US Pharm. 2011;36(4):20-28
The thyroid gland facilitates normal growth and maturation. This is achieved through the maintenance of a level of metabolism in tissues that is optimal for normal function.1 Hyperthyroidism refers to a state of excessive thyroid hormone availability to peripheral tissues and is characterized by hypermetabolism and elevated serum levels of free thyroid hormones.2,3 It is estimated that 10% to 17% of all hyperthyroid patients are older than 60 years.2 In elderly hyperthyroid patients with atrial fibrillation, there is a risk for systemic embolization and stroke, particularly in those with comorbid cardiac disease.2 The clinical presentation of hyperthyroidism may be dramatic or subtle, with many common symptoms and signs similar to those of adrenergic excess such as nervousness, palpitations, increased sweating, heat hypersensitivity, weight loss, fatigue, increased bowel movements, and tremor.3 The diagnosis of this condition is made clinically and with thyroid function testing. Treatment is based on the cause of the disorder.
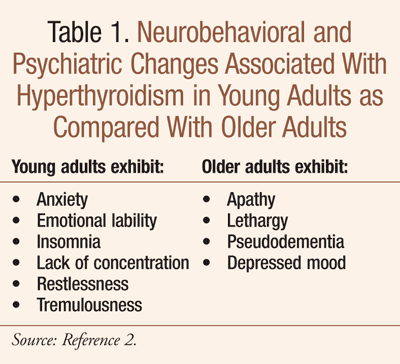
In the elderly, however, the onset of hyperthyroidism may present atypically. Referred to as apathetic or masked hyperthyroidism, symptoms resemble depression or dementia (TABLE 1).2,3 This is especially the case with toxic nodular goiter; toxic multinodular goiter is more common in the geriatric population and has been reported in approximately 50% of older patients with hyperthyroidism.2,3 Seniors are more likely to experience atrial fibrillation, syncope, altered sensorium, heart failure, and weakness.3,4 Some elderly patients with apathetic hyperthyroidism present with weight loss, anorexia, nausea, vomiting, and constipation, instead of the classic gastrointestinal signs and symptoms of hyperthyroidism such as weight loss with increased appetite and rapid intestinal transit (i.e., frequent defecation).2 In one study, weight loss was noted as a major diagnostic finding in 80% of patients older than 70 years; experts indicate hyperthyroidism should be ruled out as a cause of weight loss in seniors before broad evaluations for gastrointestinal disease or occult malignancy.2
Most elderly patients do not exhibit tremor or exophthalmos, an abnormal protrusion of the eye.3 Ocular signs seen in the elderly are largely due to excessive adrenergic stimulation and include stare, eyelid lag, eyelid retraction, and mild conjunctival injection. Symptoms and signs of hyperthyroidism in seniors may involve only a single organ system and usually remit with successful treatment.3
Graves’ Disease
Graves’ disease (toxic diffuse goiter), named after Robert J. Graves, MD, is the most common cause of hyperthyroidism.3,5,6 In seniors, Graves’ disease is a common cause of hyperthyroidism, but other etiologies are more frequent.2 It is classified as an autoimmune thyroid disorder in which thyroid-stimulating immunoglobulins bind to and activate thyrotropin receptors; the thyroid gland grows as a result causing thyroid follicles to synthesize more thyroid hormone.6,7 Graves’ disease is characterized by hyperthyroidism (due to circulating autoantibodies) and one or more of the following: goiter, exophthalmos, and infiltrative dermopathy (also referred to as pretibial myxedema) (TABLE 2).3,6 It is thought that this condition may represent an element of more extensive autoimmune processes that lead to dysfunction of multiple organs in some patients.6 Graves’ disease is associated with other conditions including pernicious anemia, vitiligo, type 1 diabetes mellitus, autoimmune adrenal insufficiency, systemic sclerosis, myasthenia gravis, Sjögren's syndrome, rheumatoid arthritis, and systemic lupus erythematosus.8

Women are seven times more likely to develop Graves’ disease than men and the development of the disease usually occurs after age 20 years; other risk factors include stress, pregnancy, and smoking.9
Infiltrative Ophthalmopathy
Infiltrative ophthalmopathy is a hallmark of Graves’ disease and a serious development that is responsible for the exophthalmos seen in this disorder.3,6 Ocular manifestations can occur years before, at the time of, or after the discovery of hyperthyroidism and are referred to as Graves’ ophthalmopathy. Approximately 25% to 30% of patients with Graves’ disease have clinical evidence of Graves’ ophthalmopathy.6
While the cause of this ophthalmopathy is unknown, its proposed mechanism is the development of retrobulbar autoimmune inflammation secondary to the release of cytokines, the thickening of extraocular muscles, and the swelling of orbital contents.2 This condition is comprised of two phases: The inflammatory stage, marked by edema and deposition of glycosaminoglycan in the extraocular muscles, and the fibrotic stage, which is a convalescent phase.6 Characteristically, patients experience irritation, lacrimation, orbital pain, photophobia, increased retro-orbital tissue, and exophthalmos; lymphocytic infiltration of the extraocular muscles causes weakness of the ocular muscles often leading to diplopia (double vision).3 Graves’ ophthalmopathy improves spontaneously in 64% of patients.6
A complete ophthalmologic exam is indicated if a patient is symptomatic; this includes a retinal examination and slit-lamp examination performed by an ophthalmologist.6 In order to clinically follow the course of ophthalmopathy, it is necessary to measure diplopia fields, eyelid fissures, range of extraocular muscles, visual acuity, and proptosis (bulging) for quantitative assessment.6 Conjunctival injection and chemosis (edema) are signs of corneal or conjunctival irritation.6 Consultation with an ophthalmologist may be necessary for unilateral or bilateral proptosis, to work up other etiologies for eye findings besides Graves’ disease, and for follow-up of visual acuity, corneal disease prevention, and eye muscle function.6 The course of this disease is one of gradual progression over many years in approximately 10% to 20% of patients, followed by clinical stability; progressive worsening, with visual impairment in some, is seen in approximately 2% to 5% of patients.6
Treatment
It is recommended that Graves’ ophthalmopathy be treated collaboratively by an endocrinologist and an ophthalmologist. Treatment may require corticosteroids, orbital radiation, and surgery.3 While correction of hyperthyroidism is important for the ophthalmopathy, pharmacists should note that antithyroid drugs (e.g., methimazole, propylthiouracil [PTU]) and thyroidectomy do not influence the course of the ophthalmopathy; radioiodine treatment may exacerbate preexisting ophthalmopathy, but this can be prevented by treatment with glucocorticoids.6
Thyroid ablation: In the long term, thyroid ablation (i.e., with radioactive iodine I131) may be beneficial for ophthalmopathy because of the decrease in antigens shared by the thyroid and the orbit in the autoimmune reactions; it is considered to be the agent of choice for Graves’ disease.6,10 In general, improvement of ophthalmopathy is seen with treatment of hyperthyroidism; however, hypothyroidism must be avoided because it worsens ophthalmopathy.6
Local therapeutic measures: Sunglasses, eye patches, artificial tears and ointments, nocturnal taping of the eyes, prisms, and elevating the head at night have the ability to control symptoms and signs in mild-to-moderate ophthalmopathy.6
Glucocorticoids: As indicated by current evidence, glucocorticoids are the only class of drug therapy that, either alone or combined with other therapies, has an unequivocal role in the management of Graves’ ophthalmopathy.3 Glucocorticoids have been shown to decrease T4-to-T3 conversion and decrease levels of thyroid hormones; the adverse effect profile of long-term glucocorticoid therapy, however, makes it unattractive for long-term management of Graves’ hyperthyroidism.6 While prednisone has been customarily used in the management of Graves’ ophthalmopathy, other glucocorticoids at equipotent doses may also be effective.6 Treatment of active disease requires: 1) high-dose glucocorticoids11; 2) orbital radiotherapy; 3) both; or 4) orbital decompression.6 In severe cases or those that are progressive, glucocorticoids at the usual dose of 40 mg/day may be introduced and should be continued until observable evidence of improvement and disease stability are achieved; a dosage taper is then initiated and continued for 4 to 12 weeks.6 Good results have also been reported with high-dose pulse glucocorticoid therapy.6
Orbital radiotherapy: With or without glucocorticoids, orbital radiotherapy may be tried if no response to therapy occurs in the inflammatory phase; this does not increase the risk for radiation-induced tumors, cataracts, and retinopathy (except in patients with diabetes with possible or definite retinopathy).6 While success with gamma knife surgery has been achieved in a limited number of patients, further studies are necessary to validate this modality.6
Miscellaneous and novel treatments: There is limited utility with diuretic therapy for edema secondary to venous engorgement of the orbit.6 The evaluation of novel treatments includes somatostatin analogues or intravenous immunoglobulins. Octreotide LAR (long-acting, repeatable) studies indicate conflicting or marginal therapeutic benefit6,12,-14; infliximab has been reported to successfully treat a case of sight-threatening Graves’ ophthalmopathy6,15; rituximab may transiently deplete B lymphocytes and potentially suppress the active inflammatory phase of Graves’ ophthalmopathy.6,16 A multicenter prospective pilot study in patients with Graves’ ophthalmopathy of recent onset suggests that periocular injection of triamcinolone may reduce diplopia and the size of extraocular muscles.6 Symptoms and proptosis in the inactive phase of Graves’ ophthalmopathy have improved with pentoxifylline, according to one prospective, randomized trial.6
Surgery for Ophthalmopathy
Generally, surgery is performed in the fibrotic phase, when the patient is euthyroid.6 It is considered to be an alternative therapeutic option that is safe in those patients who have moderate-to-severe ophthalmopathy, are noncompliant with (or cannot tolerate) antithyroid drugs, or refuse or cannot undergo radioiodine therapy.6 Thyroidectomy (i.e., near-total) has little, if any, effect on the course of ophthalmopathy.6 It is often necessary to perform extraocular muscle or eyelid surgery (i.e., rehabilitative); eyelid surgery can be performed to improve exposure keratitis.6
Subclinical Hyperthyroidism
Subclinical hyperthyroidism, characterized by low serum thyroid stimulating hormone (TSH) in patients with normal serum free T4 and T3 and absent or minimal symptoms of hyperthyroidism, is far less common than subclinical hypothyroidism. Individuals with serum TSH <0.1 mU/L have 1) an increased incidence of atrial fibrillation, especially in seniors; 2) reduced bone mineral density; 3) increased fractures; and 4) increased mortality.3 Pharmacists should note that many patients with subclinical hyperthyroidism are taking L-thyroxine; a reduction in dosage is the most appropriate management unless the goal of therapy is to target thyroid cancer or nodules.3 Therapy is indicated for patients with endogenous subclinical hyperthyroidism (serum TSH <0.1 mU/L), especially when evidence of atrial fibrillation or reduced bone mineral density is present. Radioactive iodine I131 is the usual treatment.3 In patients with milder symptoms, such as nervousness, a trial of antithyroid drug therapy (e.g., methimazole; it is preferred over PTU since it can be given once daily and has a better side-effect profile) is considered worthwhile.3
Conclusion
Elderly patients often present without classic signs and symptoms of hyperthyroidism. Pharmacists should be aware of apathetic hyperthyroidism in the elderly and how it compares with hyperthyroidism in younger adults. Infiltrative ophthalmopathy, causing exophthalmos—a hallmark of Graves’ disease—should be recognized and treated as a serious development of the disorder.
REFERENCES
1. Finkel R, Clark MA, Cubeddu LX, et al, eds. Lippincott’s Illustrated Reviews: Pharmacology. 4th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2009:275-284.
2. Hershman JM, Hassani S, Samuels MH. Thyroid diseases. In: Halter JB, Ouslander JG, Tinetti ME, et al, eds. Hazzard’s Geriatric Medicine and Gerontology. 6th ed. New York, NY: McGraw-Hill; 2009:1287-1303.
3. Hyperthyroidism. Section: Endocrine disorders. Subject: Thyroid disorders. Merck Manuals.com. Revised June 2008.
www.merckmanuals.com/
4. Monaghan MS, Kissinger JF, Archer J. Endocrine disorders: thyroid and adrenal conditions. In: Youngkin EQ, Sawin KJ, Kissinger JF, et al. Pharmacotherapeutics: A Primary Care Guide. Upper Saddle River, NJ: Pearson Prentice Hall; 2005:649-667.
5. Ellis H. Robert Graves: 1796-1852. Br J Hosp Med (Lond). 2006;67(6):313.
6. Yeung SJ, Habra MA, Chiu AC. Graves’s disease. eMedicine from WebMD. Medscape.com. Updated: April 26, 2010.
http://emedicine.medscape.com/
7. Dorland’s Pocket Medical Dictionary. 28th ed. New York, NY: Elsevier Saunders; 2009.
8. Cruz AA, Akaishi PM, Vargas MA, de Paula SA. Association between thyroid autoimmune dysfunction and non-thyroid autoimmune diseases. Ophthal Plast Reconstr Surg. 2007;23(2):104-108.
9. Grave’s disease.
www.MayoClinic.com. July 7, 2009.
www.mayoclinic.com/health/
10. Sherman SI, Talbert RL. Thyroid disorders. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 7th ed. New York, NY: McGraw-Hill Inc; 2008:1243-1263.
11. Macchia PE, Bagattini M, Lupoli G, et al. High-dose intravenous corticosteroid therapy for Graves’ ophthalmopathy. J Endocrinol Invest. 2001;24:152-158.
12. Dickinson AJ, Vaidya B, Miller M, et al. Double-blind, placebo-controlled trial of octreotide long-acting repeatable (LAR) in thyroid-associated ophthalmopathy. J Clin Endocrinol Metab. 2004;89(12):5910-5915.
13. Wemeau JL, Caron P, Beckers A, et al. Octreotide (long-acting release formulation) treatment in patients with Graves’ orbitopathy: clinical results of a four-month, randomized, placebo-controlled, double-blind study. J Clin Endocrinol Metab. 2005;90:841-848.
14. Stan MN, Garrity JA, Bradley EA, et al. Randomized, double-blind, placebo-controlled trial of long-acting release octreotide for treatment of Graves’ ophthalmopathy. J Clin Endocrinol Metab. 2006;91(12):4817-4824.
15. Durrani OM, Reuser TQ, Murray PI. Infliximab: a novel treatment for sight-threatening thyroid associated ophthalmopathy. Orbit. 2005;24(2):117-119.
16. Salvi M, Vannucchi G, Campi I, et al. Treatment of Graves’ disease and associated ophthalmopathy with the anti-CD20 monoclonal antibody rituximab: an open study. Eur J Endocrinol. 2007;156(1):33-40.
To comment on this article contact rdavidson@uspharmacist.com.





