US Pharm. 2015;40(8):HS12-HS15.
ABSTRACT: Uremic pruritus (UP) is a common complication in patients with chronic kidney disease. Although the etiology of UP is not fully understood, several mechanisms for its pathogenesis have been proposed. UP can manifest in a single localized area or throughout the body, and it frequently affects the patient’s quality of life. Although there are treatment options that demonstrate efficacy against UP, no single therapy has been confirmed as the cornerstone of management for this condition. Pharmacotherapeutic management depends on symptom severity and the patient’s response to initial therapies.
Uremic pruritus (UP; chronic itching in patients with chronic kidney disease) has a prevalence of 15% to 49% and impacts more than one-third of patients undergoing dialysis.1,2 Of this subset of patients, 30% to 60% experience symptoms for more than a year.1,3 There seem to be no differences in prevalence or severity between peritoneal dialysis and hemodialysis (HD); however, the incidence of UP has decreased over time owing to improved dialysis techniques.1 UP symptoms, which are subjective, can range from mild, localized itching to widespread, severe irritation. With its long duration, frequency, and high intensity, UP has been linked to sleep disturbances, depression, impaired quality of life, and mortality.1-5
Etiology
The etiology of UP imperfectly understood. Studies have shown that patients with severe pruritus are male and have a high predialysis concentration of blood urea nitrogen, hypercalcemia, hyperphosphatemia, secondary hyperparathyroidism, and hypermagnesemia.6 Medications that may contribute to the development of UP have also been investigated. Of note, ACE inhibitor treatment has been associated with UP, whereas in patients without UP furosemide treatment is more common.1 A possible explanation for this is the adverse effect (AE) of pruritus from ACE inhibitor treatment.
Pathophysiology
Although UP is a common complication in patients with end-stage renal disease (ESRD), its pathophysiology is poorly understood and thought to be multifactorial. Early theories suggest that UP may be caused by hyperparathyroidism, hypercalcemia, hyperphosphatemia, and increased histamine secretion from proliferated mast cells.7 These electrolyte imbalances, in addition to decreased calcitriol production, can increase parathyroid production and hyperplasia of parathyroid cells, thereby resulting in secondary hyperparathyroidism. As a result, hyperparathyroidism can stimulate mast cells to release histamine and increase the deposition of calcium and magnesium salts on the skin.8 However, not all patients with severe hyperparathyroidism have pruritus. Additionally, the literature has yet to confirm these theories. Most recently, hypotheses of UP consider the inflammatory response of the immune system, in addition to opioidergic receptor activation.9,10
The immune hypothesis of UP postulates that the condition originates in an inflammatory response rather than a local skin disorder.9 Studies have shown that immunomodulating therapies such as ultraviolet B phototherapy, thalidomide, and calcineurin inhibitors are effective in decreasing the inflammatory responses seen in UP patients.9,11,12 ESRD pruritus is associated with elevated C-reactive proteins and other inflammatory cytokines, hinting that there is an upregulated inflammatory component in this form of pruritus.9 These observations suggest that UP has a correlation with the inflammatory response, as better outcomes are achieved by suppressing many of the inflammatory mediators.
While the immune hypothesis proposes an inflammatory etiology, the opioid hypothesis speculates that UP may be due to imbalances in the expression of mu and kappa receptors. Mu receptor agonists are known to induce pruritus through mu receptor activation and kappa receptor blockade.10 Further, changes in the endogenous opioidergic systems may be the result of an increased serum beta-endorphin to dynorphin ratio, which is commonly observed in patients undergoing HD.13
Clinical Manifestations
A few studies have evaluated the clinical characteristics of UP. The disorder generally presents with cutaneous manifestations—such as excoriations (from scratching) with or without impetigo, prurigo lesions, and lichenification—that occur as secondary phenomena.8 UP is characterized by daily itching episodes with a symmetric distribution; the intensity worsens during the night, thus causing sleeplessness in 61% of patients.1 Additionally, in about 25% of patients, pruritus is most severe during or immediately after dialysis.1,11 More than one-third of patients have generalized UP in areas including the back (70%), abdomen (46%), head (44%), and arms (43%), whereas those with localized UP experience symptoms primarily in areas of the back, face, and shunt arm.1 Additional symptoms associated with UP include depression and agitation, occurring in approximately 8% and 36% of patients, respectively.
Diagnosis
By definition, to be diagnosed with UP, a patient must experience pruritus shortly before or during dialysis, although in only 7% of patients is UP confined to this period.1,11 More importantly, UP is a diagnosis of exclusion, with other potential etiologies ruled out.1 Xerosis (abnormal dryness) is common in dialysis patients as a result of atrophied sweat glands, impaired secretions, and reduced hydration.2 This mechanism, an associated cause of pruritus alone, may reduce the threshold for itching and contribute to the symptomology of UP.1 Therefore, xerosis should be resolved before it is concluded that UP is the cause of itching in chronic kidney disease or dialysis patients.
Management
The management of UP is not completely understood, and the only definitive treatment is renal transplantation. Pharmacotherapeutic management of UP depends on the severity of symptoms, as well as the patient’s response to initial therapies. To date, treatment options are based on a small number of randomized, controlled trials and published case studies. The ability to prove benefit of treatment versus placebo also poses difficulty, as UP has a contributory psychological component.4 Therapies may be categorized as physical (dialysis-related), topical, or systemic. Treatment often begins with first-line agents such as skin emollients and gabapentin, proceeding to other systemic modalities in refractory patients (TABLE 1). These modalities include selective serotonin reuptake inhibitors (SSRIs), glucocorticoids, omega-3 fatty acids, and thalidomide. Finally, the kappa-opioid receptor agonist nalfurafine is a potential future option, with clinical trials currently in progress.
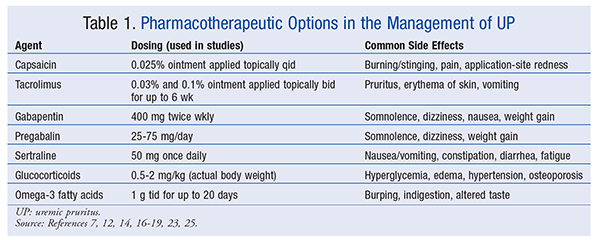
Capsaicin: Topical capsaicin can be beneficial in ESRD patients experiencing UP, as it eliminates substance P by binding to type C sensory neurons with subsequent reductions in the synthesis, transport, and storage of this molecule.5 Substance P is thought to be a neuropeptide contributing to the conduction of pain and itching impulses. Despite strong reports that placebo topical emollients have provided relief of xerosis, previous studies evaluating capsaicin therapy have been favorable. Additionally, studies have reported marked relief or remission of symptoms in patients using capsaicin, although the studies were severely limited in terms of population size and sufficiency of data reported.14,15
A study revealed cutaneous side effects of moderate-to-severe burning and/or stinging in 32.2% of patients.5 Frequent application of capsaicin and adherence to therapy are necessary to obtain the full effect of treatment, but this may be difficult to achieve outside of a study population.15 Sites and surface areas of pruritus, as well as potentially damaged skin, must also be considered given the potential limitations in patient flexibility and capsaicin application.
Tacrolimus: Tacrolimus ointment has been investigated only in small sample sizes for a period of up to 6 weeks.12 Results revealed a >80% reduction in the severity of pruritus.12 No side effects were observed during or after the treatment period, and systemic absorption did not appear to be a concern in these patients, since blood concentrations were negative in clinical study. However, sustained use (>6 weeks) of this topical agent is not advised, owing to the risk of skin cancer.16
Gabaminergic Agents: Gabapentin and pregabalin have been shown to provide relief to patients with UP. Gabapentin is an antiepileptic agent that modulates various receptor sites and alters dopamine, serotonin, and norepinephrine release. However, in the treatment of UP, gabapentin’s mechanism is thought to be its effect on voltage-dependent calcium-ion channels. By blocking neuronal calcium influx, gabapentin may inhibit ectopic discharge from injured nerves, thus interrupting the pathway that triggers the itching sensation in UP patients.17
In a double-blind, placebo-controlled trial, adult patients on maintenance hemodialysis received 4 weeks of treatment with either gabapentin or placebo administered twice weekly subsequent to HD sessions. After 4 weeks of treatment, the mean decrease in pruritus scores in the gabapentin group was beneficial compared with that in the placebo group.17 Somnolence, dizziness, and nausea were the most commonly reported side effects. AEs were of mild-to-moderate severity and usually subsided within 5 to 10 days after the first dose of gabapentin. Pregabalin is also suggested as a viable treatment option for UP. In a recent study, pregabalin administered to HD patients resulted in a swift reduction in itching within the first week of treatment.18 Most patients responded to 25 mg daily, and the most frequently reported AEs leading to discontinuation of therapy were dizziness (4%) and somnolence (3%).
SSRIs
Although SSRIs have been used to treat many causes of pruritus, their effectiveness in UP has been documented in only a few case reports, case series, and controlled trials. The proposed mechanism is postulated to be a reduction in itching perception or sensation.19 Of the various SSRIs, sertraline has been most studied for UP management. Although trials of sertraline in ESRD patients have been limited by sample size, positive results have been demonstrated. A study published in 2012 evaluated the effectiveness of sertraline in 19 ESRD patients experiencing UP.19 After 4 months of treatment, 50% of patients with severe pruritus at baseline experienced reduction of symptoms to weak pruritus and 40% experienced reduction to moderate pruritus. Sertraline was well tolerated, with no reported side effects. A retrospective study published in 2013 also yielded positive results for sertraline use in 20 patients.20
Another benefit of sertraline therapy is the convenience of dosing, as no adjustments are needed in patients with renal insufficiency or ESRD. AEs of sertraline use in this patient population include increased bleeding, gastrointestinal symptomology, sexual dysfunction, and cardiovascular effects, all of which must be weighed against the potential benefits of reduced UP.21
Glucocorticosteroids: Systemic glucocorticosteroids have been found beneficial in UP patients. These agents exert their effect by modulating cytokines, chemokines, lipid mediators (phospholipase A2, prostanoids, and leukotrienes), nitric oxide, and signal transduction pathways such as nuclear factor kappa-B.7 The ability of glucocorticoids to decrease inflammation, leukocyte migration, and phagocytosis renders these agents highly effective in many pruritic conditions. The use of these agents should be limited to controlling acute, severe forms of pruritus. Use is also limited by side effects such as adrenocorticotropic hormone suppression, hyperglycemia, osteoporosis, hypertension, infection, glaucoma, and mental dysfunction.
Omega-3 Fatty Acids: These fatty acids contain eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), which are precursors to certain signaling molecules involved in inflammation reduction. Moreover, omega-3 fatty acids reduce free radicals and leukotrienes, thereby adding to their effectiveness in UP management. One study found that fatty acids are present in abnormal proportions in the serum of HD patients.22 Because of the imbalance of fatty acids, patients exhibited symptoms characteristic of essential fatty acid deficiency, such as dry and scaly skin, abnormal perspiration, and susceptibility to infections. This deficiency could be a contributing factor to pruritus in HD patients. In the study, after 8 weeks of treatment, patients who used fish oil had a greater reduction in pruritus scores.22 In an anecdotal placebo-controlled, double-blind study, omega-3 fatty acids were shown to be more effective than placebo.23 This study found that the use of omega-3 fatty acids reduced the severity of UP by 65%.23
Nalfurafine: Nalfurafine is a kappa-opioid receptor agonist that inhibits the signals of mu-opioid receptors’ itch response. Although it is not yet available in the United States, phase II studies are under way based on positive results in other countries. In placebo-controlled studies, about one-third of patients experienced a response (defined by a 50% reduction in symptoms) to nalfurafine therapy.24 This statistically significant improvement occurred after only 1 to 2 weeks of therapy, and only mild side effects were reported.4,24 The most common effects associated with nalfurafine use included headache, nausea, vomiting, insomnia, and vertigo.4 In the future, nalfurafine may be used patients with UP.
Conclusion
UP is an extremely common complication in patients with ESRD, but a definitive pathogenesis and a fully delineated treatment algorithm have not yet been established. There is evidence that UP may be due either to an inflammatory response or to imbalances in mu or kappa receptors. Published literature supports both of these hypotheses, and other studies suggest additional contributing factors. Agents that are used to decrease inflammatory responses by targeting specific mediators have demonstrated benefits in UP patients despite the lack of robust randomized, controlled trials. Current and prospective therapies that affect mu or kappa receptors have produced promising results, but study sizes often have been limited. Other therapies that modulate pruritus signals have these same shortcomings; however, these therapies could be additional options for patients with UP. Since UP is affected by patient perception and can vary significantly in severity, extent, and duration, there is no single therapy that is right for all patients. Instead, healthcare providers and UP patients must together consider the risks and benefits of each treatment option to determine an appropriate patient-specific match.
REFERENCES
1. Zucker I, Yosipovitch G, David M, et al. Prevalence and characterization of uremic pruritus in patients undergoing hemodialysis: uremic pruritus is still a major problem for patients with end-stage renal disease. J Am Acad Dermatol. 2003;49:842-846.
2. Narita I, Iguchi S, Omori K, Gejyo F. Uremic pruritus in chronic hemodialysis patients. J Nephrol. 2008;21:161-165.
3. Ko MJ, Wu HY, Chen HY, et al. Uremic pruritus, dialysis adequacy, and metabolic profiles in hemodialysis patients: a prospective 5-year cohort study. PLoS One. 2013;8:e71404.
4. Wikström B, Gellert R, Ladefoged SD, et al. Kappa-opioid system in uremic pruritus: multicenter, randomized, double-blind, placebo-controlled clinical studies. J Am Soc Nephrol. 2005;16:3742-3747.
5. Makhlough A, Ala S, Haj-Heydari Z, et al. Topical capsaicin therapy for uremic pruritus in patients on hemodialysis. Iran J Kidney Dis. 2010;4:137-140.
6. Narita I, Alchi B, Omori K, et al. Etiology and prognostic significance of severe uremic pruritus in chronic hemodialysis patients. Kidney Int. 2006;69:1626-1632.
7. Steinhoff M, Cevikbas F, Ikoma A, Berger TG. Pruritus: management algorithms and experimental therapies. Semin Cutan Med Surg. 2011;30:127-137.
8. Tajbakhsh R, Joshaghani HR, Bayzayi F, et al. Association between pruritus and serum concentrations of parathormone, calcium and phosphorus in hemodialysis patients. Saudi J Kidney Dis Transpl. 2013;24:702-706.
9. Berger TG, Steinhoff M. Pruritus and renal failure. Semin Cutan Med Surg. 2011;30:99-100.
10. Mettang T, Pauli-Magnus C, Alscher DM. Uraemic pruritus—new perspectives and insights from recent trials. Nephrol Dial Transplant. 2002;17:1558-1563.
11. Gilchrest BA, Rowe JW, Brown RS, et al. Ultraviolet phototherapy of uremic pruritus. Long-term results and possible mechanism of action. Ann Intern Med. 1979;91:17-21.
12. Kuypers DR, Claes K, Evenepoel P, et al. A prospective proof of concept study of the efficacy of tacrolimus ointment on uraemic pruritus (UP) in patients on chronic dialysis therapy. Nephrol Dial Transplant. 2004;19:1895-1901.
13. Kuypers DR. Skin problems in chronic kidney disease. Nat Clin Pract Nephrol. 2009;5:157-170.
14. Tarng DC, Cho YL, Liu HN, Huang TP. Hemodialysis-related pruritus: a double-blind, placebo-controlled, crossover study of capsaicin 0.025% cream. Nephron. 1996;72:617-622.
15. Gooding SM, Canter PH, Coelho HF, et al. Systematic review of topical capsaicin in the treatment of pruritus. Int J Dermatol. 2010;49:858-865.
16. FDA. Information for healthcare professionals: tacrolimus (marketed as Protopic). www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm126497.htm. Accessed April 28, 2015.
17. Naini AE, Harandi AA, Khanbabapour S, et al. Gabapentin: a promising drug for the treatment of uremic pruritus. Saudi J Kidney Dis Transpl. 2007;18:378-381.
18. Shavit L, Grenader T, Lifschitz M, Slotki I. Use of pregabalin in the management of chronic uremic pruritus. J Pain Symptom Manage. 2013;45:776-781.
19. Shakiba M, Sanadgol H, Azmoude HR, et al. Effect of sertraline on uremic pruritus improvement in ESRD patients. Int J Nephrol. 2012;2012:363901.
20. Chan KY, Li CW, Wong H, et al. Use of sertraline for antihistamine-refractory uremic pruritus in renal palliative care patients. J Palliat Med. 2013;16:966-970.
21. Hedayati SS, Yalamanchili V, Finkelstein FO. A practical approach to the treatment of depression in patients with chronic kidney disease and end-stage renal disease. Kidney Int. 2012;81:247-255.
22. Peck LW, Monsen ER, Ahmad S. Effect of three sources of long-chain fatty acids on the plasma fatty acid profile, plasma prostaglandin E2 concentrations, and pruritus symptoms in hemodialysis patients. Am J Clin Nutr. 1996;64:210-214.
23. Ghanei E, Zeinali J, Borghei M, Homayouni M. Efficacy of omega-3 fatty acids supplementation in treatment of uremic pruritus in hemodialysis patients: a double-blind randomized controlled trial. Iran Red Crescent Med J. 2012;14:515-522.
24. Kumagai H, Ebata T, Takamori K, et al. Effect of a novel kappa-receptor agonist, nalfurafine hydrochloride, on severe itch in 337 haemodialysis patients: a Phase III, randomized, double-blind, placebo-controlled study. Nephrol Dial Transplant. 2010;25:1251-1257.
25. Micromedex 2.0 (electronic version). Greenwood Village, CO: Truven Health Analytics; 2015. www.micromedexsolutions.com. Accessed April 28, 2015.
To comment on this article, contact rdavidson@uspharmacist.com.






