US Pharm.
2007;32(10):HS-24-HS-HS-32.
Currently, there are 20.8 million people, or
7% of the U.S. population, with diabetes. By 2050, this number is projected to
more than double to 48.3 million.1 The majority of these patients
are diagnosed with type 2 diabetes, which is characterized by insulin
resistance and diminished beta-cell function.2 Lifestyle
modifications, including medical nutrition therapy, weight management, and
physical activity, are the foundations of treatment. In addition to lifestyle
interventions, there are now nine classes of antihyperglycemic agents.
Sitagliptin (Januvia), the first in a new
class of antihyperglycemic agents, was approved in October 2006. It inhibits
the enzyme dipeptidyl peptidase-4 (DPP-4), which causes the degradation of
glucagon-like peptide-1 (GLP-1). The only other agent on the market that
affects levels of GLP-1 is exen atide (Byetta). Exenatide is an
injectable GLP-1 agonist, which mimics the action of naturally occurring
GLP-1. Indications for sitagliptin include use in type 2 diabetes as
monotherapy or in combination with metformin or thiazolidinediones.3
Clinical Effects
The two incretin hormones that
have been identified are glucose-dependent insulinotropic polypeptide (GIP)
and GLP-1.4 Incretin hormones are released by cells in the
gastrointestinal tract in response to food, and they stimulate insulin
secretion to maintain glucose homeostasis. Research shows a 15% reduction in
postprandial levels of GLP-1 in patients with type 2 diabetes.5
Circulating levels of GIP are near normal in type 2 diabetes, but the peptide
has decreased ability to stimulate insulin secretion levels. The problem is
that GLP-1 is rapidly inactivated (half-life of approximately one minute) by
the enzyme DPP-4.6 Inhibiting the DPP-4 enzyme enhances the action
of GLP-1 by prolonging the half-life and increasing circulating GLP-1. GLP-1
causes pancreatic beta-cells to increase insulin secretion when glucose levels
are elevated due to a meal; therefore, hypoglycemia is less likely.4
DPP-4 inhibitors have actions similar to
those of the GLP-1 agonist, such as stimulation of insulin secretion,
inhibition of glucagon secretion, and preservation of beta-cell function.4
In contrast, DPP-4 inhibitors do not slow gastric emptying or cause weight
loss, possibly due to lower concentrations of GLP-1 postprandially compared to
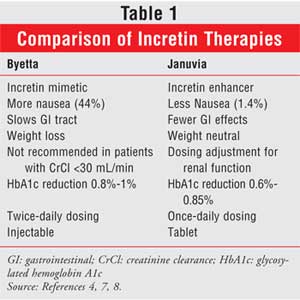
the GLP-1 agonist (see Table 1). DPP-4 inhibitors raise incretin levels
to normal or slightly above normal levels.7 The advantage of DPP-4
inhibitors is oral administration; the GLP-1 receptor agonist is injectable.
Adverse Effects
The most common adverse effects
are upper respiratory tract infection, nasopharyngitis, and headache.
Nasopharyngitis was reported in 5.2% of patients taking sitagliptin 100 mg as
monotherapy versus 3.3% of patients taking placebo.3 When
sitagliptin plus pioglitazone was compared with pioglitazone plus placebo, the
most common side effects reported included upper respiratory tract
infection (6.3% vs. 3.4%, respectively) and headache (5.1% vs. 3.9%,
respectively).9 The mechanism for the nasopharyngitis is unknown,
but the intensity, duration, and recurrence rate was the same as the placebo
group, and this did not cause discontinuation from the clinical trial.3
The DPP-4 enzyme is also found in other tissues, including those from the
liver, kidneys, pancreatic duct, and lymphocytes.4 Interestingly,
early trials suggested DPP-4 may affect the immune system due to inhibition of
T-cell activity in vitro.11 It was later determined that higher
concentrations were necessary to produce the same effect in vivo. Studies have
shown that sitagliptin is a selective and potent inhibitor (greater than 80%)
of the DPP-4 enzyme.3,10 Selectivity is important, because
inhibition of two other enzymes, DPP-8 and -9--both in the same family as
DPP-4--resulted in serious toxicities and impaired T-cell activation.
6,7 Long-term trials evaluating the effects on the immune system may be
warranted owing to increased reports of nasopharyngitis, urinary tract
infection, and small elevations in white blood cell count.12-14 The
overall incidence of hypoglycemia and gastrointestinal adverse events in
patients taking sitagliptin 100 mg was similar to that of placebo.10
Drug Interactions
Sitagliptin does not inhibit or
activate the cytochrome P-450 enzyme system, but it is a P-glycoprotein
substrate.7 After 10 days, sitagliptin 100 mg increased the area
under the curve (11%) and the mean peak drug concentration (Cmax,
18%) of digoxin.3 Therefore, patients receiving digoxin should be
monitored when starting sitagliptin.
Dosage
The recommended dosage of
sitagliptin is 100 mg once daily with or without food. The majority of
sitagliptin (79%) is excreted unchanged in the urine. For patients with
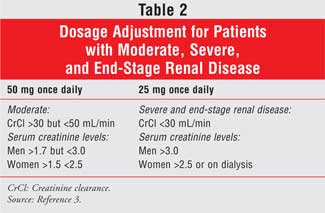
moderate, severe, and end-stage renal disease, a dosage adjustment is required
(see Table 2).
Sitagliptin is available in 25-, 50-, and
100-mg tablets. The different strengths are available for dosage adjustment in
renal insufficiency and should not be used for dosage titration.
Place In Therapy
In type 2 diabetes, there is a
progressive loss of beta-cell mass and function. GLP-1 increases endogenous
insulin secretion, which supports the use of DPP-4 inhibitors early in the
disease process.
To date, there are few head-to-head trials
comparing DPP-4 inhibitors to other antihyperglycemic agents. A noninferiority
trial compared sitagliptin to glipizide as add-on therapy in 1,172 patients
inadequately controlled using metformin alone. After 52 weeks of treatment,
the change in the glycosylated hemoglobin A1c (HbA1c) level from baseline was
–0.67% for both groups. There was more hypoglycemia and weight gain (+1.1 kg)
in the glipizide group, whereas the sitagliptin group had weight loss (-1.5
kg).12
Sitagliptin is not as potent in lowering
HbA1c levels (0.6%-0.85%) as metformin (1%-2%).15 However, the
recent FDA approval of sitagliptin/metformin (Janumet) may make sitagliptin a
more reasonable first-line choice. The combination of sitagliptin and
metformin is indicated in patients who are not adequately controlled on
metformin or sitagliptin alone or in patients already taking both agents.
16 This approval was based on a randomized, double-blind,
placebo-controlled trial of 701 patients inadequately controlled on metformin
alone. The average duration of diabetes was 6.2 years, and patients were on at
least 1,500 mg of metformin with a mean HbA1c of 8%. The HbA1c level decreased
by an additional 0.7% using sitagliptin. Fasting plasma glucose decreased by
16.2 mg/dL, compared to 9.0 mg/dL with placebo. Two-hour postprandial glucose
decreased by 61.3 mg/dL using the combination drug versus 10.8 mg/dL with
placebo. Most impressive was that more patients taking the combination drug
(47%) reached an HbA1c level of less than 7% during the 24 week study than did
those taking placebo (18%).14 Pairing sitagliptin with metformin is
a good combination, because metformin is commonly used as initial therapy due
to the absence of weight gain and hypoglycemia.
Most patients will require more than one
agent to control diabetes. Many of the antihyperglycemic therapies currently
available are associated with weight gain (i.e., sulfonylureas,
nonsulfonylureas, thiazolidinediones, and insulin). The DPP-4 inhibitors offer
the advantage of no weight gain.7,11 It is unknown if DPP-4
inhibitors can prevent weight gain in the long term. DPP-4 inhibitors may be a
treatment option for patients in whom metformin is contraindicated due to
decreased renal function or intolerable gastrointestinal side effects. The
biggest disadvantage is cost, since sitagliptin is approximately three times
more expensive than metformin and about ten times more expensive than the
sulfonylureas.17
Long-term effects of GLP-1 in animal studies
demonstrated increases in beta-cell mass and beta-cell efficiency.4
Can these agents that increase the levels of GLP-1 alter the course of type 2
diabetes in humans? Some of the clinical studies did document improvement in
the markers of alpha- and beta-cell function (glucagon suppression, indices of
insulin secretion, measures of beta-cell sensitivity, proinsulin–insulin
ratios, and Hemeostatis Model Assessment of beta cell function [HOMA-beta]) in
patients taking a DPP-4 inhibitor.7 However, long-term studies are
needed to confirm these findings and document a delay in disease progression.
The mechanism of action of the DPP-4
inhibitors raises the question: Will these agents be effective in patients who
have had diabetes for 10 to 15 years and who may no longer have any beta-cell
function? In clinical practice, sitagliptin is often used as add-on therapy to
delay insulin therapy. The fasting and postprandial reductions are not as
impressive. To date, there are no trials assessing patients who have had
diabetes for more than 6.2 years.14 Long-term studies and
postmarketing data may help answer this question.
HbA1cGoals
The United Kingdom Prospective Diabetes Study
demonstrated that for every percentage point reduction in HbA1c, there was a
35% reduction in the risk of complications in patients with type 2 diabetes.
18 For type 1 diabetes, the Diabetes Complications and Control Trial
showed that complications--including retinopathy, nephropathy and
neuropathy--can be delayed or prevented with intensive insulin therapy.
19 These large, controlled clinical trials have been the basis for
recommendations aimed at preventing long-term complications. Extensive
research supports tighter glucose control to reduce complications. Despite our
best efforts, the treatment of diabetes remains suboptimal. The National
Health and Nutrition Examination Survey 2001–2002 sample data indicate that
only half (49.8%) of diabetic adults aged 18 years or older met the
recommended HbA1c level of less than 7%.20
It is important to remember that both
fasting and postprandial glucose contribute to HbA1c. In patients with
near-normal fasting glucose who are unable to reach an HbA1c level of less
than 7%, postprandial glucose should be the target.21 Fasting
hyperglycemia is the dominant contributor when the HbA1c level rises above
8.4%.22 Clinical trials for sitagliptin reflect a more dramatic
reduction in postprandial glucose (–49 to –62 mg/dL) compared to fasting
glucose (–10 to –25 mg/dL).7,10
The American Diabetes Association (ADA)
continues to recommend an HbA1c level of less than 7%, but in 2006, it stated
that the HbA1c level should be "as close to normal (<6%) as possible without
significant hypoglycemia."23 On the other hand, the American
Association of Clinical Endocrinologists (AACE) recommends an HbA1c level of
less than 6.5%.24 Recommendations for glycemic control from both
organizations are listed in Table 3. Both the ADA and the AACE agree
that the HbA1c goals should be individualized, but despite all the treatment
options available, good glycemic control is not always attainable.

Barriers
Glycemic control in patients with type 2 diabetes
remains poor despite the use of old and new medications available for the
treatment of diabetes. There are numerous factors that affect adherence,
including patient and/or physician resistance to intensifying treatment
regimens, financial burden, and the complexity of the medication regimen.
25 Many times, pharmacotherapy is postponed at the time of diagnosis in
the hopes that diet and exercise therapy will be successful. A retrospective
analysis of 9,416 patients over a three-year period showed the mean HbA1c
level was 8.4% when antihyperglycemic therapy was started.26 The
majority (67%) of the patients reviewed had a mean baseline HbA1c of 9.5% when
therapy was initiated, which supports the theory of lack of action in response
to abnormal findings. Infrequent monitoring of HbA1c was another reason
identified for not intensifying treatment regimens. Data showed that patients
had, on average, one HbA1c test annually.26 The ADA recommends
monitoring HbA1c levels twice a year for patients at goal and four times a
year for patients not at goal.2 Data show that more than 50% of
patients started on monotherapy will require a second medication to control
diabetes after three years.27 Also, studies have concluded that it
is very difficult and expensive to treat patients to an HbA1c level of less
than 7%.28 The combination of multiple medications and/or the
financial burden may be too much for some patients.
Other reasons associated with poor glycemic
control include physicians' lack of knowledge about diabetes treatment
guidelines or their choice not to follow them.
What's Next?
Development of new therapies for
the treatment of diabetes continues to focus on enhancing the positive effects
of GLP-1. In February 2007, Novartis received an approval letter from the FDA
for vildagliptin (Galvus), another DPP-4 inhibitor.29 One study of
interest looked at vildagliptin in patients with type 2 diabetes who required
insulin. Findings revealed that patients were able to take lower dosages of
insuline and had fewer episodes of hypoglycemia.30 The side-effect
profile of vildagliptin is similar to that of sitagliptin. In addition, the
rate of hypoglycemia in patients taking vildagliptin is similar to that of
patients taking placebo. There is no anticipated dosage adjustment for renal
insufficiency.30 So far, the studies show a mixed effect on
cholesterol, with one study showing a small increase in total cholesterol and
another study producing a decrease in HDL cholesterol.29 Two other
DPP-4 inhibitors, saxagliptin and denagliptin, are in development.31
Currently, clinical trials are evaluating
the effects of a long-acting-release form of exenatide. Preliminary data show
greater reductions in fasting glucose and HbA1c with a weekly injection.31
Lastly, a longer-acting synthetic derivative
of GLP-1, liraglutide, is in the pipeline. Liraglutide is more like human
GLP-1 than exenatide and is injected once daily. Disadvantages, compared to
sitagliptin, include an injectable dosage form and gastrointestinal side
effects. The most frequent side effects are nausea and diarrhea. Liraglutide
increases insulin secretion, suppresses postprandial glucagon secretion, and
decreases weight and HbA1c.32
Summary
DPP-4 inhibitors may have the
greatest impact in patients who are early in the disease process. Sitagliptin
is another weapon in the arsenal of oral antihyperglycemic agents; however,
whether it will be enough to get more patients to reach their goal HbA1c level
or whether it will simply delay the start of insulin therapy remains in
question. The pharmacist can have a key role in educating patients and
providers about the importance of regular diabetes follow-up, home glucose
monitoring, frequent HbA1c testing, and why multiple medications are needed.
The ultimate objective is to increase the number of people with diabetes with
an HbA1c level of less than 7%, but it will take a team effort.
References
1. Narayan KM, Boyle JP, Geiss MA, et al. Impact of recent increase in incidence on future diabetes burden, U.S.: 2005-2050. Diabetes Care. 2006;29:2114-2116.
2. American Diabetes Association. Standards of medical care in diabetes--2007 [position statement]. Diabetes Care . 2007;30:S4-S41.
3. Januvia (sitagliptin) [package insert]. North Wales, PA: Merck & Co., Inc.; 2007.
4. Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet. 2006;368:1696-1705.
5. Meier JJ, Nauck MA. Incretins and the development of type 2 diabetes. Curr Diab Rep. 2006;6:194-201.
6. Lankas GR, Leiting B, Roy RS, et al. Dipeptidyl peptidase IV inhibition for the treatment of type 2 diabetes: potential importance of selectivity over dipeptidyl peptidases 8 and 9. Diabetes. 2005;54:2988-2994.
7. Deacon CF. Dipeptidyl peptidase 4 inhibition with sitagliptin: a new therapy for type 2 diabetes. Expert Opin Investig Drugs. 2007;16:533-545
8. Byetta (exenatide) [package insert]. San Diego, CA: Amylin Pharmaceuticals; 2007.
9. Rosenstock J, Brazg R, Andryuk PJ, et al. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin added to ongoing pioglitazone therapy in patients with type 2 diabetes: a 24-week, multicenter, randomized, double-blind, placebo-controlled parallel-group study. Clin Ther. 2006;28:1556-1568.
10. Herman GA, Stein PP, Thornberry NA, Wagner JA. Dipeptidyl peptidase-4 inhibitors for the treatment of type 2 diabetes: focus on sitagliptin. Clin Pharmacol Ther. 2007;81:761-767.
11. Miller S, St Onge EL. Sitagliptin: a dipeptidyl peptidase IV inhibitor for the treatment of type 2 diabetes. Ann Pharmacother. 2006;40:1336-1343.
12. Nauck MA, Meininger G, Sheng D, et al. Efficacy and safety of dipeptidyl peptidase-4 inhibitor, sitagliptin, compared with the sufonylurea, glipizide, in patients with type 2 diabetes inadequately controlled on metformin alone: a randomized, double-blind, non-inferiority trial. Diabetes Obes Metab. 2007;9:194-205.
13. Aschner P, Kipnes MS, Lunceford JK, et al. Effect of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy on glycemic control in patients with type 2 diabetes. Diabetes Care. 2006;29:2632-2637.
14. Charbonnel B, Karasik A, Liu J, et al. Efficacy and safety of dipeptidyl peptidase-4 inhibitor sitagliptin added to ongoing metformin therapy in patients with type 2 diabetes inadequately controlled with metformin alone. Diabetes Care. 2006;29:2638-2643.
15. Inzucchi SE. Oral antihyperglycemic therapy for type 2 diabetes: scientific review. JAMA. 2002;287:360-372.
16. Sitagliptin/metformin (Janumet) for type 2 diabetes. Med Lett Drugs Ther. 2007;49:45-47.
17. Sitagliptin (Januvia) for type 2 diabetes. Med Lett Drugs Ther. 2007;49:1-3.
18. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet.1998;352:837-853.
19. The effect of intensive treatment of diabetes in the development and progression of long term complications in insulin dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med. 1993;329:977-986.
20. Resnick HE, Bardsley J, Foster GL, et al. Achievement of American Diabetes Association clinical practice recommendations among US adults with diabetes, 1999-2002. Diabetes Care. 2006;29:531-537.
21. Monnier L, Colette C, Dunseath G, et al. The loss of postprandial glycemic control precedes stepwise deterioration of fasting with worsening diabetes. Diabetes Care. 2007;30:263-269.
22. Monnier L, Lapinski H, Colette C. Contributions of fasting and postprandial plasma glucose increments to the overall diurnal hyperglycemia of type 2 diabetics patients: variations with increasing levels of HbA1c. Diabetes Care. 2003;26:881-885.
23. Nathan DM, Buse JB, Davidson MB, et al. Management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy. Diabetes Care. 2006;29:1963-1972.
24. American Association of Clinical Endocrinologists Diabetes Mellitus Guidelines--2207. Available at: www.aace.com/pub/pdf/guidelines/
DMGuidelines2007. Accessed June 29, 2007.
25. Berger J. Economic and clinical impact of innovative pharmacy benefit designs in the management of diabetes pharmacotherapy. Am J Managed Care. 2007;13(2 Suppl):S55-S58.
26. Riedel AA, Plauschinat CA. Clinical inertia in the management of type 2 diabetes. Presented at: 66th Scientific Sesson of the American Diabetes Association. Washington, DC; 2006. Abstract 552-P.
27. Turner RC, Cull CA, Frighi V, et al. Glycemic control with diet, sulfonylurea, metformin or insulin in patients with type 2 diabetes mellitus: progressive requirement for multiple therapies (UKPDS 49). JAMA. 1999;281:2005-2012
28. Clark NG. Defending ADA's A1c target. DOC News-American Diabetes Association. 2005;2:3. Available at: docnews.diabetes journals.org. Accessed June 22, 2007.
29. Lauster CD, McKaveney TP, Muench SV. Vildagliptin: a novel oral therapy for type 2 diabetes mellitus. Am J Health-Syst Pharm. 2007;64:265-273.
30. Dejager S, Baron MA, Schweizer A. DPP-4 Inhibitors: review of vildagliptin phase 3 data. Rev Endocrinol. 2007;1(2):47-51.
31. Chirico M, Cherian S, Anderson S, et al. New agents for the treatment of diabetes. Rev Endocrinol. 2007;1:42-46.
32. Trujillo J. Incretin hormones in the treatment
of type 2 diabetes. Formulary. 2006;41:130-141.
To comment on this article, contact
editor@uspharmacist.com.





