US Pharm. 2018;43(4):33-36.
ABSTRACT: Specialty medications are expected to account for 50% of drug spending within the next 2 years. The management of patients with specialty conditions is often a high-touch, interprofessional, and complex undertaking. Jobson Research, together with Pharm/alert Research, conducted a follow-up survey of community pharmacists regarding knowledge and perception of specialty products. The results of the survey show a need to foster innovation in care delivery between specialty pharmacists and community pharmacists. Legislative changes have created a sense of urgency, which can kindle the paradigm shift of community practice.
In 2016, specialty medications accounted for over 30% of the total prescription drug spend, and they are expected to represent 50% of total drugs sales by 2020.1 Mercer published a similar study predicting that between 40 and 50 new specialty drugs will hit the market each year for the next 5 years and that drug growth is not slowing down anytime soon, with 2,320 specialty products in active development.2
In May 2016, U.S. Pharmacist published an article titled “Specialty Pharmacy: What’s the Impact on Community Practice?”3 This analysis of community pharmacists’ perceptions of specialty medications investigated the burden of specialty medications on community pharmacists and how adequately prepared or knowledgeable they felt they were at managing the care of a specialty patient.3 A follow-up survey was conducted that is the basis for this analysis.
Specialty Pharmacy
Specialty pharmaceuticals are among the most valuable developments in healthcare, assisting patients with complex disease states. For example, in 2017, the FDA approved Spark Therapeutics’ Luxturna (voretigene neparvovec-rzyl), which cures a rare type of blindness at a cost of $850,000.4 The specialty niche in pharmacy is approximately 30% of all prescriptions sold and is expected to reach 50% over the next 3 years.5 By 2020, growth trajectories in specialty spending are estimated to quadruple, resulting in $400 billion annually.6
Specialty drugs treat a variety of medication conditions, including hepatitis C, rheumatoid arthritis, multiple sclerosis, and oncolytic disorders. Although they are often able to alter the prognosis, specialty drugs have a high price tag. Insurance and pharmaceutical companies define medications as “specialty” mainly by their cost, and Medicare classifies a specialty medication as costing more than $670 per month.7 According to IMS Health, these medications must meet at least five of the following eight criteria to be classified as specialty: biotech product, injectable formulation, part of the Risk Evaluation and Mitigation Strategy (REMS) program, treats a chronic condition, specialist-initiated, requires special handling, costs in excess of $6,000 per year, and has limited distribution.8
Specialty drugs are often more intricate than traditional medications owing to the narrow therapeutic window and cost, making it advisable for pharmacists to play a vital role in distribution and therapy management. Pharmacies must be held to a higher standard to maintain patient-centered care, given the continuous rise in specialty drugs being dispensed.
The use of specialty pharmaceutical products is often driven by big data. Value-based pharmacy management, or paying for medications based on outcomes, is the new avenue for reimbursement of specialty products.9 Manufacturers have to closely track real-world outcomes associated with specialty products to ensure positive outcomes and net financial benefit. This model of care is similar to the United Kingdom’s National Health Service medication payment based on quality-adjusted life year, wherein quality of life, life expectancy, and cost are combined and compared for coverage.10 Essentially, specialty pharmacies’ ability to track and report data will be the practice of the future.
One way to standardize the requirement for big data is through accreditation programs. Accreditation is defined as a process whereby a professional association or nongovernmental agency grants recognition for demonstrated ability to meet predetermined criteria for established standards.11 If accredited, a pharmacy is recognized as having a highly trained staff, pronounced expertise, and competent patient services.11 An accreditation in specialty pharmaceuticals provides a means for third-party payers to recognize a pharmacy for its unique capabilities.
Presently, it is practically mandatory for pharmacies dealing in specialty pharmaceuticals to be accredited in order to receive payer coverage, as well as authorization to distribute these products.12 Pharmacy benefit managers require specialty pharmacy accreditation for inclusion in their preferred networks.6 Industry and accreditation organizations have collaborated to ensure quality throughout all facets of specialized pharmacies.6
Currently, four accrediting bodies for specialty pharmacies exist: the Accreditation Commission for Health Care, the Community Health Accreditation program, the Center for Pharmacy Practice Acreditation, and the Utilization Review Accreditation Commission. These organizations observe the following standards, albeit with marginal differences: organization and administration, pharmacy operations, clinical management, and quality reporting and improvement.13
Community Pharmacy
As of 2015, over 67,750 community-based pharmacies are operating in the United States.14 This includes large chain retail stores, independently owned pharmacies, and pharmacies in supermarkets and mass merchandisers. In 2016, over 4.5 billion prescriptions were dispensed in the U.S.; many of these prescriptions were from community pharmacies.15
Community pharmacies are one of the few business models that have not undergone a major transformation in the past few decades. Most of the change has come through the advent of major community pharmacy chains or mergers, such as CVS Health and Walgreens Boots Alliance. However, a sense of urgency is starting to kindle advances in community pharmacy practice. For example, the completion rate of Comprehensive Medication Reviews was only 21%.16 Community pharmacists are capable of impacting patients through countless services that are often underutilized.
The future of community pharmacy lies with the transition to a service-based approach. Pharmacists are the most accessible healthcare professionals to the public. Limiting the practice of community pharmacists solely to dispensing is to limit the expansion of healthcare. Community-based practice is the epitome of platforms for offering quality healthcare intertwined with convenience. Various community-pharmacy organizations are starting to innovate practice—from proposed upward vertical mergers of CVS Health with Aetna to the creation of high-performing networks such as CPESN USA.
The community pharmacists of the future are focused on improving the lives of their patients, similar to the concept of value-based purchasing— reimbursement on service-based outcomes, not products. The paradigm shift is occurring through nondispensing services, such as chronic care management, administering immunizations, smoking-cessation education, home care services, hospice care, medication therapy management, diabetes self-management education, point-of-care testing, and weight management.17 Additionally, community pharmacists may play a role in the day-to-day management of patients with specialty medications through these services.
Results of the Survey
Jobson Research, together with Pharm/alert Research, conducted a quantitative online survey of 830 community pharmacists from across the country in 2017; this is almost double the number of responses in the previous survey.3 Each pharmacist was presented with a list of the top 10 leading specialty products as of June 2015 per Quintiles Health (previously IMS Health) and asked to think of those agents when answering five questions about specialty products. The 10 products pharmacists were asked to think of when considering specialty medications were the following: Harvoni (ledipasvir/sofosbuvir), Humira (adalimumab), Enbrel (etanercept), Sovaldi (sofosbuvir), Remicade (infliximab), Copaxone (glatiramer acetate), Neulasta (pegfilgrastim), Rituxan (rituximab), Tecfidera (dimethyl fumarate), and Avastin (bevacizumab).3
The demographics of the respondents may be found in TABLE 1. The pharmacists averaged 26.9 years in pharmacy practice, with a majority of the respondents identified as independent pharmacists. These pharmacies were located across the U.S., with larger proportions based in the following states: California (7.5%), New York (7.3%), Texas (6.7%), and Florida (6.5%).
TABLE 2 compares community pharmacists’ responses to the standardized questions in 2015 and 2017. The questions focused on awareness, significance, and counseling habits for specialty products in the community setting. A larger percentage—80.2% versus 70.3%—of pharmacists strongly agreed that they were generally aware of their patients’ specialty conditions. Additionally, the pharmacists felt a greater sense of importance that they were knowledgeable about these specialty medications—78.3% versus 71.4%—compared with the previous cohort.3 However, the frequency of pharmacists very often counseling patients decreased drastically to 13.4% in 2017 from 25.8% in 2015; pharmacists who identified as counseling “sometimes” decreased to 35.2% in 2017 from 47.2% in 2015.
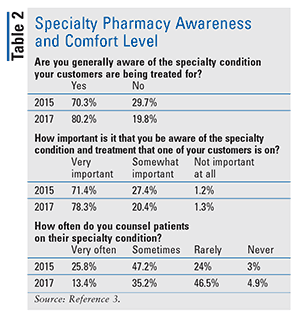
Based on these data, community pharmacists’ awareness of and reverence toward specialty products has increased; however, patient interventions have decreased. The data show the need for community pharmacists to be knowledgeable about specialty medications.
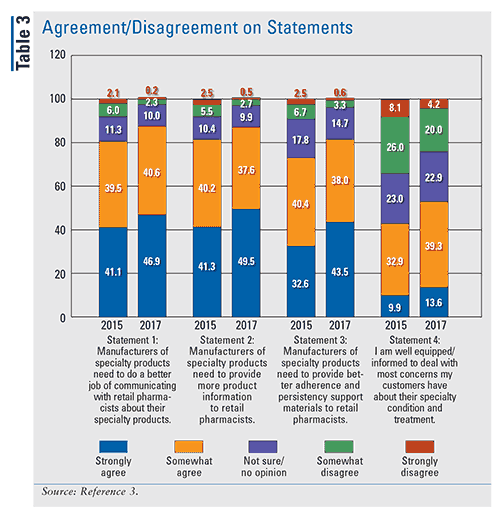
The second half of the survey focused on the needs of community pharmacists relative to product drug information through specialty product manufacturers. The survey reviews four core statements: communication, providing product information, provision of adherence and persistency reports, and pharmacist resources. The data are summarized in TABLE 3, with the 2015 data given as comparison. All statements increased in agreement in 2017 compared with 2015, and the data show there is still room for improvement. More pharmacists strongly agreed that they were well equipped to deal with most concerns of customers with specialty concerns (13.6% vs. 9.9%); however, nearly half (46.9%) strongly agreed with the need for manufacturers to do a better job of communicating product information to community pharmacists. Finally, community pharmacists increased in agreement (87.1% and 81.5%) on the need for product and adherence information to provide higher quality care.
The final survey question focused on primary concerns and specified resources to better help patients in the community setting. The primary concern for community pharmacists involved patients and patient education. The need for patient education resonated with community pharmacists in the needs they expressed: 71.6% of pharmacists stated there is value in continuing-education programs on specialty products, and 71.3% stated there is value in patient-education materials. Community pharmacists did not place much value on medical websites, portals dedicated to specific disease states, or the patient saving materials.
Community Pharmacy and Specialty Pharmacy 3.0
A significant takeaway from the survey is that community pharmacists have a high awareness of patients and specialty medical conditions, yet showed a minimal role in counseling and providing interventions. The answer may not be a misunderstanding of the pathophysiological conditions specialty pharmacy patients have, but rather a paucity of knowledge about the specialty products. The survey shows that 87.5% of community pharmacists agree with the need for manufacturers to provide more product information (87.1%).
One avenue to provide better patient care is through the juxtaposition of community pharmacy and specialty pharmacy. Community pharmacists are often the most accessible healthcare provider and are a part of the day-to-day lives of many patients. Moreover, specialty pharmacies are often accredited and equipped to handle the intricate needs of specialty medications.
Change-management thought leader John Kotter proposed eight steps for successful change management.18 The first step is creating a sense of urgency—over half of all organizational changes fail because of a lack of urgency. The Patient Protection and Affordable Care of Act of 2010 (ACA) and the Medicare Access and CHIP Reauthorization Act of 2015 (MACRA) have created a sense of urgency. Both the ACA and MACRA have pushed for quality of healthcare delivery, either through the establishment of a national quality strategy or, in the case of MACRA, by changing payment reform through merit-based incentive payment systems.
The changes implemented through ACA and MACRA allow specialty pharmacists to partner with community pharmacists, with the community pharmacist highlighting the problems found in day-to-day management and specialty pharmacists providing the solutions using quality metrics as drivers to change.
Conclusion
Community pharmacists and specialty pharmacists should accept the evolution in the outpatient medication market and identify opportunities to optimize patient care. This survey of community pharmacists has identified many gaps in the specialty products supply chain—specifically in the day-to-day management of individual patients. New legislative initiatives create a sense of urgency to allow pharmacists to change and rise up to this new challenge.
REFERENCES
1. McCann D. Specialty drug costs to soar again in 2018. CFO. www.cfo.com/health-benefits/2017/09/specialty-drug-costs-soar-2018/. September 2017. Accessed January 2, 2018.
2. Grover D. Health care costs expected to rise because of new specialty drugs. Huffington Post. www.huffingtonpost.com/entry/health-care-costs-expected-to-rise-because-of-new-specialty-drugs_us_59bfeac5e4b02da0e143c985. September 2017. Accessed January 2, 2018.
3. DeLoach LA, Leonard CE, Galdo JA. Specialty pharmacy: what’s the impact on community practice? US Pharm. 2016;1(5):35-40.
4. Tirrell M. A US drugmaker offers to cure rare blindness for $850,000. NBC. www.cnbc.com/2018/01/03/spark-therapeutics-luxturna-gene-therapy-will-cost-about-850000.html. January 2018. Accessed February 1, 2018.
5. Morgan K. Trends and challenges in specialty drugs. Drug Channels. www.drugchannels.net/2017/06/trends-and-challenges-in-specialty-drugs.html. June 2017. Accessed January 18, 2018.
6. The Management of Specialty Drugs. Washington, DC: Specialty Pharmaceutical Care Management Association. 2017.
7. Specialty medications. National Pharmaceutical Services. www.pti-nps.com/nps/index.php/specialty-medications/. 2016. Accessed January 18, 2018.
8. Duffant B, Garg A, Shah K. Overview of the specialty drug trend: succeeding in the rapidly changing U.S. specialty market. IMS Health. 2014. http://docplayer.net/4230764-Overview-of-the-specialty-drug-trend.html. Accessed January 18, 2018.
9. Paying for drugs based on outcomes. CVS Health. https://payorsolutions.cvshealth.com/insights/paying-for-drugs-based-on-outcomes. Accessed February 17, 2018.
10. Rawlins M. NICE: moving onward. N Engl J Med. 2013;369:1(July 4).
11. Millonig M. Pharmacy accreditation: is it worth the effort? Wolters Kluwer.
www.wolterskluwercdi.com/blog/pharmacy-accreditation-it-worth-effort/. 2017. Accessed January 18, 2018.
12. Steiber D. Specialty pharmacy: no longer optional. www.specialtypharmacytimes.com/publications/specialty-pharmacy-times/2016/april-2016/accreditation-in-specialty-pharmacy-no-longer-optional. 2016. Accessed January 18, 2018.
13. Fein A. Pharmacy accreditation boom continues. Drug Channels. www.drugchannels.net/2016/03/the-specialty-pharmacy-accreditation.html. 2016. Accessed January 18, 2018.
14. Qato DM, Zenk S, Wilder J, et al. The availability of pharmacies in the United States: 2007–2015. PLoS ONE. 2017;12(8): e0183172. https://doi.org/10.1371/journal.pone.0183172.
15. Total number of prescriptions dispensed in US. www.statista.com/statistics/238702/us-total-medical-prescriptions-issued/. Accessed February 17, 2018.
16. Quality measures for pharmacists. Pharmacist’s Letter/Prescriber’s Letter. January 2015.
17. NCPA Innovation Center. www.ncpanet.org/innovation-center/diversified-revenue-opportunities/specialty-pharmacy. Accessed January 18, 2018.
18. Kotter J. Leading change: why transformation efforts fail. HBR. June 1995.
To comment on this article, contact rdavidson@uspharmacist.com.





