US Pharm. 2015;40(11):63-64.
Through large or repetitious exposure to infected blood, hepatitis C virus (HCV) is a blood-borne pathogen that causes damage to the liver. Though such an infection is often caused by injection drug use (i.e., sharing needles, etc.) and accidental needle sticks, other pathways of transmission include vertical transmission from mother to fetus, sharing personal items that could contain blood (e.g., razors, toothbrushes, etc.), and unprotected sex with an HCV-positive person.1
Predominantly a chronic infection, hepatitis C (Hep C) may be classically asymptomatic for many years. As such, in the face of slow disease progression, it may be years before a patient is diagnosed. In fact, most patients are unaware of their status until a healthcare provider detects elevated liver enzymes, primarily alanine aminotranferase, through routine blood screening.
Symptoms experienced by patients are directly proportional to the duration of infection. Approximately 20% to 30% of newly infected people will experience acute symptoms, such as pain in the right upper quadrant, poor appetite, jaundice, fatigue, and fever. Further, symptoms of chronic hepatitis parallel those of hepatic failure: Bleeding/bruising easily, ascites, hepatic encephalopathy, weight loss, and edema may be seen.
Epidemiology
Approximately 2.7 million people in the United States have chronic Hep C. In 2013, according to the CDC, there were an estimated 30,000 new cases. Of those newly infected, 75% to 85% will progress to chronic Hep C. However, complete spontaneous remission has been observed in 15% to 25% of infected individuals.2
Etiology
HCV is a spherical, enveloped, single-stranded RNA virus. Though there are six genotypes, genotypes 4, 5, and 6 do not cause significant infection rates in the U.S. Each genotype may be further divided into subtypes, affording providers a more defined causative homology. Genotype 1 is the most common worldwide, accounting for approximately 40% to 80% of total infections. Moreover, genotype 1 is associated with severe liver damage and total disease progression.3
Diagnosis
There are two distinct methods for diagnosing Hep C. The initial test, an enzyme-linked immunosorbent assay (ELISA), screens for antibodies to the HCV (anti-HCV). A confirmatory examination, the HCV polymerase chain reaction (PCR), is the gold standard as it scans specifically for the RNA of HCV.
As of late, problems with diagnosis of Hep C have been the production of false-positive and -negative results. A false-positive occurs when the ELISA test confirms the presence of anti-HCV; yet, the patient is not infected. False-negative results occur when the patient is infected but the antibodies are undetectable. The reason for this is the window period, which is defined as the time it takes for the body to produce antibodies. The window period usually lasts 4 to 8 weeks after infection. A false-negative can be more detrimental because infected patients will not receive treatment in a timely fashion.
Elecsys Anti-HCV Immunoassay
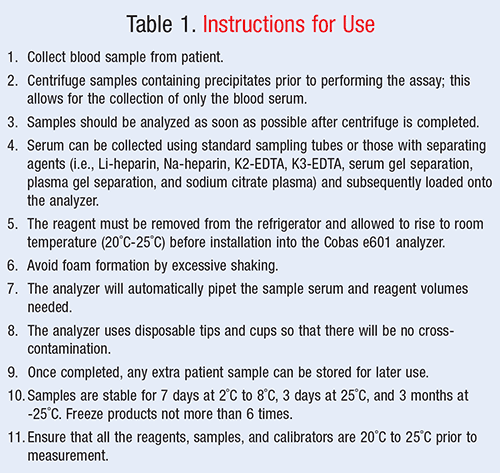
Developed by Roche Diagnostics, the Elecsys anti-HCV immunoassay is used to detect anti-HCV through the use of electrochemiluminescence (ECL) technology. ECL technology allows for amplification of anti-HCV signals, affording it the ability to parallel a PCR test in sensitivity. As such, patients may test positive as early as 2 to 3 weeks after infection, compared to the traditional 6 to 8 weeks required for the ELISA test.4 ECL technology mitigates the number of false-negatives produced because of its increased sensitivity to HCV antibodies. The Elecsys anti-HCV immunoassay is shown in FIGURE 1, and directions for its use can be found in TABLE 1.
Cobas e601 Analyzer
This new immunoassay was built for use with the Cobas e601 analyzer. Serum or plasma from blood is mixed with test chemicals. If anti-HCV is remarkable, reagents will bind with it, generating light, or ECL. The analyzer measures the light released, providing results in as few as 18 minutes.5 The FDA has not licensed this product for use as a final diagnostic test for HCV or for mass screening of donated tissue products.
Clinical Trial
According to a prospective study, the Elecsys assay had a positive percent agreement (PPA) of 99.64% for adult patients in the study. Additionally, the authors concluded that the negative percent agreement (NPA) was 98.81%.6 PPA is defined as the number of patients with HCV RNA divided by the number of true-positive and false-positive results of ELISA testing. On the other hand, NPA is the number of patients having no HCV RNA divided by the number of true-negatives and false-negatives produced from ELISA testing. The specificity was evaluated and tested by examining the cross reactivity with various medical conditions, multiple drugs, and antibodies. All categories showed minimal reactivity; as such, it can be surmised that the rate of false-positives/-negatives will be equally low. These values show the new device’s ability to determine when the patient is infected. In a separate study, Yoo et al reinforced the aforementioned findings and sensitivity and specificity values of 100% and >99.60%, respectively.6 Overall, the studies performed show the capacity to accurately detect antibodies to HCV.
Conclusion
Elecsys anti-HCV immunoassay is unique in that it employs ECL technology to gain more precise and consistent results. It is safe for all patients who are able to give blood for testing and capable of delivering dependable marks without frequent calibration. The ease of use and efficiency of this new immunoassay makes the device an attractive alternate to other available tests. For more information contact Roche Diagnostics at 1 (800) 428-5076.
REFERENCES
1. Yeung LT, King SM, Roberts EA. Mother-to-infant transmission of hepatitis C virus. Hepatology. 2001;34(2):223-229.
2. CDC. Hepatitis C information, Division of Viral Hepatitis. 2015. www.cdc.gov/hepatitis/hcv/. Accessed September 11, 2015.
3. Dhawan VK. Hepatitis C: practice essentials, background, pathophysiology. Emedicinemedscapecom. 2015. http://emedicine.medscape.com/article/177792-overview#a4. Accessed September 9, 2015.
4. Esteban J, van Helden J, Alborino F, et al. Multicenter evaluation of the Elecsys® anti-HCV II assay for the diagnosis of hepatitis C virus infection. J Med Virol. 2013;85(8):1362-1368.
5. Yoo S, Wang L, Ning H, et al. Evaluation of the Elecsys® Anti-HCV II assay for routine hepatitis C virus screening of different Asian Pacific populations and detection of early infection. J Clin Virol. 2015;64:20-27.
6. FDA.gov. SSED for Elecsys anti-HCV immunoassay. 2015. www.accessdata.fda.gov/cdrh_docs/pdf14/P140021b.pdf. Accessed September 7, 2015.
To comment on this article, contact rdavidson@uspharmacist.com.





