US Pharm. 2013;38(4):23-26.
ABSTRACT: Foot infections are a common and serious complication of diabetes. While gram-positive cocci—particularly staphylococci and streptococci—are the most common causes of mild-to-moderate infections, mixed gram-positive cocci and gram-negative bacilli with or without anaerobic organisms tend to cause chronic infections. Mild infections should be treated on an outpatient basis with oral antibiotics directed against staphylococci and streptococci, and severe infections should be treated initially with broad-spectrum parenteral antibiotics on an inpatient basis, followed by oral antibiotics when possible. Pharmacists play an important role in educating health care professionals and patients about the proper treatment and prevention of diabetic foot infections.
Foot infection, a common and serious complication of diabetes, increases the risk of hospitalization, amputation, and death. According to the CDC, 25.8 million Americans have diabetes, and these patients have up to a 3% annual risk and a 25% lifetime risk of developing a foot ulcer.1,2 In 2007, hospitalization for ulcer, inflammation, and/or infection as a primary diagnosis was 5.7 per 1,000 diabetic patients.1 Diabetic ulceration is the primary cause of 85% of all lower-extremity amputations.2,3 Mortality following amputation ranges from 13% to 67% after 1 year, 35% to 65% after 3 years, and 31% to 39% after 5 years.4,5 Given this increase in morbidity and mortality, the treatment and prevention of diabetic foot infections (DFI) are key. It is estimated that 40% of amputations could be prevented with appropriate wound care.2 The Infectious Diseases Society of America (IDSA) updated its guidelines for the treatment of DFIs in 2012.6 This review is intended to educate pharmacists about the proper management of DFIs according to current guidelines.
Etiology and Risk Factors
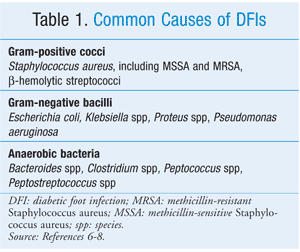
DFIs may be caused by a number of infecting bacteria, including gram-positive cocci (GPC), gram-negative bacilli (GNB), and anaerobic organisms.7 GPC, particularly Staphylococcus aureus and beta-hemolytic streptococci, are the most common infecting organisms in patients with mild-to-moderate DFIs and patients who did not receive antibiotics in the previous month.6-8 Patients who experience chronic infections and those who have undergone recent antibiotic therapy tend to develop mixed infections of GPC and GNB with or without anaerobic organisms.6-8 The presence of obligate anaerobes is associated with necrotic, gangrenous, or ischemic tissue, and this is usually the case in chronic and severe infections.6-8 Infection with drug-resistant organisms like methicillin-resistant S aureus usually occurs in patients exposed to the health care system and in communities with a high prevalence of drug resistance.6-8 Pseudomonas aeruginosa infections typically are associated with warm climate and exposure to water.6-8 Types of infecting organisms are listed in TABLE 1.6-8
Although any wound, ulcer, or traumatic injury involving the foot may lead to infection, certain factors increase the risk at least twofold.9 In a 2-year prospective study of 1,666 diabetic patients at an outpatient clinic, the following risk factors were associated with DFI development: wound depth to bone, wound duration exceeding 30 days, recurrent foot wound, traumatic wound etiology, and peripheral vascular disease (PVD).9 In addition to these risk factors, the IDSA guidelines include previous lower-extremity amputation, loss of protective sensation, renal insufficiency, and a history of walking barefoot.6 A DFI that extends to the leg can prolong healing time and increase the risk of amputation.10 In a retrospective case-control study of 48 diabetic patients with DFI, risk factors associated with extension of the infection into the leg were previous toe amputation, presence of systemic signs of infection, necrosis, large size of primary wound, ulcer on the heel, and advanced Wagner and infection grades.10
Pathophysiology
Although DFIs can develop in the absence of an ulcer or traumatic injury, most cases begin with a wound.8,11 Typically, these wounds begin superficially but spread to deeper structures, potentially leading to deep abscesses, cellulitis, chronic ulcers, and osteomyelitis (OM).8,11 Neuropathy, ischemia from peripheral arterial disease (PAD), and diminished immunity put diabetic patients at greater risk for developing DFIs.2,8 Neuropathy affects sensory, motor, and autonomic innervation. Often, loss of sensation causes wounds to go unnoticed. The injury becomes progressively worse from weight-bearing pressure. Damage to the motor nerves and the foot muscles causes an imbalance in flexion and extension, leading to foot deformities and abnormal pressure points, which contribute to skin breakdown and ulceration. Autonomic neuropathy results in decreased functioning of sweat and oil glands, which makes the skin dry and more susceptible to tearing and infection.2,11
PAD is another contributor to the development of foot ulcers. Long-term hyperglycemia causes endothelial and smooth-muscle cell dysfunction in the peripheral arteries. The breakdown of glucose also depletes nicotinamide adenine dinucleotide phosphate, which normally detoxifies reactive oxygen species and aids in the production of the vasodilator nitric oxide. Smoking, hypertension, and hyperlipidemia are other factors that contribute to the development and worsening of existing PAD. This increase in oxidative stress and vasoconstriction promotes poor healing, ischemia, and cell death and further contributes to the development of DFI.2 The resulting angiopathy, low oxygen environment, and chronic hyperglycemia decrease cell-mediated immunity, rendering diabetic patients more susceptible to infection.11
Clinical Presentation and Diagnostic Considerations
Patients may present with classic signs and symptoms of inflammation (erythema, warmth, tenderness, pain, or induration) and with purulent, possibly foul-smelling, discharge from an ulcer.8,11 It is important to note that pain and tenderness may be reduced in patients with neuropathy and that erythema may be decreased in patients with vascular disease.8,11 Most patients with DFIs do not have systemic signs of infection (e.g., fever, chills); however, the presence of these signs indicates a severe and deep infection.8,11
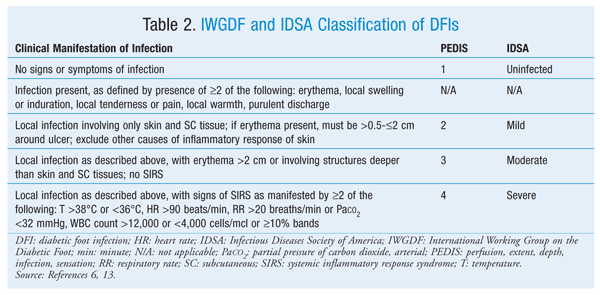
In DFI diagnosis, it is important to establish the extent of the infection. TABLE 2 illustrates the two commonly used and validated systems for wound classification: PEDIS (Perfusion, Extent/size, Depth/tissue loss, Infection, Sensation)—established by the International Working Group on the Diabetic Foot—and IDSA.6,12,13 For clinically uninfected wounds, the IDSA guidelines do not recommend that a specimen be collected for culture.6 For infected wounds, however, the guidelines generally recommend that appropriate specimens be obtained before empiric therapy is initiated. Wounds should be properly cleaned and debrided before specimens are obtained, and specimens should preferably be collected from deep tissue or by bone biopsy.6
Plain radiographs to assess bone abnormalities and soft-tissue gas are recommended for all patients presenting with a new DFI.6 MRI is indicated if radiography is inconclusive and OM is suspected. OM is a serious complication, and diagnosis may be difficult. Laboratory studies, such as WBC count and erythrocyte sedimentation rate, have little predictive value. Visible bone and palpable bone detected by probe-to-bone testing are suggestive of underlying OM in patients with DFI.6,8,14 The probe-to-bone test has low positive predictive value, but a negative test may exclude a diagnosis of OM.14 Imaging studies and radionuclide scans are more accurate, but confirmation requires a bone culture if imaging is inconclusive.6
Treatment
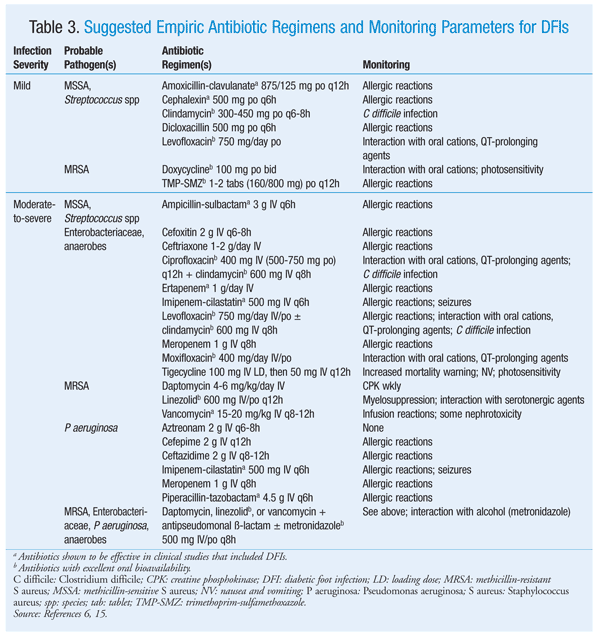
To manage DFIs, the IDSA guidelines recommend a multidisciplinary diabetic foot-care team consisting of infectious disease specialists, clinical microbiology specialists, foot surgeons, and wound-care specialists.6 Empiric antibiotic treatment depends upon the severity of the infection (TABLE 3).6,15 Uninfected wounds should not be treated with topical or systemic antibiotics, and mild infections should be treated with oral antibiotics on an outpatient basis for 1 to 2 weeks.6 Moderate infections should be treated initially with oral antibiotics or with IV antibiotics, followed by oral therapy for 1 to 3 weeks.6 Severe infections require treatment with IV antibiotics in an inpatient setting followed by oral therapy, when possible, for a total of 2 to 4 weeks.6
Patients with complicating features, such as severe PAD, lack of home support, or nonadherence, should be hospitalized for the entire course of therapy.6 Patients with OM require a longer therapy duration: 4 to 6 weeks in the case of residual infected viable bones, and more than 3 months in the case of residual dead bones postoperatively.6 Clinicians should streamline antibiotic therapy based upon appropriately obtained culture and sensitivity reports.6
All DFI patients should receive wound care, which may consist of incision and drainage to decrease bacterial load, debridement to remove necrotic tissues, offloading to redistribute pressure off the wound, and appropriate dressings (TABLE 4) to allow for optimal wound healing.6 Finally, hyperbaric oxygen therapy, platelet-derived growth factors, granulocyte colony-stimulating factors, bioengineered skin equivalents, and topical negative pressure may be considered as adjunctive treatment in certain settings to further promote wound healing.6

Prevention
Comprehensive foot-care programs that include risk assessment, education, treatment of foot problems, and appropriate referrals can reduce amputation rates by 45% to 85%.1 In order to reduce contributing factors to DFI, it is recommended to use monofilament testing to screen for neuropathy and to use the ankle-brachial index to screen for PAD.4 Studies have shown that improved glycemic and blood pressure (BP) control prevent microvascular complications and lessen the risk of infection. In fact, for every percentage-point drop in hemoglobin A1C, microvascular complications are reduced by 40%, and for every 10-mmHg reduction in systolic BP, any complication related to diabetes is reduced by 12%.1
The Pharmacist’s Role
As the most accessible health care professional, the pharmacist occupies a prime position in helping prevent DFI. Pharmacists should frequently remind their diabetic patients to wear socks and properly fitting closed-toe footwear, to moisturize and visually inspect their feet nightly, and to see a physician for evaluation of cracks, sores, and other injuries. Pharmacists should empower patients with diabetes to achieve optimal glucose and BP control through individualized diet and exercise and medication adherence, since worsening control contributes to the development of neuropathy, PVD, and infections. Once a foot infection develops, the pharmacist should engage with other health care professionals to select the most appropriate antimicrobial regimen for the patient, including dosage, route of administration, frequency, and duration of therapy. The pharmacist is also well positioned to assist in monitoring the efficacy and safety of the patient’s prescribed antibiotic regimen.
REFERENCES
1. CDC. National Diabetes Fact Sheet: National Estimates and General Information on Diabetes and Prediabetes in the United States, 2011. Atlanta, GA: U.S. Department of Health and Human Services, CDC; 2011.
2. Clayton W Jr, Elasy TA. A review of the pathophysiology, classification, and treatment of foot ulcers in diabetic patients. Clin Diabetes. 2009;27:52-58.
3. CDC. Diabetes data & trends. Age-adjusted hospital discharge
rates for peripheral arterial disease (PAD),
ulcer/inflammation/infection (ULCER), or neuropathy as first-listed
diagnosis per 1,000 diabetic population, United States, 1988–2007.
www.cdc.gov/diabetes/statistics/hosplea/diabetes_complications/fig2_pop.htm.
Accessed November 27, 2012.
4. Singh N, Armstrong DG, Lipsky BA. Preventing foot ulcers in patients with diabetes. JAMA. 2005;293:217-228.
5. Schofield CJ, Libby G, Brennan GM, et al. Mortality and
hospitalization in patients after amputation: a comparison between
patients with and without diabetes. Diabetes Care. 2006;29:2252-2256.
6. Lipsky BA, Berendt AR, Cornia PB, et al. 2012 Infectious Diseases
Society of America clinical practice guideline for the diagnosis and
treatment of diabetic foot infections. Clin Infect Dis. 2012;54:e132-e173.
7. Lipsky BA. Empirical therapy for diabetic foot infections: are there clinical clues to guide antibiotic selection? Clin Microbiol Infect. 2007;13:351-353.
8. Bader MS. Diabetic foot infection. Am Fam Physician. 2008;78:71-79.
9. Lavery LA, Armstrong DG, Wunderlich RP, et al. Risk factors for foot infections in individuals with diabetes. Diabetes Care. 2006;29:1288-1293.
10. Adam KM, Mahmoud SM, Mahadi SI, et al. Extended leg infection of diabetic foot ulcers: risk factors and outcome. J Wound Care. 2011;20:440-444.
11. Fish DN, Pendland SL, Danziger LH. Skin and soft-tissue infections. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 8th ed. New York, NY: McGraw-Hill Medical; 2011:1893-1912.
12. International Working Group on the Diabetic Foot. Ulcer
classification.
www.iwgdf.org/index.php?option=com_content&task=view&id=84&Itemid=116.
Accessed November 27, 2012.
13. Bakker K, Apelqvist J, Schaper NC; International Working Group on
Diabetic Foot Editorial Board. Practical guidelines on the management
and prevention of the diabetic foot 2011. Diabetes Metab Res Rev. 2012;28(suppl 1):225-231.
14. Lavery LA, Armstrong DG, Peters EJ, Lipsky BA. Probe-to-bone test
for diagnosing diabetic foot osteomyelitis: reliable or relic? Diabetes Care. 2007;30:270-274.
15. Lexi-Comp Online [Internet database]. Hudson, OH: Lexi-Comp, Inc; 2012. www.lexi.com. Accessed November 27, 2012.
To comment on this article, contact rdavidson@uspharmacist.com.





