US Pharm. 2015;40(9):13-19.
Vulvovaginal complaints are among the most common reasons women seek medical care. Vaginitis, defined as a variety of inflammatory conditions that can result in vaginal and sometimes vulvar symptoms such as itching, burning, irritation, odor, and vaginal discharge, accounts for approximately 10 million physician office visits annually.1,2 Among the many causes of vaginitis, vulvovaginal candidiasis (VVC), also referred to as a yeast infection, is the second most common cause after bacterial vaginosis.3 VVC, caused by fungi of the genus Candida, is diagnosed in up to 40% of females presenting with vaginal complaints in the primary care setting.1 The prevalence of VVC is highest among women of reproductive age; it is estimated that about 75% of women will have at least one episode of VVC, and 40% to 45% will have two or more episodes.4 Approximately 5% to 8% of VVC cases are recurrent, defined as four or more episodes per year.5
VVC is the only vaginal infection for which OTC treatments are available, allowing women to self-diagnose and self-treat their condition. Since the introduction of these OTC products in the early 1990s, many women have associated vaginitis-type symptoms with yeast infections, leading to increased self-treatment of presumed VVC. From 1993 to 1997, OTC sales of vaginal antifungal products and feminine hygiene products increased from $90 million to $250 million6; during this same time period, physician office visits for vaginal conditions and prescriptions decreased by 15%.7 Today, healthcare costs are estimated to be between $4 and $5 billion annually, with OTC sales reaching $295 million.8,9
Although there are many advantages to having vaginal antifungals readily available, there is a potential for inappropriate therapy. Some women may have difficulties identifying VVC based on symptoms alone, and others may have problems selecting and using these products appropriately. One study found that only one-third of women correctly diagnosed themselves; prior diagnosis only moderately affected their ability to correctly diagnosis vaginal Candida infection.10 Similarly, another study reported that many women who purchased an OTC antifungal vaginal product did not have VVC, and many utilized the product up to three times in a 6-month period.11 Pharmacists play a pivotal role in managing VVC by helping patients understand the importance of proper diagnosis, product selection, and use.
Etiology
Under normal circumstances, the vagina maintains a balance among organisms that make up the vaginal microflora. Vaginal pH is maintained between 3.8 and 4.2 by Lactobacillus acidophilus, diphtheroids, and Staphylococcus epidermidis.12,13 The normal vaginal environment protects women against vaginal infections. Any alterations in this environment allow for the overgrowth of organisms normally suppressed, including Candida.
Candida albicans is the most common pathogen identified, responsible for 85% to 95% of VVC episodes.14 Asymptomatic colonization is also common; it is identified in almost one-third of women without any symptoms.15 The second most common pathogen is Candida glabrata, which is isolated in 7% to 16% of all cases.5 The incidence of non–C albicans VVC is increasing, and this may be attributed to overuse of OTC antifungal vaginal products, single-dose treatments, and low-dosage azole maintenance regimens.14
Although many women will develop VVC sporadically, there are several factors that can increase the risk.13-15 These factors include antibiotic use, hormones, immune system status, diabetes, and lifestyle habits. Broad-spectrum antibiotic use alters the bacterial microflora, allowing for the overgrowth of Candida organisms. Colonization with C albicans increased from 10% to 30%, and VVC was diagnosed in 28% to 33% of cases.14 Women who are already colonized may have a greater risk.16 However, some studies have failed to show a link between antibiotic use and development of VVC.17 Elevated estrogen levels, either through oral contraceptives, hormone replacement, or pregnancy, enhance adherence of Candida to vaginal epithelial cells.13,14 Immunosuppression lowers the patient’s ability to fight infections, reducing vaginal protection by immunoglobulins.13 Diabetic patients who are uncontrolled are more prone to VVC, as hyperglycemia also enhances binding of Candida to the vaginal epithelial wall.13 Those who are nondiabetic but consume a diet high in refined sugars may be susceptible to VVC as well.14
Lifestyle habits also play an important role by introducing microorganisms to the vaginal environment and altering the normal flora, as by not cleansing correctly after bowel movements or wearing tight-fitting clothing that promotes a warm, moist environment. VVC is often associated with sexual activity, although women who are not sexually active can develop infections. Asymptomatic colonization of the male genitalia with Candida is four times more common in the sexual partners of infected females.14 Use of diaphragms, intrauterine devices, and sponges may also pose a risk; these items can increase the adherence of a yeast, potentiating the development of VVC in susceptible patients.18,19
Classification
VVC can be classified as either un-complicated or complicated based on clinical presentation, microbiology, host factors, and response to therapy.4 Uncomplicated VVC occurs sporadically or infrequently, is mild-to-moderate in nature, is most likely caused by C albicans, and affects nonimmunocompromised women. VVC is considered complicated if it is recurrent, severe in nature, and caused by non-albicans, or if it affects women with diabetes, immunocompromising conditions, debilitation, or those on immunosuppressive therapy.
Clinical Presentation
The symptoms of VVC are usually nonspecific and can be due to a variety of causes. The hallmark symptoms in most women are vulvar pruritus and burning. This is usually accompanied with soreness and irritation, which can lead to dyspareunia and dysuria in more severe cases.1 Other common symptoms include vaginal soreness, irritation, and a white vaginal discharge, which varies from watery to homogenously thick, described typically as cottage cheese–like.14 If there is an odor present, it is usually very faint.14 Upon examination, the labia and vulva will be erythematous and swollen. Approximately 25% of patients will have fissures and excoriations on the external genitalia.20 Usually, symptoms worsen the week before menses and during pregnancy.14,20
Assessing Self-Treatment
Self-treatment with the available OTC vaginal antifungals is only appropriate in patients with uncomplicated, infrequent episodes of VVC with symptoms ranging from mild-to-moderate in severity. It is important to be able to differentiate between VVC and other causes of vaginitis, including bacterial vaginosis and trichomoniasis, so that appropriate treatment is not delayed.1,21 The pharmacist can assist patients in determining the most appropriate course of action by first obtaining pertinent information about their symptoms. Pharmacists can also recommend the use of vaginal screening kits to help patients identify, confirm, or rule out VVC. When compared to standard diagnostic testing for VVC, rapid testing was shown to be accurate and affordable.22 The patient should be referred for a medical diagnosis if this is her first episode of VVC; she is pregnant; experiences recurrent episodes or recurrence of symptoms within 2 months after treatment; experiences persistent symptoms despite treatment; has concomitant symptoms of fever, lower abdominal pain, or pain in the back or shoulders; has a malodorous vaginal discharge; or is <12 years of age.4,13,23
Nonprescription Treatment
All OTC vaginal antifungal products are approved for the treatment of VVC. Short-course topical formulations or single-dose oral treatment has been shown to effectively treat up to 90% of uncomplicated VVC.4 No single agent, topical or oral, has demonstrated superiority; they all have shown equivalent results.24 The various topical azoles available OTC include clotrimazole, miconazole, and tioconazole. They come in 1-, 3-, and 7-night regimens, in a variety of formulations including suppositories (vaginal tablets/ovules), creams, and ointments and in combination packages (TABLE 1).4,25,26
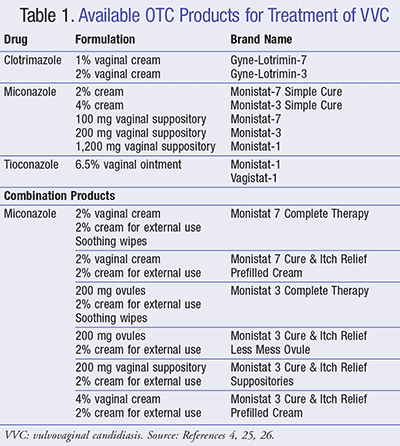
These products are generally considered safe, although some patients have complained about a burning sensation.14 Single-dose or short-course topical therapy is the preferred regimen of the CDC.4 Studies to date have not demonstrated that one duration of therapy is significantly better.27,28 Combination products, containing both a vaginal insert and external cream, have an additional use for the relief of itching and irritation on the skin outside the vulva. One comparative study concluded that the combined use of both clotrimazole vaginal suppositories and clotrimazole cream for external use was better at treating VVC than the suppositories alone.29
Product selection will be based mostly on patient preference. Factors to be considered include cost, length of therapy, formulation, convenience, and ease of use. Creams should be used at bedtime, preferably with a sanitary pad to help absorb any leaking. Ovules have the advantage that they may be used at any time of day. If a patient is currently taking warfarin, any product containing miconazole should be used cautiously. Although there is limited absorption with topical agents, an increased INR has been documented with concomitant use of warfarin and intravaginal miconazole.30
Patient counseling should include information on proper use, including instructions on cleaning the applicator if using reusable applicators. Symptoms should improve within 2 to 3 days after initiating therapy. Patients need to be informed that the length of therapy does not indicate when symptom resolution will occur; full resolution usually takes up to 1 week. A full course of therapy should be completed even after resolution of symptoms. Therapy should also be continued even if the patient’s menstrual period begins. In addition, patients need to be informed that the creams and suppositories are oil-based, which may weaken latex condoms and diaphragms.4
Some patients have turned to probiotics for preventing VVC. In VVC infections, Lactobacillus bacteria help maintain the normal vaginal flora and prevent the overgrowth of Candida organisms. Published studies have shown conflicting results, but Lactobacillus rhamnosus GR-1, Lactobacillus fermentum RC-14, and L acidophilus have been shown to be somewhat effective; eating 8 oz of yogurt with live cultures daily may also provide some benefit.31 Further research is needed before specific recommendations on its use can be made.
Conclusion
VVC is a very common gynecologic condition that will affect almost every woman at least once in her lifetime. With the availability of many OTC treatment options, women have the ability to quickly and effectively treat this condition; however, pharmacists play an important role in helping patients accurately assess and properly use these products.
PATIENT INFORMATION

What Causes Vulvovaginal Candidiasis (VVC)?
VVC is most commonly caused by a fungus known as Candida albicans (it can also be caused by other fungi). C albicans is normally found in the vaginal environment; overgrowth of this fungus, or any other fungi, could lead to VVC. Several conditions and behaviors allow for fungal overgrowth, predisposing individuals to VVC. Conditions include pregnancy, diabetes, HIV/AIDS, or any immunocompromised state. Behavioral risk factors include the use of high-dose estrogen oral contraceptives, antibiotics, systemic corticosteroids, tight synthetic underwear, spermicides, and oral sexual contact.
VVC vs. Bacterial Infections
It is imperative to differentiate between yeast infections and bacterial infections; only yeast infections are self-treatable. Antifungal medications would not be effective in treating bacterial infections. Symptoms of VVC include burning pain, itchiness, swelling, and redness of the vulva, which may lead to painful/difficult urination. A thick, odorless, cottage cheese–like discharge is a paramount predictor of VVC; bacterial infections tend to present with foul odor.
If unable to determine whether the infection is of fungal origin, you can use an OTC screening kit (e.g., Fem-V or Vagisil Screening Kit). These test the pH of vaginal secretions. A pH higher than 4.5 indicates that the infection is not likely fungal, making antifungal treatments useless. You cannot use these kits until 72 hours after the use of any vaginal or antifungal product, 48 hours after sexual intercourse or douching, and/or 5 days after menstruation.
Uncomplicated vs. Complicated Infections
Uncomplicated infections may be self-treated, but complicated cases require a medical consult. Complicated infections include recurrent VVC (4 or more episodes within 12 months or recurrent symptoms within 2 months), VVC with severe symptoms, and/or VVC while pregnant, diabetic, or immunocompromised.
OTC Treatments
There are many OTC antifungal agents that can treat VVC. They generally differ in cost, formulation, and duration of treatment (1, 3, or 7 days), but are all equally effective. These include miconazole (Monistat), tioconazole (Vagistat), and clotrimazole (Gyne-Lotrimin). Although adverse effects are not common, you may experience vaginal burning and/or irritation. It is vital to know that length of treatment does not always correspond with resolution of symptoms, particularly for shorter treatments; symptoms may take up to a week to resolve.
A sitz bath may be used to provide prompt relief. Daily consumption of yogurt with live cultures may help prevent VVC recurrences. It is also important to refrain from sexual intercourse during and for 3 days after treatment; antifungals may damage condoms and diaphragms, leading to unreliable contraception.
When to Seek Medical Attention
Several factors require a medical consult. These include age under 12 years; pregnancy; fever; pain in the lower abdomen, lower back, or shoulder; complicated VVC; use of corticosteroids, antineoplastic drugs, or immunosuppressants; and failure of symptoms to resolve after 1 week of treatment.
Remember, if you have questions, Consult Your Pharmacist.
REFERENCES
1. Anderson MR, Klink K, Cohrssen A. Evaluation of vaginal complaints. JAMA. 2004;291(11):1368-1379.
2. Hainer BL, Gibson MV. Vaginitis. Am Fam Physician. 2011;83(7):807-815.
3. Martin Lopez JE. Candidiasis (vulvovaginal). BMJ Clin Evid. 2015;2015:pii0815.
4. Workowski KA, Bolan GA. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm Rep. 2015;64(RR-03):1-137.
5. Dovnik A, Golle A, Novak D, et al. Treatment of vulvovaginal candidiasis: a review of the literature. Acta Dermatovenerol Alp Pannonica Adriat. 2015;24(1):5-7.
6. Theroux R. Factors influencing women’s decisions to self-treat vaginal symptoms. J Am Acad Nurse Pract. 2005;17(4):156-162.
7. Lipsky MS, Waters T, Sharp LK. Impact of vaginal antifungal products on utilization of health care services: evidence from physician visits. J Am Board Fam Pract. 2000;13(3):178-182.
8. Consumers Healthcare Products Association. OTC sales by category 2011-2014. www.chpa.org/OTCsCategory.aspx. Accessed July 20, 2015.
9. NovaDigm Therapeutics. Vulvovaginal candidiasis. www.novadigm.net/development-programs/vulvovaginal-candidiasis.php. Accessed July 30, 2015.
10. Ferris DG, Nyirjesy P, Sobel JD, et al. Over-the-counter antifungal drug misuse associated with patient-diagnosed vulvovaginal candidiasis. Obstet Gynecol. 2002;99(3):419-425.
11. Sihvo S, Ahonen R, Mikander H, Hemminki E. Self-medication with vaginal antifungal drugs: physicians’ experiences and women’s utilization patterns. Fam Pract. 2000;17(2):145-149.
12. Egan ME, Lipsky MS. Diagnosis of vaginitis. Am Fam Physician. 2000;62(5):1095-1104.
13. Schlesselman L. Superficial fungal infections. In: Chisholm-Burns MA, Wells BG, Schwinghammer TL, et al, eds. Pharmacotherapy: Principles and Practice. 3rd ed. New York, NY: McGraw-Hill; 2013:1425-1438.
14. Sobel JD. Vulvovaginal candidosis. Lancet. 2007;369(9577):1961-1971.
15. Achkar JM, Fries BC. Candida infections of the genitourinary tract. Clin Microbiol Rev. 2010;23(2): 253-273.
16. Pirotta MV, Garland SM. Genital Candida species detected in samples from women in Melbourne, Australia, before and after treatment with antibiotics. J Clin Microbiol. 2006;44(9):3213-3217.
17. Glover DD, Larsen B. Relationship of fungal vaginitis therapy to prior antibiotic exposure. Infect Dis Obstet Gynecol. 2003;11(3):157-160.
18. Camacho DP, Consolaro ME, Patussi EV, et al. Vaginal yeast adherence to the combined contra-ceptive vaginal ring (CCVR). Contraception. 2007;76(6):439-443.
19. Chassot F, Negri MF, Svidzinski AE, et al. Can intrauterine contraceptive devices be a Candida albicans reservoir? Contraception. 2008;77(5): 355-359.
20. Ilkit M, Guzel AB. The epidemiology, patho-genesis, and diagnosis of vulvovaginal candidosis: a mycological perspective. Crit Rev Microbiol. 2011;37(3):250-261.
21. Farage MA, Miller KW, Ledger WJ. Determining the cause of vulvovaginal symptoms. Obstet Gynecol Surv. 2008;63(7):445-464.
22. Chatwani AJ, Mehta R, Hassan S, et al. Rapid testing for vaginal yeast detection: a prospective study. Am J Obstet Gynecol. 2007;196(4):309.e1-309.e4.
23. Goad JA, Hess KM. Sexually transmitted diseases. In: Alldredge BK, Corelli RL, Ernst ME, et al, eds. Applied Therapeutics: The Clinical Use of Drugs. 10th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2013:1619-1647.
24. Pappas PG, Kauffman CA, Andes D, et al. Clinical practice guidelines for the management of candidiasis: 2009 update by the Infectious Diseases Society of America. Clin Infect Dis. 2009;48(5): 503-535.
25. Monistat product information. Prestige Brands Holdings, Inc. 2015. www.monistat.com. Accessed July 30, 2015.
26. Vagistat product information. Novartis Consumer Health, Inc. 2015. www.vagistat.com. Accessed July 30, 2015.
27. Edelman DA, Grant S. One-day therapy for vaginal candidiasis. A review. J Reprod Med. 1999;44(6):543-547.
28. Upmalis DH, Cone FL, Lamia CA, et al. Single-dose miconazole nitrate vaginal ovule in the treatment of vulvovaginal candidiasis: two single-blind, controlled studies versus miconazole nitrate 100 mg cream for 7 days. J Womens Health Gend Based Med. 2000;9(4):421-429.
29. Mendling W, Schlegelmilch R. Three-day combination treatment for vulvovaginal candidosis with 200 mg clotrimazol vaginal suppositories and clotrimazol cream for the vulva is significantly better than treatment with vaginal suppositories alone—an earlier, multi-centre, placebo-controlled double blind study. Geburtshilfe Frauenheilkd. 2014;74(4):355-360.
30. Wooltorton E. Drug advisory: the interaction between warfarin and vaginal miconazole. CMAJ Can Med Assoc J. 2001;165(7):938.
31. Falagas ME, Betsi GI, Athanasiou S. Probiotics for prevention of recurrent vulvovaginal candidiasis: a review. J Antimicrob Chemother. 2006;58(2):266-272.
To comment on this article, contact rdavidson@uspharmacist.com.






