US Pharm. 2017;42(1):33-36.
ABSTRACT: Patients who experience headaches related to strenuous physical activity may find it difficult to exercise at sufficient durations or intensities. These headaches, which are known as primary exercise headaches or exertional headaches, impact people during or immediately following exercise. Although the pathophysiology of exertional headaches is not well defined, there are two hypotheses that may explain them. These theories help determine appropriate prevention and treatment strategies for patients who get exertional headaches. The adoption of appropriate nonpharmacologic and pharmacologic preventive and treatment measures improves exercise tolerance in patients who experience exercise-induced headaches.
Patients who experience headache upon strenuous physical activity (exertional headache) may find it difficult to exercise at sufficient durations or intensities. The International Classification of Headache Disorders defines primary exercise headache as “headache precipitated by any form of exercise in the absence of any intracranial disorder.”1 Diagnostic criteria include two or more headaches that are brought on by, and occurring during or after, strenuous physical activity and that last more than 48 hours. Primary means that these headaches are not caused by an underlying neurologic problem and are benign other than the discomfort they cause the patient, whereas secondary headaches may result from a serious disease or condition.2 Primary exercise headache is a type of exertional headache; other types include primary cough headache and headache associated with sexual activity.
One etiological study described the approximate prevalence of primary exertional headaches as 1.5% of all headache consultations.3 In patients with chronic headaches, approximately one-third are associated with exercise, with about 15% being migrainous.4,5
Presentation
Exertional headaches generally present unilaterally with moderate-to-severe pain that is pulsating in nature.1 In one retrospective analysis, approximately one-third of patients reported neck pain as the initial symptom at headache onset.5 This symptom was more commonly associated with exercise headaches than with typical migraines, especially in men.
Although the diagnosis of primary exercise headache remains distinct from that of migraine, these two conditions often share similar characteristics, such as nausea, vomiting, scotomas, and photophobia.1,6 If a patient experiences an aura, an additional diagnosis of migraine with aura should be considered. The element unique to exercise headache is its onset during or shortly after completing exercise. Several studies analyzing exercise headaches found that they occurred most commonly after weightlifting, distance running, or other aerobic activity, possibly because of increases in intracranial pressure during these activities.5-8 Other triggers include insufficient warm-up, extreme heat or cold, dehydration, hypoglycemia, and fatigue or extreme exercise.6,8
In a study conducted by Williams and Nukada, a survey was administered to 129 patients experiencing sports-related headache.8 In patients with nontraumatic exertional headaches, the reported headache duration ranged from 5 minutes to 24 hours, with differences based on the headache trigger. For instance, headaches occurring shortly after intense sprinting or heavy weightlifting usually resolved more quickly than those that were precipitated by factors established over a longer time frame, such as dehydration or endurance exercises. Headaches lasting more than 1 hour were always associated with continuous aerobic effort.8
The presentation of exertional headache may be similar to that of many other neurologic conditions, so a thorough differential diagnosis should be conducted to identify possible causes of exertional headache, determine the corresponding treatment, and correct any potential underlying condition.1,9
Pathophysiology
The pathogenesis of migraine is not fully understood, but two main mechanisms have been hypothesized.6,9-13 First, migraines are thought to be associated with pain triggers generated in the trigeminal nerve pathways.6,12,13 The presence of the pain generator in the brainstem causes the release of neuroinflammatory chemicals and results in headache. This pain generator is believed to arise from the cerebral vasculature; the cerebral arteries, large veins, and venous sinuses are the most important skull structures that register pain.12,13 The second hypothesized mechanism involves the dysregulation of vasoactive neurotransmitters (serotonin, acetylcholine, and other neuropeptides) that control pain pathways and result in headache onset.6,13 Serotonin receptor modulation and activity on cerebral vasculature are particularly important in the pharmacologic management of migraines, as shown with the triptan class, and migraine symptoms have been associated with drops in serotonin platelet levels.6
Neither of these mechanisms explains the cause of exertional headaches, but both may shed light on the pathophysiology. The mechanism of exertional headaches is usually attributed to increased intracranial pressure and its impact on cerebral blood vessels.9-11 Transient increases in intracranial pressure caused by physical exertion are thought to produce venous or arterial distention that triggers pain-inducing neurotransmitter pathways and headache symptoms.9-11 In studies of weightlifters, systolic blood pressures exceeded 400 mm Hg and diastolic blood pressures exceeded 300 mm Hg during maximal lifts.13 Exertion during physical activity can produce increases of varying degrees in blood pressure and intracranial pressure, which is why athletes can experience headaches of differing intensity and duration depending on the sport performed.8
Prevention
Despite the overwhelming benefits of exercise for the general population, the majority of individuals who experience primary exercise headache report stopping, or lessening the intensity of, their exercise routine to avoid headaches.5 First-line therapy for exercise headache is the implementation of appropriate prevention strategies. Since most exercise headaches are caused by a combination of factors, athletes should be mindful of the variety of prevention mechanisms available.6 Food triggers, including alcohol and caffeine, should be limited or avoided when possible. Foods that cause headache are patient-specific, but may include aged cheeses, cured meats, the additive monosodium glutamate, nuts, beans, tomato-based products, and certain fruits.14 Eating three to six small, well-balanced meals throughout the day and drinking plenty of water to avoid dehydration may be helpful in reducing primary exercise headache.6 Proper sleep hygiene is also an important preventive measure. Patients should establish a regular sleep cycle and aim for 8 hours of restful sleep each night. It is also helpful to limit sleep disruptions such as lights, excess noise, or an uncomfortably warm environment.
Proper intensity and duration of warm-up are crucial in preventing headaches related to exercise.15 A study by Varkey and colleagues assessed the implementation of an exercise program in patients with migraines in an attempt to increase maximum oxygen uptake without worsening migraine status.16 Participants completed a 15-minute warm-up of light to somewhat-hard intensity before engaging in 45 minutes of somewhat-hard to hard physical activity. There was no worsening of migraine control despite extensive exercise. Implementation of an appropriate warm-up can help prevent exercise-induced headaches in people engaging in more strenuous physical activity, as well as help patients develop a tolerance to exercise-induced headaches and reduce the overall monthly occurrence of migraines.
Medication lists should be assessed for agents that may be associated with headaches (TABLE 1), as these agents may exacerbate exertional headaches.13
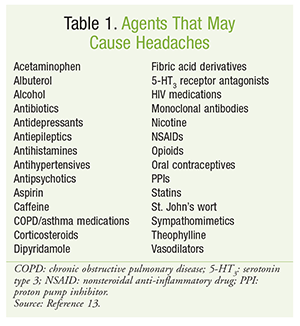
Although guidelines for the prevention of migraines and headaches do not address potential prophylactic options for exertional headaches, application of these guidelines to exertional headaches may still provide viable options and benefits for these patients.1,17
Magnesium: Guidelines classify magnesium as “probably effective” for migraine prevention and note that it should be considered.17 Hypomagnesemia and low levels of magnesium in the cerebrospinal fluid have been associated with exercise-induced migraines.6,18 Approximately 30% of migraineurs have low levels of magnesium, and supplementation with magnesium sulfate 400 mg daily may help reduce the frequency and/or intensity of migraine attacks in these individuals. A recent literature review concluded that the evidence supporting oral magnesium supplementation is limited and that it may be advantageous to focus on increasing dietary magnesium intake rather than relying on magnesium supplements.19 Diarrhea and gastrointestinal (GI) discomfort are common adverse effects of magnesium supplementation and are an important counseling point if therapy is initiated.
Riboflavin and CoQ10: Both riboflavin and ubiquinone (coenzyme Q10, or CoQ10) were shown to decrease migraine frequency compared with placebo and are preventive options for exercise-induced migraines. The mechanism for reducing migraine attacks concerns the impact of these agents on mitochondrial energy.6 Riboflavin and CoQ10 are involved in cellular energy production and are required for the electron-transport chain within the mitochondrial inner membrane. Impaired mitochondrial oxygen metabolism or deficient mitochondrial energy production is thought to play a role in migraine pathogenesis.6,20
Schoenen and colleagues conducted a 3-month, randomized, double-blind, placebo-controlled trial comparing riboflavin 400 mg per day versus placebo for the prevention of migraines in 55 patients.20 Riboflavin showed statistically significant differences in the proportions of patients who improved by at least 50% in migraine-attack frequency (56% vs. 19%, P = .01), headache days (59% vs. 15%, P = .002), and the migraine index (headache days and mean severity, 41% vs. 8%, P = .02). Riboflavin is generally well tolerated; common adverse effects include yellow discoloration of urine, polyuria, and diarrhea.
Sándor and colleagues conducted a 3-month, randomized, double-blind, placebo-controlled trial comparing CoQ10 100 mg three times daily versus placebo for migraine prevention in 42 patients.21 CoQ10 showed significant improvement in the 50% responder rate for migraine-attack frequency (47% vs. 14%, P = .02). The CoQ10 group had a statistically significant improvement in the change between months 1 and 4 in the number of days with nausea and in the number of headache days. The researchers commented that CoQ10’s excellent tolerability could render it a treatment option for children or women of childbearing age.
Feverfew: Feverfew is another option, and several studies support its use for headaches. The recommendation in the American Academy of Neurology (AAN) guidelines for migraine prevention is based on five studies with positive results, one negative study, and one study that did not show differences versus placebo when feverfew was given as part of a combination.17 The mechanism for feverfew’s antimigraine effects may be related to a reduction in platelet aggregation and the inhibition of serotonin release from platelets.22 Feverfew at a dosage of 300 mg twice daily can help reduce the frequency of migraines and some of the associated symptoms, such as pain, nausea and vomiting, and phonophobia and photophobia; this is advantageous because previously discussed therapies have benefited only prevention, not symptom reduction.22,23 Adverse effects, which are generally mild and transient, include certain GI disturbances (heartburn, nausea, diarrhea) and mild heart palpitations.
Butterbur: Butterbur was previously recommended by the AAN guidelines for migraine prevention.17 In September 2015, however, butterbur was removed from the guidelines by the AAN Board of Directors based on serious health concerns regarding one of its components, pyrrolizidine alkaloids (PAs)—toxic substances that cause hepatotoxicity in humans. Butterbur should no longer be recommended for migraine prophylaxis.
Treatment
Indomethacin is the first-line choice for short-term treatment of exercise-induced headache.3,9-13 Indomethacin may be taken either on an as-needed basis prior to a known exertional trigger or as scheduled dosing for headache prophylaxis. Indomethacin should be administered 30 to 60 minutes before the known exertional trigger, when the trigger cannot be avoided; when the headache frequency is high or triggers cannot be anticipated, indomethacin is often dosed every 8 hours.3,9-13 Dosages of 25 to 250 mg/day have been recommended for the treatment of exertional headache.3,9-13
The mechanism behind indomethacin’s therapeutic efficacy is not fully understood, but it may be due to the drug’s ability to reduce intracranial pressure.10 Indomethacin is a direct and potent cerebral arteriolar vasoconstrictor, which is likely a significant contributing factor in regulating cerebral blood flow and decreasing intracranial pressure.10,24 Bioavailability of orally administered indomethacin is virtually 100%, and peak concentrations are reached within 0.5 to 2.0 hours.10,25,26 Other cyclooxygenase inhibitors, such as ibuprofen, diclofenac, and naproxen, do not share indomethacin’s cerebral vasoconstrictor effects. This suggests that indomethacin may have an additional mechanism that provides direct vasoconstriction of cerebral blood.10,24
Indomethacin is recommended for short-term use because of adverse effects associated with chronic use of nonsteroidal anti-inflammatory drugs (NSAIDs). When long-term or daily headache prophylaxis is needed, beta-blockers are recommended. The beta-blockers propranolol and nadolol 1 to 2 mg/kg/day are most commonly used for exertional headaches, but metoprolol, atenolol, and timolol also have good supporting data for migraine prevention.3,9,11,12,27 Beta-blockers are an alternative to indomethacin when headache frequency is high, triggers cannot be anticipated, or contraindications to NSAIDs exist. Caution should be taken when beta-blockers are used in athletes because reductions in heart rate and exercise tolerance may occur.6,12
Triptans are another treatment option, particularly when there is a known exertional trigger that cannot be avoided or when the patient does not tolerate indomethacin.11,12,27 The timing of administration prior to exertion is based on patient-specific and pharmacologic parameters of the triptan of choice. Onset, duration, and route of administration vary among triptans. The choice of triptan should be based on the patient’s specific needs, and trial and error may be required to determine which triptan provides optimal efficacy. If prophylactic measures fail, triptans still may provide relief for acute treatment and termination of migraines.27
Patient Case
Presentation: CT, a 48-year-old male, presented with complaints of migraines occurring during exercise. He had a previous diagnosis of migraine with aura; for the past several months, he experienced a migraine after each running session. CT reported that he alternated jogging and walking, which helped prevent the migraines, but he was dissatisfied with this strategy because he could not work out for the duration or intensity he desired. What should be recommended to CT?
Recommendation: It was suggested that CT extend the time of his warm-up and gradually increase his exercise intensity over a longer period of time; it was also recommended that he ensure adequate hydration, especially on days he was going to run. In addition, he was started on magnesium supplementation. These measures have allowed CT to run for longer distances and intensities without experiencing an exercise-related migraine for the last 3 months.
Conclusion
Implementation of proper warm-up and nonpharmacologic measures can help prevent exercise-induced migraines in patients who experience them. In patients who need pharmacologic therapy, preventive options such as magnesium, riboflavin, and NSAIDs should be considered. Appropriate abortive therapy, such as triptans, should be readily available to the patient during exercise for times when preventive measures fail.
REFERENCES
1. Headache Classification Committee of the International Headache Society. The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia. 2013;33:629-808.
2. Johns Hopkins Medicine. Neurology and neurosurgery. www.hopkinsmedicine.org/neurology_neurosurgery/centers_clinics/headache/conditions. Accessed July 25, 2016.
3. Pascual J, González-Mandly A, Martín R, Oterino A. Headaches precipitated by cough, prolonged exercise or sexual activity: a prospective etiological and clinical study. J Headache Pain. 2008;9:259-266.
4. Williams SJ, Nukada H. Sport and exercise headache: part 1. Prevalence among university students. Br J Sports Med. 1994;28:90-95.
5. Koppen H, van Veldhoven PL. Migraineurs with exercise-triggered attacks have a distinct migraine. J Headache Pain. 2013;14:99.
6. Nadelson C. Sport and exercise-induced migraines. Curr Sports Med Rep. 2006;5:29-33.
7. Chen SP, Fuh JL, Lu SR, Wang SJ. Exertional headache—a survey of 1963 adolescents. Cephalalgia. 2009;29:401-407.
8. Williams SJ, Nukada H. Sport and exercise headache: part 2. Diagnosis and classification. Br J Sports Med. 1994;28:96-100.
9. Halker RB, Vargas BB. Primary exertional headache: updates in the literature. Curr Pain Headache Rep. 2013;17:337.
10. VanderPluym J. Indomethacin-responsive headaches. Curr Neurol Neurosci Rep. 2015;15:516.
11. Allena M, Rossi P, Tassorelli C, et al. Focus on therapy of the Chapter IV headaches provoked by exertional factors: primary cough headache, primary exertional headache and primary headache associated with sexual activity. J Headache Pain. 2010;11:525-530.
12. Kernick DP, Goadsby PJ. Guidance for the management of headache in sport on behalf of The Royal College of General Practitioners and The British Association for the Study of Headache. Cephalalgia. 2011;31:106-111.
13. McCrory P. Headaches and exercise. Sports Med. 2000;30:221-229.
14. Cleveland Clinic. Headaches and food. http://my.clevelandclinic.org/health/diseases_conditions/hic_Overview_of_Headaches_in_Adults/hic_Headaches_and_Food. Accessed August 22, 2016.
15. Lambert RW Jr, Burnet DL. Prevention of exercise induced migraine by quantitative warm-up. Headache. 1985;25:317-319.
16. Varkey E, Cider A, Carlsson J, Linde M. A study to evaluate the feasibility of an aerobic exercise program in patients with migraine. Headache. 2009;49:563-570.
17. Holland S, Silberstein SD, Freitag F, et al. Evidence-based guideline update: NSAIDs and other complementary treatments for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1346-1353.
18. Sun-Edelstein C, Mauskop A. Role of magnesium in the pathogenesis and treatment of migraine. Expert Rev Neurother. 2009;9:369-379.
19. Teigen L, Boes CJ. An evidence-based review of oral magnesium supplementation in the preventive treatment of migraine. Cephalalgia. 2015;35:912-922.
20. Schoenen J, Jacquy J, Lenaerts M. Effectiveness of high-dose riboflavin in migraine prophylaxis. A randomized controlled trial. Neurology. 1998;50:466-470.
21. Sándor PS, Di Clemente L, Coppola G, et al. Efficacy of coenzyme Q10 in migraine prophylaxis: a randomized controlled trial. Neurology. 2005;64:713-715.
22. Feverfew. In: Natural Medicines [online database]. Somerville, MA: Therapeutic Research Center; 2015. https://naturalmedicines.therapeuticresearch.com. Accessed December 7, 2016.
23. Pittler MH, Ernst E. Feverfew for preventing migraine. Cochrane Database Syst Rev. 2004(1):CD002286.
24. Baccaro FG, Romano M, Ciapponi A, López-Alcalde J. Indomethacin in controlling intracranial hypertension secondary to severe traumatic brain injury. Cochrane Database Syst Rev. 2015(6):CD011725.
25. Indomethacin. In: Clinical Pharmacology [online database]. Tampa, FL: Elsevier; 2016. www.clinicalpharmacology.com. Accessed September 5, 2016.
26. Indomethacin immediate-release capsule package insert. Philadelphia, PA: Iroko Pharmaceuticals; October 2011.
27. Silberstein SD, Holland S, Freitag F, et al. Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1337-1345.
To comment on this article, contact rdavidson@uspharmacist.com.





