US Pharm. 2016;41(4):HS5-HS12.
ABSTRACT: Intra-abdominal infection is a broad term that encompasses a number of infectious processes, including peritonitis, diverticulitis, cholecystitis, cholangitis, and pancreatitis. Empirical treatment is based on whether the infection is healthcare- versus community-acquired, which organs are infected, and whether the infection is complicated or uncomplicated. Organism resistance is of concern; thus, it is imperative that pharmacists have the necessary education on etiology and treatment of intra-abdominal infection.
Intra-abdominal infection (IAI) is a broad term that encompasses a number of infectious processes, including peritonitis, diverticulitis, cholecystitis, cholangitis, and pancreatitis. A common cause of IAI is appendicitis. According to DeFrances et al, each year >300,000 people develop appendicitis, resulting in over 1 million hospital days.1 Further-more, 30% of people are diagnosed with diverticulosis by age 60 years; 10% to 25% of these patients will eventually develop diverticulitis.2 This group of infections is the second leading cause of infection-related mortality in the ICU.3
The necessary empirical treatment is established by identifying whether the infection is healthcare- versus community-acquired, which organs are infected, and whether the infection is complicated or uncomplicated. According to the Infectious Diseases Society of America (IDSA), complicated IAI (cIAI) is defined as an infection that extends beyond the wall of a hollow viscus of origin into the abdominal cavity while being associated with an abscess or peritonitis.3,4
Peritonitis is classified as primary (also referred to as spontaneous bacterial peritonitis [SBP] in patients with ascites and hepatic failure), secondary, or tertiary (also labeled as complicated secondary peritonitis). Primary peritonitis is an infection of the peritoneal cavity without breach of the intact gastrointestinal (GI) tract.5 This infection occurs as a result of either an exogenous source, such as the introduction of a peritoneal catheter for dialysis, or an endogenous source, which is most commonly seen in patients with cirrhotic ascites.6 Per Mujais, on average an individual undergoing chronic ambulatory peritoneal dialysis develops peritonitis every 33 months.7
Primary peritonitis is considered a serious infection with significant mortality. Peritonitis causes a mortality rate of 16% in peritoneal dialysis patients.8 Additionally, a 30% to 50% mortality rate is observed in patients with cirrhosis-related SBP.9 It has been estimated that 10% to 30% of cirrhotic patients admitted to the hospital develop SBP.6 Nonetheless, secondary peritonitis occurs as a result of trauma or necrosis of the gut wall, where organisms can translocate into the peritoneal cavity. Tertiary peritonitis is a severe form of secondary peritonitis, where the infection fails to respond to, or reoccurs after, 48 hours of appropriate therapy.5
Etiology
The organism responsible for the IAI can be inferred based on the location of the organ originally infected, since the resident GI flora are typically the culprits in the IAI. For example, infections occurring in the stomach and proximal small intestine mostly involve gram-positive organisms, whereas those in the distal small intestine involve more gram-negative aerobic and facultative anaerobic bacilli. Even more distally, infections in the colon consist mainly of obligate anaerobic organisms.4,5
Primary peritonitis is monomicrobial, where the most common isolates are aerobic gram-negative organisms (Escherichia coli and Klebsiella species), with an increasing occurrence of gram-positive organisms and extended-spectrum beta-lactamase (ESBL)–producing Enterobacteri-aceae.5,10 Secondary peritonitis is usually polymicrobial, with aerobic gram-positive cocci (predominately enterococci, 18%-24%), gram-negative bacilli (E coli, 32%-61%), and anaerobes (Bacteroides species, 25%-80%). Resistant gram-negative bacilli, enterococci, and yeast can be found in tertiary peritonitis.4,5
The main pathogens associated with community-acquired IAIs are Enterobacteriaceae (predominantly E coli ) and anaerobes, particularly Bacteroides fragilis. Healthcare-associated IAIs occur secondary to resistant nonfermenting gram-negative Pseudomonas aeruginosa, Acinetobacter species, ESBL Klebsiella species, and E coli. Additionally, Enterobacter and Proteus species, methicillin-resistant Staphylococcus aureus (MRSA), and Candida organisms may also be present in healthcare-associated IAIs.3
Resistant microorganisms are becoming problematic for IAI cases. In 2011, a study conducted in 19 U.S. hospitals for patients treated for IAIs investigated the trends in microorganisms.11 A total of 1,442 gram-negative isolates were identified as part of the Study for Monitoring Antimicrobial Resistance Trends (SMART). The study evidenced that the incidence of ESBL isolates was 12.7% for Klebsiella pneumoniae, 9.7% for E coli, 3.6% for Proteus mirabilis, and 3.1% for Klebsiella oxytoca.11 Nonetheless, for community-acquired IAIs, increased resistance from gram-negative organisms is also growing. According to Patterson et al, the prevalence of ESBL organisms is 3% for E coli, 7% for Klebsiella species, and 18% for Enterobacter species.12
Clinical Presentation and Diagnosis
Some of the nonspecific physical findings of IAIs are vomiting, constipation, diarrhea, and abdominal distention. In cIAI, the highly vascular and innervated peritoneum ultimately releases cytokines to mount a host response to the bacteria.2,13 This physiological response can lead to an inflammatory state, vasodilation, and hypotension, all of which can be specific variables for sepsis.2,13 A diagnosis of IAI should be considered with the aforementioned physical assessment findings along with sepsis variables. Serum lactic acid, arterial blood gases, and oxygen saturation levels are usually tested, especially if sepsis is suspected. Moreover, radiographic images using CT scans with oral and IV contrast can also be useful for visualization of the abdominal cavity.2
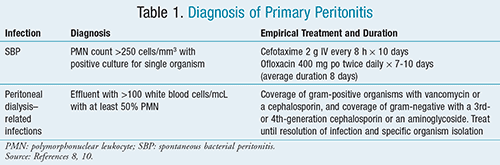
A diagnosis of SBP can be confirmed based on paracentesis, if the ascetic fluid has a polymorphonuclear leukocyte (PMN) of ≥250 cells/mm3 with a positive culture for a single organism (TABLE 1).8,10 SBP may also be identified by paracentesis that is characterized by a PMN of ≥250 cells/mm3 with a positive culture for multiple organisms and at least two of the following: total protein >1 g/dL, lactate dehydrogenase greater than the upper limit of normal (ULN), or glucose <50 mg/dL.10
Treatment and Duration of Therapy
Source control is a vital nonpharmacologic intervention for the treatment of IAIs. Source control is defined as interventional procedures used to help prevent or contain the source of the IAI. Examples may include drainage of an abscess, debridement of necrotic tissue, or surgical repair of perforations. For uncomplicated IAI, source control may serve as the mainstay of treatment with minimal antibiotic use. According to a recent systematic review, prior to an appendectomy, only a single broad-spectrum preoperative dose is warranted for patients with uncomplicated appendicitis.14 Although duration of therapy is still debatable, the guidelines recommend a full 24-hour prophylactic course of antibiotics for appendicitis without perforation.3 In cIAI, source control does not completely eradicate the infection; rather, it aids with infection reduction in order for the antibiotic therapies to establish more effective outcomes.4
Pharmacologic treatment ranges widely from fluids and vasopressor for hemodynamic support in sepsis patients to antimicrobial therapy targeting the source of infection. The remaining portion of this pharmacologic section will focus on antibiotic therapies.
Appropriate antibiotic selection is dependent upon the location, nature (i.e., healthcare- vs. community-acquired), and severity of the infection. Low-risk or mild-to-moderate severity infection is used to describe patients with improved prognosis, low risk of mortality, and no risk for multidrug-resistant organisms. High-risk/high-severity patients are classified as having an APACHE (Acute Physiology and Chronic Health Evaluation) score >15 (predictor of mortality), inability to control the source, age >70 years, immunosuppression, and/or delay of >24 hours to initial intervention.2 Per Swenson et al’s recent study, it was discovered that healthcare-associated pathogens, corticosteroids, solid-organ transplant, liver and pulmonary disease, and duodenal source of infection increase the risk of resistant pathogens.15 The study evidenced a strong relationship between solid-organ transplant plus having resistant pathogens and death (odds ratio [OR] = 5.61, 95% CI, 1.60-16.8; P = .0061).
Primary Peritonitis: Treatment of primary peritonitis varies depending on whether the infection is SBP-related (in patients with ascites and hepatic failure) or peritoneal dialysis–related (see TABLE 1). In SBP diagnosis, broad-spectrum antibiotics (e.g., cefotaxime) are used for most patients, until fluid cultures and sensitivity results are available. Nonetheless, controversy does exist regarding treatment duration. Studies with both ceftriaxone and cefotaxime have demonstrated effectiveness for a 5-day course; however, most clinicians continue to prefer a treatment course of 10 to 14 days.10 Ofloxacin may be utilized as an oral alternative for patients who have not had prior quinolone exposure, vomiting, shock, encephalopathy, or serum creatinine >3 mg/dL.10 Recent studies on recurrence rates are lacking; however, older studies suggest that the 1-year probability of a recurrent episode of SBP is as high as 69%.16 Long-term secondary prophylaxis is recommended using norfloxacin 400 mg daily or double strength trimethoprim-sulfamethoxazole (TMP-SMX) daily due to the high incidence of recurrence.10 Moreover, primary prophylaxis is recommended for high-risk patients (i.e., patients with cirrhosis and variceal/GI bleeding). The guidelines recommend norfloxacin 400 mg orally twice a day for 7 days or ceftriaxone 1 g IV daily for 7 days in hospitalized patients.10
Peritonitis related to peritoneal dialysis should be treated empirically for both gram-positive organisms (i.e., with vancomycin or cephalosporin antibiotics) and gram-negative organisms (i.e., with aminoglycoside or 3rd- or 4th-generation cephalosporin antibiotics)8; see TABLE 1. Antibiotic therapy should begin promptly if the effluent is cloudy, even though cultures may be pending. In peritonitis treatment, the intraperitoneal route is preferred over the IV route. Meanwhile, dosing frequencies may be either intermittent (once daily) or continuous (in each exchange).8
Complicated IAIs: As described previously, cIAIs extend beyond the hollow viscus of origin and into the abdominal cavity with either abscess formation or peritonitis development. This type of infection requires source control plus antibiotic therapies (TABLE 2).3 Empirical antibiotic treatment is determined based on whether the infection is community- or healthcare-acquired, in addition to the severity of infection. According to the IDSA guidelines, empirical broad-spectrum therapy for mild-to-moderate community-acquired cIAI is recommended. However, antipseudomonal, enterococcal, and antifungal agents are not warranted in this subset of patients.3 Nonetheless, the authors caution the use of ampicillin-sulbactam, cefotetan, and clindamycin secondary to its increased resistance. In high-risk community-acquired cIAI, broad-spectrum antibiotics should also be used, either as a single agent or in combination with metronidazole (TABLE 2). The quinolones should only be chosen if the local susceptibilities show >90% sensitivity to E coli.3 Double coverage against gram-negative organisms is unnecessary, unless a resistant organism is suspected. Additionally, MRSA and fungal coverage are not required unless cultures are positive for those organisms.

Resistant organisms are typically present in healthcare-acquired cIAI; thus, empirical antimicrobial coverage should target those microbiologic patterns at the local level. Coverage with an antifungal agent should be initiated if cultures are positive. In Candida albicans infections, fluconazole is the agent of choice, while an echinocandin is preferred if resistance develops.3 Empirical coverage for enterococci is recommended, especially in patients who are immunocompromised or have valvular heart disease or prosthetic intravascular devices.3 Vancomycin is the agent of choice in patients who have a history of MRSA or are colonizers.3
Duration of Therapy: The antimicrobial therapy duration for cIAIs should be 4 to 7 days, unless there is inadequate source control.3 Nonetheless, treatment duration of 10 to 14 days continues to be widely practiced by clinicians.17 A recently conducted study evaluated 518 patients, with a primary goal of determining the optimal duration of therapy.17 The study’s control group consisted of 260 patients with cIAI having adequate source control. These patients received antibiotics until 2 days after resolution of infection for a maximum of 10 days of therapy. On the other hand, the experimental group consisted of 257 patients and was limited to a fixed course of 41 days of antibiotic therapy. One patient withdrew consent after randomization. It was discovered that the primary outcomes of surgical site infection, recurrent IAI, or death were similar among both groups; occurring 21.8% (n = 56) in the experimental group and 22.3% (n = 58) in the control group (P = .92).17 However, the sample size to show equivalence was not obtained; therefore, it cannot be statistically concluded that the groups are equivalent. More trials are needed to properly determine a fixed duration.
Specific Organs and Conditions: Specific organ infections should be treated based on the most common pathogens associated with that body area. As previously discussed, appendicitis is treated with source control and up to 24 hours of prophylactic coverage of aerobic and anaerobic organisms. However, if perforation occurs, treatment utilizing the cIAI recommendations is warranted.3 The IDSA makes specific recommendations for empirical treatment in patients with cholecystitis or cholangitis (TABLE 3).3 Anaerobic coverage is not required for this condition unless biliary-enteric anastomosis has occurred. Additionally, in community-acquired biliary infections, coverage for enterococci is unnecessary. Patients with uncomplicated diverticulitis may be treated with antibiotics for 5 to 7 days, typically with the same agents used to treat cIAI.18 Moreover, infectious pancreatitis is managed by source control in which surgical debridement removes the infected necrotic tissue. If infection is confirmed, antimicrobial therapies that penetrate pancreatic necrosis are recommended (e.g., carbapenems or quinolones plus metronidazole).19

Novel Pharmacologic Agents
Ceftolozane-tazobactam (Zerbaxa) and ceftazidime-avibactam (Avycaz) are two new agents that were brought to the market after the updated IDSA guidelines were issued in 2010. Both agents are among the cephalosporin class, coformulated with a beta-lactamase inhibitor.20-22
Ceftolozane-tazobactam has an FDA approval for cIAI, when given in conjunction with metronidazole. This agent covers gram-negative organisms, such as P aeruginosa and Enterobacteriaceae. Additionally, it has some anaerobic activity (B fragilis) and is gram-positive (Streptococcus species). This agent has an advantage for coverage against resistant organisms such as Class A, AmpC, and OXA beta-lactamases. Nonetheless, it lacks coverage against K pneumoniae carbapenemase (KPC) or metallo-beta-lactamases.20
The ASPECT-cIAI (Assessment of the Safety Profile and Efficacy of Ceftolozane/Tazobactam in Complicated Intra-abdominal Infections) trial is a randomized, double-blind, phase III noninferiority trial that was conducted in patients with cIAI for 4 to 14 days from the start of therapy.21 The trial compared ceftolozane-tazobactam 1.5 g IV every 8 hours plus metronidazole 500 mg IV every 8 hours to meropenem 1g IV every 8 hours. The trial’s primary outcome was clinical cure rates at 24 to 32 days from therapy initiation. The study evidenced how ceftolozane-tazobactam was noninferior to meropenem with 83% (n = 323) versus 87.3% (n = 364), respectively, reaching the primary outcome (95% CI, -8.91 to 0.54). The incidence of GI adverse effects of nausea, vomiting, and diarrhea were similar in both groups.21
In addition, ceftazidime-avibactam has an FDA approval for use in cIAI when combined with metronidazole.22 Similar to ceftolozane-tazobactam, this agent possesses activity against P aeruginosa and Enterobacteriaceae. This agent also covers against Class A, AmpC, and OXA beta-lactamases, with the addition of KPC coverage. However, this agent does not have coverage activities against metallo-beta-lactamases. Given the historical clinical usage of ceftazidime, this agent received a priority review based on phase II data. Therefore, safety and efficacy have not been fully evaluated with the combination agent. According to the FDA, this agent should be reserved until after all other options have failed.22
A randomized, double-blinded phase II study was conducted to help bring this agent to market for cIAI.22 The study compared ceftazidime-avibactam 2.5 g IV every 8 hours plus metronidazole 500 mg IV every 8 hours with meropenem 1g IV every 8 hours for 5 to 14 days. The primary objective of this trial was clinical cure at 2 weeks. Data evidenced how 91.2% (n = 62/68) of the meropenem group and 93.4% (n = 71/76) of the ceftazidime-avibactam plus metronidazole group achieved a favorable clinical response (95% CI, -20.4% to 12.2%). GI symptoms of nausea, vomiting, and diarrhea were similar in both groups.22
Conclusion
Comprehending the etiology and treatment of IAI is vital for pharmacists. Timing of the first agent is critical for sepsis-related IAI as well as nonseptic-related IAI. Moreover, pharmacists may impact patients’ outcomes by appropriately selecting initial antibiotic treatment, screening for duplication of therapy, and identifying the duration of therapy, regardless of whether a facility holds an antimicrobial stewardship program in place.
REFERENCES
1. DeFrances CJ, Cullen KA, Kozak LJ. National Hospital Discharge Survey: 2005 annual summary with detailed diagnosis and procedure data. Vital Heal Stat. 2007;165(13):1-209.
2. Shirah GR, O’Neill PJ. Intra-abdominal infections. Surg Clin N Am. 2014;94:1319-1333.
3. Solomkin JS, Mazuski JE, Bradley JS. Diagnosis and management of complicated intra-abdominal infection in adults and children: guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Clin Infect Dis. 2010;50:133-164.
4. Mazuski JE, Solomkin JS. Intra-abdominal infections. J Surg Clin N Am. 2009;89:421-437.
5. Marshall J, Innes M. Intensive care unit management of intra-abdominal infection. Crit Care Med. 2003;31:2228-2237.
6. Mowat C, Stanley AJ. Review article: spontaneous bacterial peritonitis—diagnosis, treatment, and prevention. Aliment Pharmacol Ther. 2001;15:1851-1859.
7. Mujais S. Microbiology and outcomes of peritonitis in North America. Kidney Int Suppl. 2006;70:S55-S62.
8. Li PK, Szeto CC, Piraino B, et al; International Society for Peritoneal Dialysis. Peritoneal dialysis-related infections recommendations: 2010 update. Perit Dial Int. 2010;30:393-423.
9. Pleguezuelo M, Benitez JM, Jurado J, et al. Diagnosis and management of bacterial infections in decompensated cirrhosis. World J Hepatol. 2013;5:16-25.
10. Runyon BA; AASLD Practice Guidelines Committee. Management of adult patients with ascites due to cirrhosis: an update. Hepatology. 2009;49(6):2087-2107.
11. Hawser SP, Badal RE, Bouchillon SK, et al. Susceptibility of gram-negative aerobic bacilli from intra-abdominal pathogens to antimicrobial agents collected in the United States during 2011. J Infect. 2014;68(1):71-76.
12. Patterson DL, Rossi F, Baquero F, et al. In vitro susceptibilities of aerobic and facultative gram-negative bacilli isolated from patients with intra-abdominal infections worldwide: the 2003 Study for Monitoring Antimicrobial Resistance Trends (SMART). J Antimicrob Chemother. 2005;55(6):965-973.
13. Dellinger RP, Levy MM, Rhodes A, et al. Serving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock, 2012. Crit Care Med. 2013;41(2):580-637.
14. Daskalakis K, Juhlin C, Pahlman L. The use of pre- or postoperative antibiotics in surgery for appendicitis: a systematic review. Scand J Surg. 2014;103(1):14-21.
15. Swenson BR, Metzger R, Hedrick TL, et al. Choosing antibiotics for intra-abdominal infections: what do we mean by “high risk”? Surg Infect. 2009;10(1):29-39.
16. Tito L, Rimola A, Gines P, et al. Recurrence of spontaneous bacteria peritonitis in cirrhosis: frequency and predictive factors. Hepatology. 1988;8(1):27-31.
17. Sawyer RG, Claridge JA, Nathens AB, et al. Trial of short-course antimicrobial therapy for intraabdominal infection. N Engl J Med. 2015;372(21):1996-2005.
18. Byrnes MC, Mazuski JE. Antimicrobial therapy for acute colonic diverticulitis. Surg Infect. 2009;10(2):143-154.
19. Tenner S, Baillie J, DeWitt J, Vege SS. American College of Gastroenterology guideline: management of acute pancreatitis. Am J Gastroenterol. 2013;108(9):1400-1415.
20. Zhanel GG, Chung P, Adam H, et al. Ceftolozane/tazobactam: a novel cephalosporin/b-lactamase inhibitor combination with activity against multidrug-resistant gram-negative bacilli. Drugs. 2014;74(1):31-51.
21. Solomkin J, Hershberger E, Miller B. Ceftolozane/tazobactam plus metronidazole for complicated intra-abdominal infections in an era of multidrug resistance: results from a randomized, double-blind, phase 3 trial (ASPECT-cIAI). Clin Infect Dis. 2015;60(10):1462-1471.
22. Lucasti C, Popescu I, Ramesh MK, et al. Comparative study of the efficacy and safety of ceftazidime/avibactam plus metronidazole versus meropenem in the treatment of complicated intra-abdominal infections in hospitalized adults: results of a randomized, double-blind, phase II trial. J Antimicrob Chemother. 2013;68(5):1183–1192.
To comment on this article, contact rdavidson@uspharmacist.com.





