US Pharm. 2011;36(9):HS-30-HS-34.
Chronic obstructive pulmonary disease (COPD) is a clinical syndrome characterized by airway obstruction and airflow limitation that interfere with normal breathing. Persons with COPD often have chronic bronchitis, which is defined as a productive cough lasting at least 3 months per year over 2 successive years. The lungs also typically display emphysema, a pattern of damage involving permanent enlargement of lung airspaces and/or destruction of the walls of the air sacs.1 Cigarette smoking—including exposure to secondhand smoke—is the primary cause of COPD.2 Other risk factors include genetic predisposition, indoor and outdoor pollution, occupational dusts and chemicals, and frequent lower respiratory infections during childhood.3
In 2008, more than 12 million U.S. adults aged 18 years and older were estimated to have COPD, and 24 million had some evidence of impaired lung function.1 COPD significantly limits physical exertion, prevents participation in social activities, and impairs the ability to work. In a report issued by the National Institutes of Health in 2011, patients with COPD had a discharge rate of 22.5 per 100,000, which equates to roughly 672,000 hospital discharges.4
COPD was recently recoded and reclassified under chronic lower respiratory diseases in the updated International Statistical Classification of Diseases and Related Health Problems.5 Current national statistics on death records indicate that COPD is the third leading cause of death in Americans, claiming more than 137,000 lives annually. COPD trails only heart disease and cancer as the leading cause of death in Americans.6 Without global intervention, deaths from COPD are projected to increase by more than 30% in the next 10 years.3 Chronic bronchitis and emphysema take a heavy toll on the economy. In 2010, the national annual cost of COPD was estimated at $49.9 billion by the National Heart, Lung, and Blood Institute.1
The Role of Gender
For years, COPD prevalence in the United States was higher in men than in women; however, a shift in the gender paradigm has occurred within the last three decades.6 Death rates for COPD declined overall among U.S. men between 1999 (57.0 per 100,000) and 2006 (46.4 per 100,000), whereas there was no significant change in death rates among women (35.3 per 100,000 in 1999 and 34.2 per 100,000 in 2006).6 Although U.S. age-adjusted COPD death rates for men were 1.3 times greater than rates for women, the overall number of women dying from COPD has surpassed the number of men owing to a higher percentage of women in the general U.S. population.1
COPD used to be more prevalent in men globally as well, but because of increased tobacco use among women in developed countries and the higher risk of exposure to indoor air pollution (such as solid fuel used for cooking and heating) in low-income countries, the disease now affects men and women almost equally worldwide.3
Several factors correlate to the gender shift. These include greater cigarette consumption in women, anatomical and hormonal disparities, differing levels of anxiety and depression, and prevalence of osteoporosis due to corticosteroid use, which more commonly occurs in female patients with COPD.
Tobacco Utilization
Smoking is the largest preventable contributor to COPD occurrence in the developed world; smokers are 12 times more likely than nonsmokers to die from the disease.7 In the early 20th century, significantly more men than women smoked cigarettes. This trend began to reverse when the tobacco industry started advertising cigarette smoking as a symbol of independence for women because men were fighting overseas. Cigarette consumption by women slowly increased in the second half of the 20th century, and by the 1980s nearly 25% of adult women in the U.S. were regular smokers.8 This smoking trend resulted in increased COPD hospitalizations and mortality for women in the early 21st century.
Smoking cessation is the most effective means of managing COPD.9 However, women face a number of psychological and physiological hindrances to their ability to quit smoking.10-12 Depression, the need for social support, and the fear of weight gain limit some women’s readiness to quit. Moreover, the potential for heightened sensitivity to nicotine and withdrawal symptoms and the timing of quit attempts relative to the menstrual cycle are physiological hurdles affecting some women’s success.13 Gender influences pharmacologic outcomes of smoking-cessation therapy, highlighting the need for deliberate selection of pharmacotherapy and counseling (TABLE 1).14
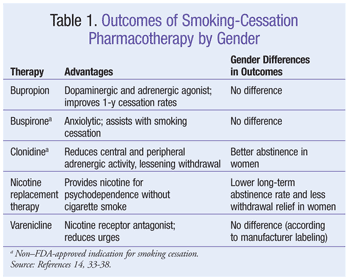
Anatomical Disparities
In contrast to the male lung, the anatomical structure of the female lung predisposes women to COPD. The “muscularity effect,” a phenomenon in which men have stronger respiratory muscles than women, leads to an inherent exchange of more air with each breath. As a result, women have a steeper increase in the ratio of residual volume to total lung capacity throughout aging.15 Additionally, adult female lungs generally are smaller and weigh less than adult male lungs, translating to smaller alveolar space and narrower airway diameter. Thus, as women age, they are more prone to the destructive effects of smoking and other noxious gases. A woman who smokes the same number of pack-years as a man is more negatively affected by concentrated tar in her smaller, less active airspace.16-18
Hormonal Disparities
Hormones play an important role in modulating inflammatory processes in the body. Chronic inflammation caused by neutrophil infiltration is the hallmark of airway destruction, leading to limitations in airflow. In laboratory studies, estrogen has been shown to delay neutrophil apoptosis and enhance degranulation of myeloperoxidase, elastase, and superoxide, leading to decreased bronchial elasticity and widespread inflammatory destruction.19,20 Clinically, estrogen enhances histamine release and testosterone possesses antihistamine properties.21 Bronchial hyperactivity in women increases throughout the menstrual cycle and peaks in the luteal and follicular phases, when female sex hormones are at their highest. This augments the correlation between the difference in sex hormones and inflammatory processes in the body.22
Anxiety and Depression
Multiple studies underscore the relationship between psychological impairment, reduction in health-related quality of life (HRQoL), repeated hospitalizations, and increased mortality in patients with COPD.23-26 A study comparing 202 COPD patients with 114 sex- and age-matched healthy controls found a higher prevalence of anxiety and depression (P <.001) in COPD patients versus controls (18.8% and 22.2% vs. 3.5% and 6.1%, respectively). Women experienced more episodes of dyspnea and symptoms that negatively affected their HRQoL. Women also had significantly higher levels of depression and anxiety in studies comparing gender differences in patients with COPD.26,27
In patients with COPD, there is a strong association between comorbid anxiety, depression, and poor health status and repeat hospitalizations.24-26 In a study by Mikkelsen et al, length of hospital stay doubled in patients with concomitant COPD and psychiatric impairment.25 Additionally, patients suffering from psychological comorbidities are more likely to feel unsupported by clinical staff and to have poor treatment adherence.26
The exact etiology of these comorbidities is inconclusive. Theories include disease severity, living alone, work status, and extent of disability.23,26-28 Psychiatric therapy may reduce hospitalizations, improve work status, and lead to improved pulmonary function.24,25 There are no definitive data on specific pharmacologic psychotherapy or its impact on physical lung function in COPD patients. However, women have more psychiatric comorbidities, thereby emphasizing the need for gender-specific consideration.
Corticosteroids
Corticosteroids, an essential component of COPD management, are administered systemically for acute exacerbations or inhaled as maintenance therapy. However, corticosteroids lower bone mineral density (BMD), increasing a patient’s susceptibility to osteoporosis and subsequent hip fractures.29 Kjensli et al found that 88 randomly selected patients with COPD and no indication of cancer, inflammatory bowel disease, hormone replacement, or bisphosphonate therapy had significantly lower BMD compared with non-COPD patients.30 Interestingly, BMD was inversely related to COPD severity.
Additional studies suggest that osteoporosis associated with COPD is caused by chronic corticosteroid utilization.31,32 In a study of more than 16,000 hip-fracture cases and almost 30,000 controls, the odds of hip fracture were an average of 1.26 times greater with use of an inhaled corticosteroid. Increased risk of hip fracture is proportional to the daily dose of inhaled corticosteroids.32 The greater risk of osteoporosis in women underscores the need for BMD monitoring, as well as calcium, vitamin D, or bisphosphonate therapy when indicated.
Conclusion
Distinctions exist between men and women with regard to cigarette consumption, anatomy, hormones, anxiety and depression, and the prevalence of osteoporosis from corticosteroid use. These factors emphasize the need for gender-specific considerations in patients with COPD. Further research is needed to support this area of practice; however, being aware of gender differences when treating patients with COPD is likely to impact outcomes.
REFERENCES
1. American Lung Association. Trends in COPD (chronic bronchitis and emphysema): morbidity and mortality. February 2010. www.lungusa.org/finding-cures/
2. U.S. Department of Health and Human Services. How Tobacco Smoke Causes Disease: The Biology and Behavioral Basis of Smoking-Attributable Disease: A Report of the Surgeon General. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2010.
3. World Health Organization. Chronic obstructive pulmonary disease (COPD). Fact Sheet No. 315. February 2011. www.who.int/mediacentre/
4. National Institutes of Health. Fact Sheet. Chronic obstructive pulmonary disease (COPD). http://report.nih.gov/
5. World Health Organization. International classification of diseases (ICD). www.who.int/classifications/
6. Kochanek KD, Xu J, Murphy SL, et al. Deaths: preliminary data for 2009. National Vital Statistics Reports. Vol. 59, No 4. Hyattsville, MD: National Center for Health Statistics; 2011.
7. U.S. Department of Health and Human Services. The health consequences of smoking: a report of the Surgeon General. www.surgeongeneral.gov/
8. Gritz ER. Cigarette smoking by adolescent females: implications for health and behavior. Women Health. 1984;9(2-3):103-115.
9. Connett JE, Murray RP, Buist AS, et al. Changes in smoking status affect women more than men: results of the Lung Health Study. Am J Epidemiol. 2003;157:973-979.
10. Chen Y, Horne SL, Dosman JA. Increased susceptibility to lung dysfunction in female smokers. Am Rev Respir Dis. 1991;143:1224-1230.
11. Langhammer A, Johnsen R, Gulsvik A, et al. Sex differences in lung vulnerability to tobacco smoking. Eur Respir J. 2003;21:1017-1023.
12. Prescott E, Bjerg AM, Andersen PK, et al. Gender difference in smoking effects on lung function and risk of hospitalization for COPD: results from a Danish longitudinal population study. Eur Respir J. 1997;10:822-827.
13. Gritz ER, Nielsen IR, Brooks LA. Smoking cessation and gender: the influence of physiological, psychological, and behavioral factors. J Am Med Womens Assoc. 1996;51:35-42.
14. Perkins KA. Smoking cessation in women. Special considerations. CNS Drugs. 2001;15:391-411.
15. Hibbert M, Lannigan A, Raven J, et al. Gender differences in lung growth. Pediatr Pulmonol. 1995;19:129-134.
16. Chapman KR. Chronic obstructive pulmonary disease: are women more susceptible than men? Clin Chest Med. 2004;25:331-341.
17. Carter R, Nicotra B, Huber G. Differing effects of airway obstruction on physical work capacity and ventilation in men and women with COPD. Chest. 1994;106:1730-1739.
18. Silverman EK, Weiss ST, Drazen JM, et al. Gender-related differences in severe, early-onset chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2000;162:2152-2158.
19. Chiang K, Parthasarathy S, Santanam N. Estrogen, neutrophils and oxidation. Life Sci. 2004;75:2425-2438.
20. Molloy EJ, O’Neill AJ, Grantham JJ, et al. Sex-specific alterations in neutrophil apoptosis: the role of estradiol and progesterone. Blood. 2003;102:2653-2659.
21. Osman M. Therapeutic implications of sex differences in asthma and atopy. Arch Dis Child. 2003;88:587-590.
22. Tan KS, McFarlane LC, Lipworth BJ. Modulation of airway reactivity and peak flow variability in asthmatics receiving the oral contraceptive pill. Am J Respir Crit Care Med. 1997;155:4:1273-1277.
23. Balcells E, Gea J, Ferrar J, et al. Factors affecting the relationship between psychological status and quality of life in COPD patients. Health Qual Life Outcomes. 2010;8:108.
24. Yohannes AM, Baldwin RC, Connolly MJ. Depression and anxiety in elderly outpatients with chronic obstructive pulmonary disease: prevalence, and validation of the BASDEC screening questionnaire. Int J Geriatr Psychiatry. 2000;15:1090-1096.
25. Mikkelsen RL, Middelboe T, Pisinger C, Stage KB. Anxiety and depression in patients with chronic obstructive pulmonary disease (COPD). A review. Nord J Psychiatry. 2004;58:65-70.
26. Gudmundsson G, Gislason T, Janson C, et al. Depression, anxiety and health status after hospitalisation for COPD: a multicentre study in the Nordic countries. Respir Med. 2006;100:87-93.
27. Di Marco F, Verga M, Reggente M, et al. Anxiety and depression in COPD patients: the roles of gender and disease severity. Respir Med. 2006;100:1767-1774.
28. Katz PP, Julian LJ, Omachi TA, et al. The impact of disability on depression among individuals with COPD. Chest. 2010;137:838-845.
29. Katsura H, Kida K. A comparison of bone mineral density in elderly female patients with COPD and bronchial asthma. Chest. 2002;122:1949-1955.
30. Kjensli A, Mowinckel P, Ryg MS, Falch JA. Low bone mineral density is related to severity of chronic obstructive pulmonary disease. Bone. 2007;40:493-497.
31. Shane E, Silverberg SJ, Donovan D, et al. Osteoporosis in lung transplantation candidates with end-stage pulmonary disease. Am J Med. 1996;101:262-269.
32. Hubbard RB, Smith CJ, Smeeth L, et al. Inhaled corticosteroids and hip fracture: a population-based case-control study. Am J Respir Crit Care Med. 2002;166:1563-1566.
33. Hatsukami D, Skoog K, Allen S, Bliss R. Gender and the effects of different doses of nicotine gum on tobacco withdrawal symptoms. Exp Clin Psychopharmacol. 1995;3:163-173.
34. Wetter DW, Fiore MC, Young TB, et al. Gender differences in response to nicotine replacement therapy: objective and subjective indices of tobacco withdrawal. Exp Clin Psychopharmacol. 1999;7:135-144.
35. Hurt RD, Sachs DP, Glover ED, et al. A comparison of sustained-release bupropion and placebo for smoking cessation. N Engl J Med. 1997;337:1195-1202.
36. Chantix (varenicline) package insert. New York, NY: Pfizer Inc; December 2010.
37. Cinciripini PM, Lapitsky L, Seay S, et al. A placebo-controlled evaluation of the effects of buspirone on smoking cessation: differences between high- and low-anxiety smokers. J Clin Psychopharmacol. 1995;15:182-191.
38. Covey LS, Glassman AN. A meta-analysis of double-blind placebo-controlled trials of clonidine for smoking cessation. Br J Addict. 1991;86:991-998.
To comment on this article, contact rdavidson@uspharmacist.com.






