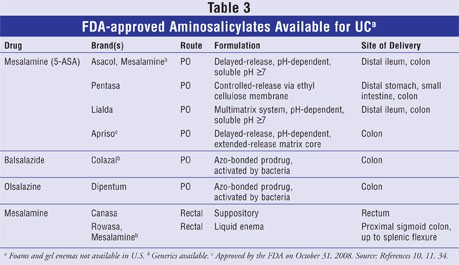
Rectal 5-ASA: For patients with distal UC (including proctosigmoiditis and proctitis), rectally administered 5-ASA is the preferred therapy for both mild to moderate active UC and maintenance of remission.13 Advantages include direct delivery of the drug to the site of action, reduction of systemic adverse effects, and improved efficacy.14 Suppositories deliver a large amount of 5-ASA that is confined to the rectum, while liquid enemas reliably reach as far as the splenic flexure.
Common complaints reported with rectally administered 5-ASA are related to patient discomfort and dislike of the drug instillation. These include abdominal bloating, discomfort during instillation, and difficulty retaining the drug.15 There is some evidence that gels or foam preparations of 5-ASA are better tolerated due to higher retention, but these products are not currently available in the U.S.16 A patient's willingness to comply and ability to retain a rectal treatment will ultimately determine the individual treatment regimen.
Randomized, controlled studies have demonstrated the superiority of rectally administered 5-ASA for active distal UC over placebo, rectally administered corticosteroids (CCSs), and oral 5-ASA.2 There also seems to be a faster onset of relief than with oral 5-ASA.17 No dose-response has consistently been documented with rectally administered 5-ASAs, and most data support between 1 and 4 g daily.2 Increasing evidence indicates that combination therapy with either oral 5-ASA and rectal 5-ASA or rectal 5-ASA and rectal CCSs may achieve better remission rates than monotherapy in patients with distal disease.18 For maintaining remission of distal UC, rectal 5-ASA is more effective than placebo and as effective as oral 5-ASA;14 however, rectal therapies are generally not accepted by patients and are also underprescribed by physicians.2
Oral 5-ASA: For patients unwilling or unable to tolerate rectal 5-ASA or for those with more extensive disease, orally administered 5-ASA is effective for mild to moderate active disease and is the mainstay of therapy for maintaining remission.3 Advantages include ease of use, wider patient acceptance, and enhanced compliance.
Different delivery systems are available that utilize physiochemical properties of the colon to release the active drug directly at the site of action. A novel multimatrix formulation of 5-ASA (MMX mesalamine [Lialda]) was recently approved; it has a pH-sensitive coating surrounding a matrix of hydrophilic polymer containing 5-ASA that is designed to slowly release the drug throughout the entire colon at relatively high concentrations.19 Similar to sulfasalazine, the azo-bonded prodrugs balsalazide and olsalazine utilize a carrier molecule, which is cleaved from the active drug by colonic bacteria.11 There does not appear to be a significant difference in efficacy between any of the currently available oral 5-ASA formulations.
A 2006 Cochrane meta-analysis reviewed the efficacy and safety of oral 5-ASA formulations compared to placebo or sulfasalazine for induction of remission in patients with active UC. The analysis included 21 studies and more than 2,000 patients. Oral 5-ASA was found to be superior to placebo, and there was a nonsignificant trend toward higher remission over sulfasalazine.8
Once remission has been achieved, it is very important to continue maintenance therapy in order to reduce the risk of relapse, proximal extension of disease, and the development of carcinoma.4 A Cochrane review of oral 5-ASA for maintenance of remission evaluated almost 2,500 patients and concluded that oral 5-ASA formulations are effective in maintaining remission of UC.9
Since there are not enough data to conclusively determine that one particular oral formulation of 5-ASA is better than another, the choice should be based on the location of active disease, cost, and administration regimens. For example, patients who forget to take multiple doses should be given once-daily formulations, and those with more distal colitis may benefit from azo-bonded formulations combined with rectal therapy.
The optimal dosing regimen for both induction and maintenance of remission is a controversial topic. Higher doses of oral 5-ASA (>=4 g/day) are more effective for inducing remission.3 There does not appear to be a dose-response relationship for maintenance therapy; thus, lower doses are recommended to minimize adverse effects (<2.4 g/day).20 There is evidence that switching between different oral 5-ASA formulations may improve response rates in individual patients, so it is worth trying an alternative before considering oral 5-ASA a treatment failure.21
Oral formulations of 5-ASA are generally well tolerated. Mild, reversible adverse effects include nausea, diarrhea, headache, and rash. More severe reactions include pancreatitis, hepatitis, blood dyscrasias, and worsening of colitis. Interstitial nephritis is a rare, idiosyncratic reaction that has been reported; however, there is some evidence that IBD itself may increase the risk of renal impairment. Patients with preexisting renal dysfunction or on high doses of oral 5-ASA should be periodically monitored for changes in renal function.10,11
Both rectal and oral 5-ASA formulations are considered safe in pregnancy (Pregnancy Category B). Therapy should be continued because active disease has more potential for maternal/fetal harm than the use of these drugs.22
Corticosteroids
Due to their potent anti-inflammatory effects, rectal, oral, and parenteral CCSs are all used for treatment of active UC, and selection depends on both the severity and extent of disease. CCSs have no role in maintenance of remission because of the many adverse effects from prolonged therapy (e.g., increased susceptibility to infections and osteoporosis).3,23 As with any drug therapy, the benefits of CCSs must be balanced by the risks.
Rectally administered CCSs (i.e., suppositories, foams, enemas) are less effective than rectal 5-ASA, but the combination is often more effective at achieving remission than either treatment alone in patients with active distal colitis. Patients using rectal CCSs on a regular basis may develop systemic adverse effects; therefore, prolonged therapy is not recommended.2 Budesonide is a potent CCS that has poor bioavailability and extensive first-pass metabolism. It acts topically on the colonic mucosa. When given orally, budesonide is released in the colon and provides therapeutic benefit with less systemic toxicity.23 Studies have shown conflicting results when oral budesonide was compared to conventional oral CCSs.2 The exact place of CCSs in therapy is unclear at this time.
Orally administered steroids, such as prednisone, are generally reserved for patients with moderate to severe active UC.23 They are also indicated for patients who have failed to respond to oral and rectal 5-ASA or rectal CCSs.3 About one-third of patients will require oral CCS therapy at some time during the course of their disease.24 The typical dose of oral prednisone is 40 to 60 mg daily, and once a response is achieved (usually 10-14 days), the dose should be tapered gradually over the next eight weeks.6
Intravenous CCSs are reserved for hospitalized patients with severe or fulminant-active UC and for those who have failed to respond to oral CCSs. There are no placebo-controlled trials with IV CCSs, but several observational studies have shown a response rate of about 50%.25 Patients will usually respond within seven to 10 days of IV treatment with the equivalent of methylprednisolone 60 mg daily. Transition to oral CCSs with plans to taper the dose should occur after the initial response.23
Long-term therapy with CCSs is not recommended secondary to significant side effects, but many patients will become dependent and experience an increase in disease activity when the dose is reduced. Despite this, even low doses of CCSs are not recommended for maintaining remission.26 Azathioprine and infliximab can be used to wean most patients off CCSs.
Other Medical Therapy
Azathioprine (AZA) and 6-mercaptopurine (6-MP) are immunomodulators, and their activity results from an unknown mechanism of their active metabolites, 6-thioguanine nucleotides. Neither is used to induce remission, as their onset of activity is about three months. AZA or 6-MP is recommended for maintaining remission in patients who frequently relapse while on adequate doses of 5-ASA or who are intolerant to 5-ASA treatment.3 They may also be used to lower or discontinue CCS therapy in patients with chronic, active UC who are dependent on CCSs.23
There have been conflicting data regarding the efficacy of AZA and 6-MP in maintaining remission of UC. A recent Cochrane review identified six randomized, controlled trials of at least one year's duration that compared AZA or 6-MP to placebo, sulfasalazine, or oral 5-ASA. AZA and 6-MP were superior to placebo in maintaining remission, but conclusions could not be drawn from the active comparator studies. The review concluded that in light of safer and more established maintenance therapy with 5-ASA, AZA or 6-MP could only be recommended in patients requiring repeated courses of CCSs.27
For long-term therapy, a large, multicenter study found that treatment with AZA or 6-MP reduced the incidence of active flares and steroid requirements in the first four years of treatment. Continuation beyond four years led to a further reduction in steroid use and improvement in disease activity.28 Patients in complete remission for many years may be able to discontinue AZA or 6-MP, but continuing therapy may be beneficial in most patients.
Cyclosporine is an immunosuppressant that works by blocking lymphocyte activation. It can be used as a rescue therapy for patients with severe, refractory UC. High doses can be effective in achieving remission in up to 80% of hospitalized patients who have been unresponsive to CCSs.29
Cyclosporine alone is not effective for maintaining remission, but coadministration with AZA or 6-MP for a few months effectively maintains remission in the majority of patients who initially responded to IV cyclosporine.29
Infliximab is a chimeric monoclonal antibody directed against the proinflammatory cytokine tumor necrosis factor. It has been used to treat Crohn's disease for many years, and now is being used for patients with active UC who have not responded to conventional therapies.30
While earlier studies have reported conflicting results, the more recent randomized controlled ACT1 and ACT2 trials have established the efficacy of infliximab for inducing and maintaining remission for outpatients with moderate to severe UC for up to one year. Almost 25% of infliximab-treated patients were completely off steroids and in clinical remission at the end of the study compared to less than 10% of controls. In addition, the study showed that patients treated with infliximab were less likely to have mucosal damage, which may be significant since normal mucosa has been associated with a reduction in the risk of colorectal cancer.31
Although methotrexate and antibiotics like metronidazole may play a role in the treatment of Crohn's disease, there is not enough evidence to support the use of either in UC. Antibiotics should only be used if there is a known or suspected infection or immediately before surgery.3
Antidiarrheals or antispasmodics may help with mild diarrhea and abdominal cramping as long as obstruction and colonic dilation are not suspected. Routine opioids should be avoided due to the risk of addiction and the potential for inducing toxic megacolon. Patients who receive frequent CCSs should receive calcium and vitamin D supplementation, as well as bisphosphonates, due to the risk of osteoporosis.26
Conclusion
With proper treatment, most patients with UC can lead normal, healthy lives. Compliance is vital for both induction and maintenance of remission, as well as for long-term colorectal cancer prophylaxis. Up to 60% of patients, however, may be noncompliant during periods of remission.32 Since patients are more likely to stay in remission with ongoing treatment, it is imperative that they are educated on the importance of medication adherence, even during symptom-free periods. Pharmacists should take advantage of frequent contact with patients to ensure that patients are taking their medications correctly and to identify any potential problems or barriers to adherence that may need to be addressed.
REFERENCES
1. Loftus EV. Clinical epidemiology of inflammatory bowel disease: incidence, prevalence, and environmental influences. Gastroenterology. 2004;126:1504-1517.
2. Regueiro M, Loftus EV, Steinhart AH, et al. Medical management of left-sided ulcerative colitis and ulcerative proctitis: critical evaluation of therapeutic trials. Inflamm Bowel Dis. 2006;12:979-994.
3. Carter MJ, Lobo AJ, Travis SP. Guidelines for the management of inflammatory bowel disease in adults. Gut. 2004;53(suppl 5):v1-v16.
4. Kornbluth A, Sachar DB. Ulcerative colitis practice guidelines in adults (update): American College of Gastroenterology, Practice Parameters Committee. Am J Gastroenterology. 2004;99:1371-1385.
5. Strober W, Fuss I, Mannon P. The fundamental basics of inflammatory bowel disease. J Clin Invest. 2007;117:514-521.
6. Langan RC, Gotsch PB, Krafczyk MA, Skillinge DD. Ulcerative colitis: diagnosis and treatment. Am Fam Physician. 2007;76:1323-1330.
7. Kornbluth A, Marion JF, Salomon P, Janowitz HD. How effective is current medical therapy for severe ulcerative and Crohn's colitis? An analytical review of selected trials. J Clin Gastroenterol. 1995;20:280-284.
8. Sutherland L, Macdonald JK. Oral 5-aminosalicylic acid for induction of remission in ulcerative colitis. Cochrane Database Syst Rev. 2006;(2):CD000543.
9. Sutherland L, Macdonald JK. Oral 5-aminosalicylic acid for maintenance of remission in ulcerative colitis. Cochrane Database Syst Rev. 2006;(2):CD000544.
10. Fernandez-Becker NQ, Moss AC. Improving delivery of aminosalicylates in ulcerative colitis: effect on patient outcomes. Drugs. 2008;68:1089-1103.
11. Nielsen OH, Munck LK. Drug insight: aminosalicylates for the treatment of IBD. Nat Clin Pract Gastroenterol Hepatol. 2007;4:160-170.
12. McCormack PL, Robinson DM, Perry CM. Delayed-release Multi Matrix System (MMX) mesalazine: in ulcerative colitis. Drugs. 2007;67:2635-2642.
13. Regueiro M, Loftus EV, Steinhart AH, Cohen RD. Clinical guidelines for the medical management of left-sided ulcerative colitis and ulcerative proctitis: summary statement. Inflamm Bowel Dis. 2006;12:972-978.
14. Marshall JK, Irvine EJ. Putting rectal 5-aminosalicylic acid in its place: the role in distal ulcerative colitis. Am J Gastroenterology. 2000;95:1628-1636.
15. Gionchetti P, Venturi A, Rizzello F, et al. Retrograde colonic spread of a new mesalazine rectal enema in patients with distal ulcerative colitis. Ailment Pharmacol Ther. 1997;11:679-684.
16. Otten MH, De Haas G, Van den Ende R. Colonic spread of 5-ASA enemas in healthy individuals with a comparison of their physical and chemical characteristics. Ailment Pharmacol Ther. 1997;11:693-697.
17. Kam L, Cohen H, Dooley C, et al. A comparison of mesalamine suspension enema and oral sulfasalazine for treatment of active distal ulcerative colitis in adults. Am J Gastroenterol. 1996;91:1338-1342.
18. Lakatos PL, Lakatos L. Ulcerative proctitis: a review of pharmacotherapy and management. Expert Opin Pharmacother. 2008;9:741-749.
19. Hu MY, Peppercorn MA. MMX mesalamine: a novel high-dose, once-daily 5-aminosalicylate formulation for the treatment of ulcerative colitis. Expert Opin Pharmacother. 2008;9:1049-1058.
20. Safdi AV, Cohen RD. Review article: increasing the dose of oral mesalazine therapy for active ulcerative colitis does not improve remission rates. Aliment Pharmacol Ther. 2007;26:1179-1186.
21. Pruitt R, Levine DS, Safdi M, et al. Balsalazide as alternative therapy to mesalamine dose-escalation for acute, mild to moderate ulcerative colitis. Gastroenterology. 2002;122:A299.
22. James SL, Irving PM, Gearry RB, Gibson PR. Management of distal ulcerative colitis: frequently asked questions. Intern Med J. 2008;38:114-119.
23. Lichtenstein G, Abreu M, Cohen R, et al. American Gastroenterological Association Institute technical review on corticosteroids, immunomodulators, and infliximab in inflammatory bowel disease. Gastroenterology. 2006;130:940-987.
24. Faubion WA Jr, Loftus EV Jr, Harmsen WS, et al. The natural history of corticosteroid therapy for inflammatory bowel disease: a population-based study. Gastroenterology. 2001;121:255-260.
25. Nakamura T, Nagahori M, Kanai T, Watanabe M. Current pharmacologic therapies and emerging alternatives in the treatment of ulcerative colitis. Digestion. 2008;77(suppl 1):36-41.
26. Hanauer SB. Inflammatory bowel disease. N Engl J Med. 1996;334:841-848.
27. Timmer A, McDonald JW, Macdonald JK. Azathioprine and 6-mercaptopurine for maintenance of remission in ulcerative colitis. Cochrane Database Syst Rev. 2007;(1):CD000478.
28. Holtmann MH, Krummenauer F, Claas C, et al. Long-term effectiveness of azathioprine in IBD beyond 4 years: a European multicenter study in 1176 patients. Dig Dis Sci. 2006;51:1516-1524.
29. Podolsky DK. Inflammatory bowel disease. N Engl J Med. 2002;347:417-429.
30. Panaccione R, Fedorak RN, Aumais G, et al. Review and clinical perspectives for the use of infliximab in ulcerative colitis. Can J Gastroenterol. 2008;22:261-272.
31. Rutgeerts P, Sandborn WJ, Feagan BG, et al. Infliximab for induction and maintenance of ulcerative colitis. N Engl J Med. 2005;353:2462-2476.
32. Kane S, Huo D, Aikens J, et al. Medication nonadherence and the outcomes of patients with quiescent ulcerative colitis. Am J Med. 2003;114:39-43.
33. Chang JC, Cohen RD. Medical management of severe ulcerative colitis. Gastroenterol Clin North Am. 2004;33:235-250.
34. Electronic Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations.www.fda.gov/cder/ob/default.htm. Accessed August 19, 2008.
To comment on this article, contact
rdavidson@jobson.com.





