US Pharm. 2019;44(8):HS-11-HS-16.
ABSTRACT: Priapism is the occurrence of a persistent penile erection lasting 4 hours beyond orgasm, or one that is unrelated to sexual stimulation. Most episodes are idiopathic; other contributors include medication therapy or drugs of abuse, perineal trauma, and hematologic dyscrasias such as sickle cell disease. There are three subtypes of priapism, with ischemic priapism causing most cases; treatment may differ by type. Priapism may be a side effect of common medications, including antipsychotics, antidepressants, and antihypertensives, and recreational drugs including alcohol, cocaine, and marijuana. Detumescence is the goal of treatment, managed through a process of aspiration followed by intracavernous drug therapy with phenylephrine, epinephrine, etilephrine, and a possible surgical shunt. Pharmacists can screen for drug-drug interactions and drug-disease interactions to minimize the use of underlying medications that that may lead to priapism or its recurrence.
Priapism is the occurrence of a persistent penile erection lasting 4 hours beyond orgasm or one that is unrelated to sexual stimulation.1 A relatively uncommon disorder, the literature consists of case reviews, and current guidelines rely on expert consensus. A Nationwide Emergency Department Sample gathered data between 2006 and 2009 to determine the number of emergency department encounters throughout the United States. An estimated 8.05 per 100,000 emergency department visits, a total of 39,964 encounters, occurred during this period. This was significantly higher than prior estimates and a more accurate estimate of acute episodes of priapism. The analysis stated that this number did not account for men who may have presented to a primary care doctor or urologist and may have detumesced prior to evaluation.2 While the majority of episodes are idiopathic—up to 33% of cases—other contributors include medication therapy or drugs of abuse (21%), perineal trauma (12%), and hematologic dyscrasias (11%; e.g., sickle cell disease [SCD]).3
Classification
There are three subtypes of priapism: ischemic priapism (veno-occlusive, low flow), shuttering priapism (intermittent), and nonischemic priapism (arterial, high flow). Prompt evaluation is essential to determine the type, because treatment approaches may differ, with detumescence of priapism as the primary outcome.1
Ischemic priapism accounts for 95% of all episodes and is a urological emergency analogous to compartment syndrome, a condition leading to increased pressure in a confined body space requiring a rapid therapeutic approach. The goal of treatment is initiation of detumescence and maintainance of flaccidity. During these episodes, a veno-occlusive effect impedes venous outflow of the corpora cavernosa, which increases pressure within the corporal bodies, facilitating accumulation of blood that stagnates in the corporal sinusoid and contributing to an environment that is hypoxic and acidic. Increasing pressure within the corporal bodies creates rigidity and a painful erection. During such episodes, persistent tissue ischemia lasting longer than 24 hours can result in endothelial and smooth-muscle cell destruction. Penile fibrosis can occur during this prolonged state, and after 48 hours, smooth-muscle necrosis can occur. With delay of treatment, the irreversible sequlae can lead to varying degrees of permeant erectile dysfunction.1,4
Shutter priapism is a distinct, recurrent form of ischemic priapism, manifesting as painful episodes that can be short-lived and self-limiting. However, the unwanted and painful erections of short duration can progress over time to become more prolonged ischemic priapism. Etiology is often idiopathic, and a history of preexisting conditions, as well as medications that are known to elicit such events, should be obtained. Patients with hematologic abnormalities are more prone to these bouts. This subtype is prevalent in patients with SCD, and the occurrence of priapism is estimated to range from 35% to 89% in this population.5 The underlying mechanism of SCD is attributed to sludging of erythrocytes leading to a veno-occlusive event. Medical consultation should be sought for management of recurrence and prolonged events.1,3,5
Nonischemic priapism, also known as high-flow priapism, is due to an unregulated or disrupted arterial inflow, allowing well-oxygenated blood in the corpora. Dysregulation of vasorelaxing and vasoconstricting factors often results from injury, affecting nerve innervation and blood supply to the genitals. Mechanism of injury can result from an array of conditions, and the time between trauma and patient presentation can range from days to years without significant impact on outcome.3
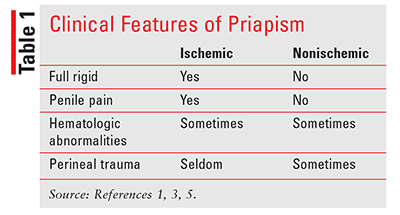
The following medical conditions have a more prevalent association with nonischemic priapism: hematological dyscrasias, SCD (most common reason for priapism), and history of malignancy (leukemia, multiple myeloma).1 Contrasting with the other subtypes of priapism, venous flow is left unimpeded, exposing the cavernosum to oxygenated blood. The erection is not fully rigid, often painless, and not associated with long-term erectile dysfunction.1,3,5 See Table 1 for clinical features of priapism.
Drug-Induced Priapism
Pharmacologically induced priapism is not often considered as a side effect in many common medications and recreational drugs. Awareness of this potential side effect may improve patient outcomes in the management of depression, hypertension, lower urinary tract disorders, and hypercoagulable disorders, such as SCD, and reduce the likelihood of priapism.6 Of the drug-induced cases of priapism, more than half are linked to the use of typical antipsychotics. Use or abuse of psychopharmaceuticals is a common factor in ischemic priapism; trazodone is the most commonly associated prescription drug, and among recreational drug abusers, cocaine is associated with priapism. Additional recreational substances associated with priapism include alcohol and amphetamines. Other pharmaceuticals associated with priapism include the newer atypical antipsychotics, antihypertensives, and anticoagulants.7,8 However, there are no indicators that can predict who may develop drug-induced priapism, which may present at any time during therapy or with the addition of new medications.9
The majority of cases of pharmacologic priapism remain to be fully explained but are likely to be multifactorial physiological disorders. Risk of priapism associated with medications is attributed to alpha-1-adrenergic receptor antagonism or veno-occlusive effects. The alpha-1-receptor is predominant in the urinary tract and vital to smooth-muscle contraction. Antagonism of alpha1-receptor may lead to arterial dilation that triggers venous stasis due to increased intracavernosal pressure, thus inhibiting penile detumescence. Veno-occlusive incidents can occur with medications or imbalance of the sympathetic autonomic nervous system that regulates phosphodiesterase inhibitors and thus prevents termination of the erectile response. In any circumstance, the offending agent, if known, should be discontinued and an early treatment plan initiated.8-10
Antipsychotics
Sexual dysfunction is prevalent among patients taking antipsychotic drugs (APDs) because they antagonize dopaminergic transmission in the central nervous system. APDs are best classified as typical and atypical, with the two classes differing by varying affinities to dopamine receptor (D2) antagonists and alpha-adrenergic blockade. Multiple cases have been documented that correlate antipsychotics with low-potency dopamine and higher alpha-adrenergic blockade and are associated with a higher number of incidences of medication-induced priapism. The imbalance of alpha-1-adrenergic blocking with a large variation in affinity for alpha-1-adrenergic receptors favors parasympathetic-mediated erection and inhibits sympathetic-mediated detumescence.9 The typical APDs include phenothiazines (chlorpromazine, thioridazine, fluphenazine) and are known to have low potency for dopamine receptors. An increased alpha-adrenergic affinity compared with high-potency drugs like haloperidol has been associated to a lesser extent.8 The atypical APDs include clozapine, which was one of the first in this class to be reported, with an alpha-adrenergic affinity half that of risperidone. Risperidone’s tendency to be associated with priapism when compared with ziprasidone, which is slightly higher in affinity for alpha-adrenergic blockade, has been documented. However, the lower incidence of priapism among ziprasidone users may be related to shorter market availability compared with risperidone. Other atypicals such as olanzapine, aripiprazole, and quetiapine also have a documented association with priapism.8,9
Antidepressants
There are a number of case reports of sexual dysfunction, including priapism, occurring with use of antidepressants. Given their actions on modulation of neurotransmission, the risk of occurrence may increase with certain combinations of fluoxetine and citalopram or with the addition of alpha-blockers such as tamsulosin.6 Trazodone is the most notable, and although the incidence rate is considered low (1:1000-10,000), it has been reported in 79% of cases of psychotropic-drug–induced priapism or prolonged erections.10 Trazodone’s actions have been attributed to a mixture of high- and moderate-affinity alpha1- and alpha2-adrenoceptors, and it has been beneficial in treating erectile dysfunction. Trazodone has been used off-label in urological patients. In one study, a Taiwanese national database from 1997 to 2008 showed 55.1% of men were assigned a code for an erectile-dysfunction–related condition. The majority of men were older than age 40 years with a 0.45% occurrence of priapism.11
Antihypertensives
Primarily developed to treat hypertension, adrenergic alpha-blockers have become the mainstay for lower urinary tract symptoms (LUTS). The complication of priapism is suggested to result from its alpha-blocker mechanism, where the corpus is a combined mixture of alpha1- and alpha2-adrenoceptors. Adrenergic alpha-blockers may be nonuroselective, the type responsible for most priapism episodes (e.g., doxazosin, prazosin, and terazosin), or uroselective (e.g., tamsulosin, alfuzosin).6,10,12 Prazosin may be most frequently associated with priapism, in part because of its nonuroselectivity and increased binding affinity to alpha-receptors. In addition, its use in hypertension requires higher dosing than in LUTS. Because of its decreased sympathetic stimuli, priapism may be less apparent in urological practice.12
Anticoagulation
Heparin is commonly used for anticoagulation and has been associated with priapism in a limited number of cases. It is not clear if the priapism is initially of the high-flow type and later becomes low-flow. It is interesting that its mechanism is related to either vasodilation or hypercoagulability or both. Hypercoagulability might occur as trauma due to high pressure in the corpora permitting blood elements to form in a hypercoagulative state, or as a rebound phenomenon. Discontinuation of heparin may promote a thrombus-enhancing effect from subtherapeutic levels in the presence of protein C deficiency or heparin-dependent immunoglobulin G platelet-aggregating antibodies. Each hypercoagulability mechanism then enables platelet aggregates that alter penile blood flow.6,13
Other Nonprescription Drugs
Illicit drug use with alcohol, cocaine, and marijuana is postulated to inhibit the reuptake of neurotransmitters. During an intoxicated state it is suggested that sexual excitement could result in an erection with impaired ability to achieve detumescence. Marijuana may promote parasympathetic-activity–promoting tumescence. Cocaine and substances made from its base (e.g., crack) deplete norepinephrine from sympathetic nerve terminals with long-term use. Recreational drugs can contribute to priapism, but cases are rare.6,14
Treatment
Historically, patients with priapism have been recommended nonmedical interventions including ejaculation, exercise, ice packs, and cold baths to end the erection by inducing vasoconstriction and possibly providing pain relief. These remedies have achieved varying levels of success; however, they lack evidence and do not correlate with the hemodynamics of priapism. These suggestions are still provided but should not delay timely medical attention.1,3 Detumescence is the goal of treatment, managed through a process of aspiration followed by intracavernous drug therapy and a possible surgical shunt.3
Intracavernous Drug Therapy
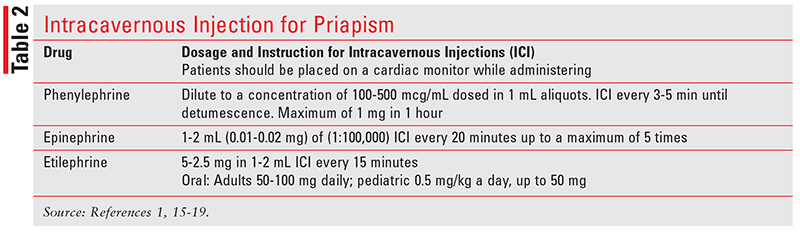
This treatment is often very effective in treating low-flow priapism. It involves intracavernous injections (ICI) of drugs known as alpha-agonists, such as diluted phenylephrine, epinephrine, and etilephrine, into the penis, which causes the veins to open. As a result, the blood circulation to the penis is reestablished. All sympathomimetics have potential side effects (e.g., tachycardia, hypertension, palpitations, subarachnoid hemorrhage, and headache); patients should be monitored and care should be taken in the preparation and administration of injections (Table 2).
Phenylephrine is a relatively selective -1-adrenergic receptor agonist without -mediated activity that may increase the risk of cardiovascular effects and is recommended by the American Urological Association (AUA) 2003 guideline as first-line treatment, due to its side-effect profile. Phenylephrine has shown 65% efficacy with aspiration, administered at 100 to 500 mcg/mL ICI with a maximum of 1 mg in an hour.1 As a single agent, phenylephrine has been shown effective in a small group that received 1 to 6 ICI of 0.5 mg, with 8 of 9 patients achieving detumescence. No cardiovascular effects were observed, and this suggests routine aspiration and irrigation may not be necessary.15 Ischemic priapism is often associated with acidosis, with higher disassociation rates suggesting decreased binding affinity. In one report, two patients with priapism lasting longer than 36 hours were successfully treated with a mean dose of 4.5 mg ICI without any adverse events. Suggesting higher doses than previously reported may be needed to overcome acidosis in the setting of prolonged ischemic episodes.16
Epinephrine use is more common in pediatrics and SCD, with a success rate of 50% after a single injection and 95% overall effectiveness.17,18 The AUA guideline showed an 81% resolution of priapism. In young patients with SCD, one study found detumescence within 1 minute in 37 out of 39 occurrences.1,17 Epinephrine’s beta-adrenergic activity promotes vasodilation, benefiting detumescence.17 One to 2 mL (1:100,000) were administered ICI every 20 minutes with no incidence of cardiovascular effects reported.17
Etilephrine is an alpha-adrenergic agonist available as oral and injectable preparations in most of Europe, but it is not widely use in the United States. One case report followed a man who had success with prior injections and was then taught to self-inject to manage recurrent episodes. After a 10-year period he reported good efficacy and preserved erectile function.18 Another review of SCD patients with recurrent priapism taking 50 to 100 mg a day orally found that 72% had a good clinical response.19 Similar efficacy and safety profiles have suggested this as a possible approach in selected cases.
Oral management of prolonged priapism is often recommended to promote continued vasoconstriction; however, only few studies have shown success. Pseudoephedrine is often first-line due to its -agonist effects analogous to the vasoconstrictive properties of phenylephrine.5,20 Terbutaline, a beta2-agonist with minor beta1 effects, is thought to have some alpha-agonist activity promoting smooth-muscle relaxation.21 Terbutaline was compared with pseudoephedrine and placebo in one study to determine the efficacy of oral agents in achieving detumescence. Successful resolution of priapism was found in each group, with terbutaline (36%) achieving significantly better results than placebo (12%) and without significant benefit compared with pseudoephedrine (28%). However, oral medication was found to be unsuccessful in 75% (56 of 75) of patients.20 In another study terbutaline compared with placebo alone found significant resolution of priapism (15%); however, terbutaline proved more effective in neurogenic (66.5%), vasculogenic (50%), and psychogenic (25%) impotence.21 Oral medications provide some benefit but are perhaps limited in reversing priapism owing to low levels of the drug in the cavernosal blood.20
The Role of the Pharmacist
Pharmacists at the bedside often play a vital role in patient management, developing a trusting relationship, being mindful of the possible embarrassment and trauma of priapism for patients, and facilitating the communication of information to physicians to aid in treatment. In priapism, as in other disease states, it is important to screen for drug-drug interactions and drug-disease interactions help to minimize the use of underlying medications that may lead to priapism or its recurrence. Pharmacists must also counsel patients on common and severe adverse effects and the need to seek immediate medical attention for erections lasting longer than 4 hours.
REFERENCES
1. Montague DK, Jarow J, Broderick GA. et al. American Urological Association guideline on the management of priapism. J Urol. 2003;170:1318-1324.
2. Stein DM, Flum AS, Cashy J, et al. Nationwide emergency department visits for priapism in the United States. J Sex Med. 2013;10:2418-2422.
3. Broderick GA, Kadioglu A, Bivalacqua TJ, et al. Priapism: pathogenesis, epidemiology, and management. J Sex Med. 2010;7:476-500.
4. Bivalacqua TJ, Burnett AL. Priapism: new concepts in the pathophysiology and new treatment strategies. Curr Urol Rep. 2006:7(6):497-502.
5. Muneer A, Minhas S, Arya M, Ralph DJ. Stuttering priapism–a review of the therapeutic options. Int J Clin Pract. 2008;62(8):1265-1270.
6. Scherzer ND, Reddy AG, Le TV, et al. Unintended consequences: a review of pharmacologcally-induced priapism. Sex Med Rev. 2019;7:283-292.
7. Gottsch HP, Berger RE, Yang CC. Priapism: comorbid factors and treatment outcomes in a contemporary series. Adv Urol. 2012; 2012:672624.
8. Sood S, James W, Bailon MJ. Priapism associated with atypical antipsychotic medications: a review. Int Clin Psychopharmacol. 2008;23(1):9-17.
9. Compton MT, Miller AH. Priapism associated with conventional and atypical antipsychotic medications: a review. J Clin Psychiatry. 2001;62(5):362-366.
10. Krege S, Goepel M, Sperling H, Michel MC. Affinity of trazodone for human penile 1-and 2-adrenoceptors. BJU Int. 2000;85:959-961.
11. Cheng WM, Lin TP, Lin AT, et al. A nationwide population study of trazodone use in urology patients. J Chin Med Assoc. 2013;76(8):432-437.
12. Spagnul SJT, Cabral PHO, Verndl DO, Glina S. Adrenergic -blockers: an infrequent and overlooked cause of priapism. Int J Impot Res. 2011;23(3):95-98.
13. Bschleipfer TH, Hauck EW, Diemer TH, et al. Heparin-induced priapism. Int J Impot Res. 2001;13(6):357-359.
14. Greiner T, Schneider M, Regente J, et al. Priapism induced by various psychotropics: a case series. World J Biol Psychiatry. 2018;1-17.
15. Muruve N, Hosking DH. Intracorporeal phenylephrine in the treatment of priapism. J Urol. 1996;155(1):141-143.
16. Wen CC, Munarriz R, McAuley I, et al. Management of ischemic priapism with high-dose intracavernosal phenylephrine: from bench to bedside. J Sex Med. 2006;3(5):918-922.
17. Mantadkis E, Ewalt DH, Cavander JD, et al. Outpatient penile aspiration and epinephrine irrigation for young patients with sickle cell anemia and prolonged priapism. Blood. 2000;95(1):78-82.
18. Teloken C, Ribeiro EP, Chammas M, et al. Intracavernosal etilefrine self-injection therapy for recurrent priapism: one decade of follow-up. Urology. 2004;65:1002.
19. Okpala I, Westerdale N, Jegede T, Cheung B. Etilefrine for the prevention of priapism in adult sickle cell disease. Br J Haematol. 2002;118(3):918-921.
20. Lowe FC, Jarow JP, . Placebo-controlled study of oral terbutaline and pseudoephedrine in management of prostaglandin E1-induced prolonged erections. Urology. 1993;42(1):51-53.
21. Priyadarshi S. Oral terbutaline in the management of pharmacologically induce prolonged erection. Int J Impot Res. 2004;16(5):424-426.
To comment on this article, contact rdavidson@uspharmacist.com.