US Pharm.2008;33(6)(Generic
Drug Review suppl):20-27.
There is little debate that
the Rx-to-OTC switch movement (when a prescription drug is reclassified
to nonprescription or OTC status) has caused major changes in the pharmacy
marketplace. When the patent on a prescription medication expires, the
manufacturer typically experiences a dip in profits due to emerging
competition from generics. Moving a prescription drug to nonprescription
status does not automatically confer increased patent life. However, if the
FDA requires the manufacturer to carry out additional studies via a
supplemental new drug application to demonstrate safety and/or efficacy for
the proposed switch, the manufacturer can gain as much as three years of
patent exclusivity. Eventually, though, even the extended patent protection
expires, and the medication often becomes available as a generic.
The Pharmacist's Dilemma
with OTC Medications
Pharmacy students
are taught to recommend nonprescription or OTC products to patients on the
basis of two fundamentally important parameters--safety and efficacy. Cost
considerations often take a backseat. However, the pharmacist counseling a
patient often notices that a discussion of the optimal nonprescription product
for a minor health care condition eventually turns to the cost of the
recommended product. The pharmacist may recommend the most appropriate OTC
product on the basis of safety and efficacy, as he or she was taught, only to
watch helplessly as the price-conscious patient chooses a less appropriate
product, solely because it is less expensive than the one recommended. For
this reason, it is helpful to the pharmacist when OTC medications are marketed
generically, as narrowing price gaps between therapeutic options allow the
patient to choose the most safe and effective product.
Advantages of OTC or
Nonprescription Generics
The chief benefit
to patients when a nonprescription medication becomes available generically is
identical to the situation with prescription medications: prices for generic
OTC versions are lower than those for the branded products. The savings vary
from product to product, but they can be as little as 11% (some omeprazole
generics) to over 75% (some loratadine generics).1 For the
price-conscious patient, the cost savings can be critical in making self-care
decisions. Further, for the price-sensitive patient with a chronic,
self-treatable medical condition, the addition of a new generic OTC with that
indication expands the range of therapeutic options.
Frequently, the newer generic
OTC medication possesses unique advantages that make it a better choice for
many patients. This is illustrated by the gradually increasing group of
options for allergic rhinitis. At one time, the patient with allergic rhinitis
had little to choose from other than older (first-generation)
antihistamines. When loratadine (Claritin) and cetirizine (Zyrtec) switched to
OTC status, their contraindications for self-treating patients differed from
those pertaining to first-generation antihistamines. These second-generation
antihistamines were not inexpensive when first introduced. However, when they
became generic, price-conscious patients could choose them for their unique
advantages and lower price.
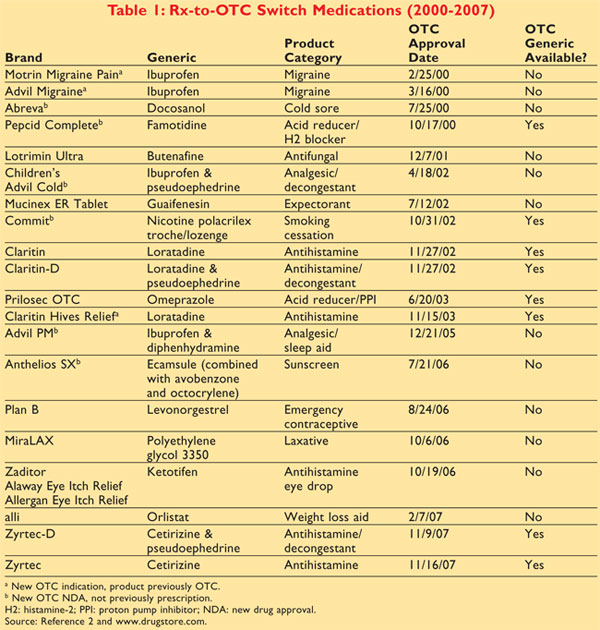
Drugs transferred from
Rx-to-OTC status by the FDA since 2000 are listed in TABLE 1.2
This review will focus on three major categories: antihistamines that treat
allergic rhinitis, acid reducers for heartburn/indigestion, and smoking
cessation products.
Allergic Rhinitis
Brompheniramine and
chlorpheniramine became the first nonprescription antihistamines when the FDA
approved their OTC sale in 1976.2 Dexbrompheniramine and
triprolidine followed in 1982, diphenhydramine (Benadryl) in 1985, doxylamine
in 1987, and clemastine (Tavist) and dexchlorpheniramine in 1992.2
By 1992, the pantheon of nonprescription first-generation antihistamines was
completed.
First-generation
antihistamines are grouped together because they share several
characteristics. One is the list of contraindications for their safe self-use.
They should not be used without physician supervision if the patient has any
of the following: emphysema, chronic bronchitis, glaucoma, or difficulty in
urination due to enlargement of the prostate gland.3 Further, all
were clearly contraindicated for self-administration in patients under the age
of 6 years to avoid excitability, despite patient dosing charts marketed by
several companies that recommended their use in children as young as 6 months
without FDA proof of safety and efficacy.3 Their labels cautioned
against use with sedatives or tranquilizers without first speaking to a doctor
or pharmacist. They cautioned that drowsiness might occur (marked drowsiness
with diphenhydramine) and cautioned against concomitant use with alcohol or
when driving or operating heavy machinery.
In 2002, the FDA approved the
nonprescription sale of Claritin. It became available generically as
loratadine within a few years.2 In 2007, a switch for Zyrtec was
approved, and generic cetirizine became available in 2008.2 These
drugs are referred to as second-generation antihistamines for several reasons.
Foremost is the fact that they are safe for self-use in patients with
emphysema, chronic bronchitis, glaucoma, and difficulty in urination due to
enlargement of the prostate gland. Thus, the price-conscious patient with
these medical conditions who wished to use a generically available
antihistamine was able to purchase loratadine and cetirizine when they became
available generically. However, they should not be used by patients with liver
or kidney disease without first speaking to a physician.3
The second-generation
antihistamines also brought another large benefit to price-conscious patients
when they became available generically. Loratadine syrup can be given to
children as young as 2 years of age, greatly extending the ability of parents
to safely treat children with allergic rhinitis.3 Eventually,
cetirizine syrup may also be available generically. As Children's Zyrtec
Allergy Syrup carries labeling for children as young as 2 years, it can be
predicted that the generic version will also carry this labeling.4
Generic availability of
loratadine and cetirizine brings another benefit to price-conscious patients.
They are widely considered to be nonsedating. The patient who must drive or
operate heavy machinery was once locked into the more sedating
first-generation antihistamines. That is no longer the case, as loratadine and
cetirizine are less likely to cause drowsiness. Loratadine also does not carry
the warnings against use when driving or operating heavy machinery or use with
alcohol, sedatives, or tranquilizers.3
However, it should be noted
that the product labels of both cetirizine and loratadine caution that
drowsiness may occur when taking normal doses (cetirizine) or when taking more
than directed (loratadine).3,5 Further, cetirizine carries the same
warnings as first-generation antihistamines in regard to use of alcohol,
sedatives, and tranquilizers and when driving or operating heavy machinery.
5
Another option for allergic
rhinitis is NasalCrom Nasal Spray.3 Currently, there is no generic
version available. However, if it is ever marketed, the generic OTC product
would also present unique advantages in regard to contraindications and other
considerations that may be important to the price-conscious patient.
Heartburn and Acid
Indigestion
Prior to 1995, the
patient with heartburn, acid indigestion, or sour stomach had little choice
for self-treatment except nonprescription antacids and antirefluxants. These
products were limited in their method of action and by their adverse
reactions. Those containing calcium (e.g., Tums) caused constipation, while
those containing magnesium (e.g., Milk of Magnesia) were more prone to cause
diarrhea.6 Products with magnesium were not for self-treatment
without physician advice if the patient had kidney disease. Those with sodium
(e.g., Alka-Seltzer) were not to be used by patients on sodium-restricted
diets without physician advice. Antirefluxants (e.g., Gaviscon) allegedly
prevented reflux through the creation of a "raft" of nonirritating alginate
that supposedly floated atop the stomach contents to prevent more irritating
materials from being refluxed.6 Antacids and antirefluxants only
neutralized acid that had already been produced, greatly limiting their
utility.
This nonprescription market
changed radically with the Rx-to-OTC switch of the histamine-2 (H2) blocking
agents famotidine (Pepcid AC) and cimetidine (Tagamet HB) in 1995, and
ranitidine (Zantac 75) and nizatidine (Axid AR) in 1996.2 The first
three OTC products are available generically. Rather than simply neutralizing
acid that has already been produced, the H2 blockers prevent acid production,
making them more powerful medications for heartburn, acid indigestion, and
sour stomach. They should not be taken by those aged under 12 years or taken
more than two weeks after onset of symptoms.6 They can be used to
treat active symptoms, as well as to prevent symptoms. They should not be used
with other acid reducers.
This market again changed
radically when the proton pump inhibitor (PPI) omeprazole (Prilosec OTC) was
given the green light for a switch by the FDA in 2003, and again when it
recently became available in a generic form.2 PPIs are generally
considered to be more effective than either antacids or H2 blockers, so its
generic availability was an advance for the price-conscious patient.
Omeprazole is the only product currently FDA approved for frequent heartburn
that occurs on two or more days per week. (For less frequent heartburn, the
patient might try H2 blockers or antacids.) Omeprazole does not provide
immediate relief. It may take one to four days for the patient to gain the
full effect.6 Patients take one tablet with a full glass of water
every 24 hours, in the morning before eating, for 14 consecutive days. The
patient cannot repeat this regimen more often than every four months. Any
patient who feels the need to take it beyond 14 days in a row or more than
every four months should be counseled to visit a physician instead. Those
under the age of 18 years should not take omeprazole.6
Patients taking omeprazole
should be told that it does not provide immediate relief. Patients may not
notice the full effect for one to four days, although some do obtain complete
relief within 24 hours. It should not be recommended if the patient has an
allergy to omeprazole, trouble or pain when swallowing food, vomiting of
blood, or bloody or black stools. Patients should consult a physician before
taking it if they have any of the following: heartburn that has persisted for
more than three months; heartburn along with lightheadedness, sweating or
dizziness; chest or shoulder pain with shortness of breath, sweating, pain
that spreads to the arms/neck/shoulders, or lightheadedness; frequent chest
pain; frequent wheezing, particularly with heartburn; unexplained weight loss;
nausea or vomiting; and stomach pain. Patients should consult a pharmacist or
physician before taking omeprazole if they are also taking warfarin,
prescription antifungal or antiyeast medications, diazepam, or digoxin.6
Smoking Cessation
Before 1996, there had never been a
safe and effective nonprescription product for smoking cessation. Several
herb-based and homeopathic products had been marketed and are still being
marketed, but their safety and efficacy have never been established. The
nonprescription smoking cessation market gained its first safe and effective
product in 1996 with FDA approval to switch nicotine polacrilex gum
(Nicorette) to nonprescription status.2 The market expanded in 1996
with the approval of nonprescription transdermal patches.2 The
final dosage form, the nicotine lozenge, was approved for OTC sale in 2002.
2 At this time, all are available generically.
The OTC smoking cessation
products all share some important warnings. They cannot be sold legally to
those under the age of 18 years, and they cannot be placed in a vending
machine or any other location where the age of the purchaser cannot be
verified. They are not safe for the pregnant or breastfeeding patient, as they
can cause tachycardia in the infant. Rather, the pharmacist should urge these
patients to attempt smoking cessation without the products. They must not be
used if the patient will continue to smoke, chew tobacco, or use snuff. They
cannot be combined with each other and should not be used with prescription
smoking cessation therapies unless a physician advises them to do so.7
Patients should not use
smoking cessation products without physician advice if they have heart disease
or irregular heartbeat, or have experienced a recent heart attack, as they can
increase the heart rate. They should not use them without physician advice if
they have hypertension uncontrolled by medication, as nicotine can increase
blood pressure. They should speak to a physician or pharmacist before use if
they are taking prescription medications for depression or asthma, as nicotine
can alter the response to theophylline or imipramine through its
enzyme-inducing properties. They should stop use and speak to a physician if
they develop irregular heartbeat or palpitations, symptoms of nicotine
overdose (e.g., nausea, vomiting, dizziness, weakness, tachycardia), or
diarrhea with the gum and lozenge products.7
Nicotine gum and lozenges
should not be used in patients with stomach ulcers because they may delay
healing. Similarly, they should be avoided in patients with diabetes as they
promote hyperinsulinemia, leading to insulin resistance. Nicotine transdermal
patches should be avoided by patients with allergies to adhesive tape or with
skin problems such as atopic dermatitis.7
Conclusion
When medications
switch from prescription to nonprescription status (the Rx-to-OTC switch),
patients benefit greatly. Price-conscious patients can realize these benefits
when the OTC medication also becomes available generically. Generic
availability of recently switched products for heartburn, allergic rhinitis,
and smoking cessation is illustrative of the expanding range of treatment
options available to price-sensitive patients.
REFERENCES
1. Omeprazole.
Loratadine. www.drugstore.com. Accessed May 5, 2008.
2. Ingredients and dosages transferred from Rx-to-OTC status (or new OTC approvals) by the Food and Drug Administration since 1975. Consumer Healthcare Products Association. November 20, 2007. www.chpa-info.org/NR/rdonlyres/E33F414C-24FE-4F34-B6FD-149D16DCE2FA/0/switch_list112007.pdf. Accessed April 21, 2008.
3. Pray WS. Allergic rhinitis. In: Pray WS, ed. Nonprescription Product Therapeutics. 2nd ed. Baltimore, MD: Lippincott Williams & Wilkins; 2006:219-234.
4. Children's Zyrtec Allergy Syrup (cetirizine) package insert. Fort Washington, PA: McNeil-PPC, Inc; 2007.
5. Zyrtec Tablets (cetirizine) package insert. Fort Washington, PA: McNeil-PPC, Inc; 2007.
6. Pray WS. Gastric distress. In: Pray WS, ed. Nonprescription Product Therapeutics. 2nd ed. Baltimore, MD: Lippincott Williams & Wilkins; 2006:81-105.
7. Pray WS. Nicotine addiction. In:
Pray WS, ed. Nonprescription Product Therapeutics. 2nd ed. Baltimore,
MD: Lippincott Williams & Wilkins; 2006:736-748.
To comment on this article,
contact
rdavidson@jobson.com.






