US Pharm. 2023;48(12):HS12-HS16.
ABSTRACT: The United States is currently experiencing a major increase in the prevalence of obesity. Bariatric surgery is an invasive weapon against the obesity foe that is recommended by clinical-practice guidelines. Various procedures are available to patients under the umbrella of bariatric surgery. The goal of all of the different types of bariatric surgery is to induce weight loss. Bariatric surgery results in weight loss through restrictive properties, malabsorptive properties, or both. Surgery-induced alterations in the gastrointestinal tract can have an impact on patients’ medications and nutritional needs. Pharmacists must understand the differences between the types of bariatric surgery, recognize special drug considerations, and be aware of recommendations for nutritional supplementation in patients who undergo bariatric procedures.
Obesity is recognized by the World Health Organization as a global noncommunicable-disease issue. The prevalence of obesity in the United States is among the highest in the world.1 Bariatric surgery is an invasive treatment option to induce weight loss that many patients undergo when it is clinically indicated. The 2019 update to the clinical practice guidelines for bariatric procedures states that patients should be offered bariatric surgery when the procedure would not be associated with excessive risk and the patient meets specific criteria, including BMI1:
• BMI >40 kg/m2 without coexisting medical problems
• BMI >35 kg/m2 and >1 severe obesity-related complication remediable by weight loss
• BMI 30 to 34.9 kg/m2 and type 2 diabetes with inadequate glycemic control despite optimal lifestyle and medical therapy.
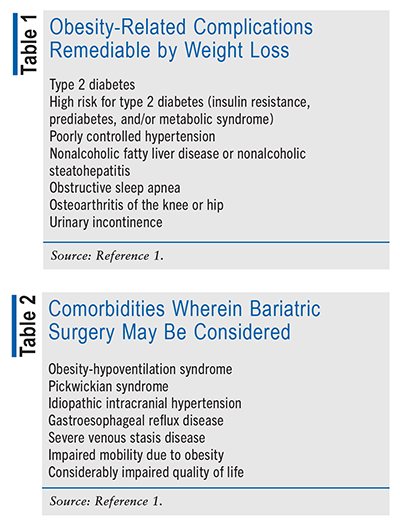
The above BMI numbers are not adjusted for ethnicity. The recommendation for Asian Americans is based on a lower BMI. TABLE 1 lists the obesity-related complications that can be remedied by weight loss.1
Patients with a BMI >35 kg/m2 and a comorbidity listed in TABLE 2 may be considered for bariatric surgery, although the strength of evidence is more variable.1 Finally, bariatric surgery may be considered in patients with health and quality-of-life issues wherein the amount of weight loss needed to prevent or treat clinically significant obesity-related complications cannot be attained using only structured lifestyle changes with medical therapy.1
Types of Bariatric Surgery
Per the American Society for Metabolic and Bariatric Surgery, rates of bariatric surgery have increased from 158,000 cases in 2011 to 262,893 cases in 2021.2 Common types of bariatric surgery include sleeve gastrectomy, laparoscopic adjustable gastric band, Roux-en-Y gastric bypass (RYGB), and biliopancreatic diversion with duodenal switch (BPD/DS). In recent years, the rates for sleeve gastrectomy have trended upward, whereas the RYGB and laparoscopic adjustable gastric band procedures have trended downward. The increase in sleeve gastrectomy rates is principally due to comparable metabolic and weight-loss outcomes, lower complication rates, and fewer nutritional deficiencies compared with RYGB.1
Bariatric surgeries can be classified as restrictive, malabsorptive, or both. Restrictive surgery reduces the volume of food that can be consumed at one time, leading to a reduced total caloric intake.3 The malabsorptive procedure creates a diversion around substantial portions of the digestive tract, causing reduced absorption of consumed products.3
Sleeve Gastrectomy: Sleeve gastrectomy is a surgical procedure in which roughly 80% of the stomach is removed, leaving a smaller, sleevelike stomach. Due to the smaller stomach size, this procedure is classified as a restrictive surgery.3 Sleeve gastrectomy’s favorable profile includes a 25% to 30% target weight loss, no anastomosis, few long-term complications, and positive metabolic effects. However, there is an increased risk for gastroesophageal reflux disease (GERD), leaks can be difficult to manage, and there are little data beyond 5 years.1
Laparoscopic Adjustable Gastric Band: In the laparoscopic adjustable gastric band procedure, an obstructive band is placed around the proximal stomach, physically restricting the rate at which food can pass into the stomach. The surgery is classified as restrictive.3 This procedure has favorable aspects, including the lack of anatomical alteration, the removable nature of the band, and the adjustability of the band. Its unfavorable aspects include the high explant rate, erosion, and the ability for the band to slip or prolapse. The target weight loss is the lowest of all the procedures discussed here, at 20% to 25%.1
RYGB: In the RYGB procedure, a small functional pouch at the top of the stomach is isolated from the rest of the stomach and directly connected to the jejunum, allowing food to bypass most of the stomach and the entire duodenum. This procedure is classified as both restrictive and malabsorptive.3 RYGB is associated with a 30% to 35% total weight loss. Favorable aspects include its strong metabolic effects, its <5% major complication rate, its effectiveness for GERD, and the ability to use it as second-stage surgery after a sleeve gastrectomy. This surgery does have drawbacks, including few revisional options for weight regain, marginal ulcers, internal hernias, and long-term micronutrient deficiencies.1
BPD/DS: The BPD/DS procedure involves removal of up to 75% of the stomach, with the small intestine reconnected to bypass the duodenum, jejunum, and all but the last 50 to 100 cm of the ileum. This procedure is classified as both restrictive and malabsorptive.4 The benefits for some patients include its very strong metabolic effects, durable weight loss, and effectiveness for very high BMI, and it can be used as second-stage surgery after sleeve gastrectomy. Drawbacks of the surgery include malabsorption, 3% to 5% protein-calorie malnutrition, GERD, the potential for internal hernias, duodenal dissection, and higher rates of micronutrient deficiencies. BPD/DS has a target weight loss at the highest amount to be 35% to 45%.1
Special Drug Considerations
When patients physically alter their gastrointestinal tract, drugs will no longer follow known pharmacokinetics. Bariatric surgery can impact absorption through changes in gastric motility, gastric volume, gastric pH, stomach surface area, bile secretions, carrier proteins, and first-pass metabolism.3 Issues with medication absorption are more likely after malabsorptive procedures.3 Generally speaking, bariatric surgery patients should switch from extended-release drug formulations to immediate-release formulations and should consider nonoral dosage forms, when available. It is also reasonable to switch to a liquid, dissolvable, or crushable dosage form when there is a suspicion that the medication is not being properly absorbed.3
Nonsteroidal Anti-inflammatory Drugs (NSAIDs) and Corticosteroids: NSAIDs and corticosteroids (to a lesser extent) should be avoided, if possible, after bariatric procedures per the guideline recommendations.1 The reason is that these drugs have been implicated in the development of anastomotic ulcerations, perforations, and leaks. When their use is unavoidable, a proton pump inhibitor should be considered for risk reduction.1 Pain management is an area in which pharmacists have extensive knowledge about treatment options. Pharmacists can work with both patients and providers to determine a safe alternative to NSAIDs for pain control. Pharmacists can also review patients’ charts to ensure that proper gastrointestinal protection is being considered when a patient with a history of bariatric surgery is prescribed corticosteroids.
Bisphosphonates: Bisphosphonates are used for the treatment of bone loss. In patients with a history of bariatric surgery, the preferred treatment agent for bone loss is an IV-administered bisphosphonate.3 There is concern about oral bisphosphonate therapy in bariatric surgery patients because there may not be adequate absorption and there is a potential for anastomotic ulceration. Some patients may undergo bariatric surgery in their younger years and then encounter bone loss multiple decades down the line. This scenario makes it easy to overlook the drug–disease state interaction. Pharmacists can double-check patients’ charts to ensure that those who have a history of bariatric surgery receive the preferred formulation of bisphosphonate when indicated.
Anticoagulation: Anticoagulation is a necessary medication for many patients with a history of atrial fibrillation or blood clots. Concern exists that bariatric surgery can alter the absorption of this critical medication. The International Society on Thrombosis and Haemostasis (ISTH) recommends not to use a direct oral anticoagulant (DOAC) for treatment or prevention of venous thromboembolism in the acute setting after bariatric surgery.5 Instead, ISTH recommends that patients use parenteral anticoagulation in the early postsurgical phase, then switch to a vitamin K antagonist (VKA) or DOAC after >4 weeks of parenteral treatment.5 When a DOAC is selected, a trough level can be used to check for drug absorption and bioavailability.5 Gastric-banding surgery and sleeve gastrectomy are unlikely to affect apixaban absorption, but they can possibly reduce absorption of dabigatran, edoxaban, and rivaroxaban. RYGB can possibly reduce absorption of all the DOAC agents. VKA agents such as warfarin should be monitored via international normalized ratio as clinically indicated.5
Contraception: Estrogen-based contraceptive therapies should be discontinued before bariatric procedures (one cycle of oral contraceptive in premenopausal women) to minimize the risk of thromboembolic events.1 After bariatric surgery, women may experience an increased fertility and need to use proper contraception aids. The optimal timing for resumption of hormonal therapies after bariatric surgery is unknown.1 However, women should avoid pregnancy for 12 to 18 months after the procedure.1 Malabsorptive procedures carry absorption concerns for oral therapy; patients should consider nonoral contraception therapies. Transdermal patches and subdermal implants are potential options, but the increased amount of flaccid skin may impede patch adherence or provide no suitable insertion site.6 Some hormonal contraceptives are associated with weight gain, and it may be preferable to avoid their use given the primary goal of weight loss. Copper or levonorgestrel intrauterine devices are commonly selected because they avoid the potential decreased medication absorption and weight gain.6
Supplements
The core concept behind bariatric surgery is weight loss due to restriction or malabsorption of food. Therefore, nutritional deficiencies are of concern. Nutritional screening should take place before any bariatric procedure, with a more extensive evaluation performed for malabsorptive procedures.1 Multivitamins with minerals are recommended. Preferred formulations include iron, folic acid, and thiamine. In the early postoperative-care period, the ideal formulation is chewable tablets. Patients who have undergone laparoscopic banding need to chew one multivitamin tablet daily, whereas patients who have had the other procedures should chew two tablets daily.1
Calcium and Vitamin D: All patients who undergo bariatric surgery should receive supplementation of calcium and vitamin D. Patients who have undergone BPD/DS should consume a higher dosage of elemental calcium—1,800 to 2,400 mg/day—in the form of calcium citrate salt. Patients who have undergone laparoscopic gastric banding, sleeve gastrectomy, or RYGB should be supplemented with 1,200 to 1,500 mg of elemental calcium (in the form of calcium citrate) daily.1 Calcium citrate is preferred over calcium carbonate because it is not reliant on gastric pH for absorption.6 All patients, regardless of their surgery type, should be supplemented with vitamin D in the amount of >2,000 to 3,000 IU/day to a therapeutic 25-hydroxyvitamin D level of >30 ng/mL.1 The supplements can be in the form of vitamin D2 or D3. Vitamin D3 is preferred because it is a more potent treatment based on the frequency and amount of drug needed for repletion.1 Patients should have their vitamin D levels evaluated at baseline and yearly, with dose adjustments as clinically indicated.1
Iron: All patients with a history of bariatric surgery should be taking a multivitamin with minerals that contains iron supplementation. In patients who have had BPD/DS, RYGB, or sleeve gastrectomy, total iron should be 18 to 60 mg via multivitamins and additional supplements.1 Additional iron supplements can be in the form of iron sulfate, fumarate, or gluconate.1 Vitamin C may be added to increase pH and enhance iron absorption.6 All patients with a history of bariatric surgery, regardless of procedure type, should be monitored for iron deficiencies 3 months after surgery, then every 3 to 6 months for the first year, followed by annual screenings.
Vitamin B12: Bariatric surgery patients should have their B12 level monitored at baseline and then annually to detect any deficiencies. Vitamin B12 can be supplemented orally, with the preference being disintegrating tablets, sublingual tablets, or liquid formulations. When B12 sufficiency cannot be attained via the oral route, intramuscular or subcutaneous formulations are preferred.1
Folic Acid: Ideally, folic acid should be present in a patient’s multivitamin at a dosage of 400 to 800 mcg/day.1 Folic acid can be supplemented when a deficiency is expected. Signs of a deficiency include skin, nail, or mucosal changes.1 Women of childbearing age should also take higher doses of folic acid to reduce the risk of fetal neural-tube defects.1
Copper and Zinc: Both copper and zinc should be included in a bariatric surgery patient’s daily multivitamin at a dosage of 2 mg/day and 8 to 22 mg/day, respectively.1 Copper and zinc screening should be considered in patients who have undergone BPD/DS or RYGB. These patients may require higher doses to avoid deficiencies. Zinc replacement can lead to copper deficiency; therefore, supplementation with 1 mg of copper is recommended for every 8 to 15 mg of elemental zinc.1
Thiamine: All bariatric surgery patients should have thiamine supplementation. The preferred dosage is >12 mg of thiamine daily. This is typically supplied in a B-complex supplement containing 50 to 100 mg of thiamine or in a high-potency multivitamin. Patients with a mild-to-severe deficiency should receive IV thiamine.1
Overall, nutritional supplementation for bariatric surgery patients is complex. With an entire pharmacy aisle of products to select from, patients may find themselves overwhelmed and confused about which option to pick. Pharmacists can help patients make a proper supplement selection for their bariatric surgery type.
Conclusion
Bariatric surgery is a common procedure many patients turn to for weight loss. The procedure is currently guideline-recommended for patients with a minimum BMI of 30 kg/m2 plus other health factors. Types of bariatric surgery include sleeve gastrectomy, laparoscopic adjustable gastric band, RYGB, and BPD/DS. Pharmacists must understand the differences between these surgery types and in what ways the restrictive surgeries, malabsorptive surgeries, and combination surgeries impact the known pharmacokinetics of medications and nutritional supplements. Pharmacists can help patients optimize their medication and supplement selections to meet their specific needs.
REFERENCES
1. Mechanick JI, Apovian C, Brethauer S, et al. Clinical practice guidelines for the perioperative nutrition, metabolic, and nonsurgical support of patients undergoing bariatric procedures—2019 update: cosponsored by American Association of Clinical Endocrinologists/American College of Endocrinology, the Obesity Society, American Society for Metabolic & Bariatric Surgery, Obesity Medicine Association, and American Society of Anesthesiologists. Surg Obes Relat Dis. 2020;16(2):175-247.
2. American Society for Metabolic and Bariatric Surgery. Estimate of bariatric surgery numbers, 2011-2021. https://asmbs.org/resources/estimate-of-bariatric-surgery-numbers. Accessed July 12, 2023.
3. Lorico S, Colton B. Medication management and pharmacokinetic changes after bariatric surgery. Can Fam Physician. 2020;66(6):409-416.
4. Miller AD, Smith KM. Medication and nutrient administration considerations after bariatric surgery. Am J Health Syst Pharm. 2006;63(19):1852-1857.
5. Martin KA, Beyer-Westendorf J, Davidson BL, et al. Use of direct oral anticoagulants in patients with obesity for treatment and prevention of venous thromboembolism: updated communication from the ISTH SSC Subcommittee on Control of Anticoagulation. J Thromb Haemost. 2021;19(8):1874-1882.
6. Bland CM, Quidley AM, Love BL, et al. Long-term pharmacotherapy considerations in the bariatric surgery patient. Am J Health Syst Pharm. 2016;73(16):1230-1242.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
To comment on this article, contact rdavidson@uspharmacist.com.






