US Pharm. 2014;39(12):44-48.
ABSTRACT: Celiac disease is a chronic condition involving an abnormal immune response to the ingestion of gluten-containing foods and products that commonly results in digestive symptoms, although other organ systems may be involved. The current mainstay of therapy is the avoidance of gluten-containing foods, beverages, and other products. However, if not equipped with the knowledge that medications, OTC products, supplements, and vitamins may contain gluten, patients with celiac disease may experience ongoing symptoms from continued ingestion of these products. Therefore, pharmacists play an essential role in educating patients and evaluating their medication use to ensure the optimal management of celiac disease.
Celiac disease (CD) is a chronic autoimmune disorder that is characterized by a multisystem immune-mediated inflammatory response to the ingestion of gluten in genetically predisposed individuals.1 Inflammation in the gastrointestinal (GI) tract results in the malabsorption of nutrients, triggering a cascade of clinical symptoms in individuals who are either unaware of the condition or noncompliant with the completely gluten-free diet (GFD) required in the case of known CD.2 Recent studies have shown that drug absorption may be impaired in CD patients who require oral medications for comorbid conditions.3 This suggests that it is imperative for the pharmacist to not only identify patients with known or suspected CD and closely monitor the efficacy and safety of any orally administered medications, but also be aware of gluten-containing excipients in medications to ensure that CD patients avoid their use.
Etiology and Risk Factors
CD affects an estimated 0.2% to 1% of the U.S. population, with a higher incidence in females, and its prevalence has increased.1,4,5 The disorder is characterized by an autoimmune reaction to gluten that causes inflammation of the mucosa of the small intestine, damaging the villi of the epithelial lining and leading to obstructed nutrient absorption.6 Gluten is a protein found in wheat, barley, and rye (containing gliadin, hordein, and secalin glycoproteins, respectively). Gliadin, which is not fully broken down by the intestine, may pass through the intestinal mucosa in patients with gluten intolerance, producing an immune response by activating class II human leukocyte antigen (HLA)-DQ2 and HLA-DQ8 T-cell genetic molecules.1,5
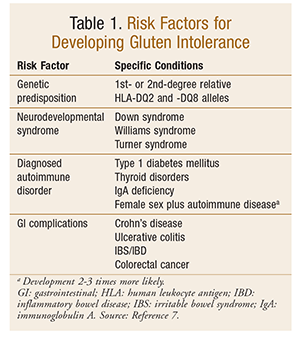
The gluten intolerance specifically characterized by CD is genetically based and typically develops in individuals with a familial (first- or second-degree relative) history of immune intolerance to the protein in gluten. Genetic predisposition of the HLA-DQ2 and HLA-DQ8 alleles is correlated with the development of gluten sensitivity.1,7 Individuals with neurodevelopmental syndromes, such as Down, Williams, and Turner syndromes, have a higher prevalence of CD. Additionally, patients with diagnosed autoimmune disease, such as type 1 diabetes mellitus, thyroid disorders, and immunoglobulin (Ig) A deficiency, are at greater risk for developing CD. See TABLE 1.7
The exact cause of autoimmune gluten intolerance is unknown, but various contributing factors to CD development have been suggested. Possible triggers include viral infections such as Giardia lamblia, rotavirus, enterovirus, Campylobacter jejuni, hepatitis C, and adenovirus type 12; gluten consumption during the first year of life; decreased protection against infection from early termination of breastfeeding; and tissue damage.8 The introduction of gluten in infant diets should occur between 3 and 7 months of age to decrease the risk of developing intolerance.1,9 Cesarean delivery and rotavirus infection have been suggested as potential risk factors for CD development owing to the disruption of normal flora in neonates and children.1 All potential risk factors for CD that occur during pediatric development have unknown mechanisms and require further investigation.
To prevent consumption of gluten, patients with CD must avoid all wheat, barley, and rye products. Exposure to as little as 30 to 50 mg of the protein (roughly 1/48th the size of a slice of bread) per day can damage the mucosa of the small intestine.5,10
Clinical Presentation and Diagnosis
CD pathogenesis involves both environmental and genetic factors, including several genes that predispose an individual to the disease.7 Depending upon the trigger, symptoms may appear in early childhood or later in life. Typically, patients present with symptoms that are secondary to malabsorption of nutrients; however, some present with nonspecific GI discomfort or extraintestinal symptoms, either alone or in addition to manifestations of malabsorption (TABLE 2). For this reason, there is often a delay in diagnosing CD, particularly because patients are commonly misdiagnosed with other disorders prior to confirmation of CD. Misdiagnoses include irritable bowel syndrome, inflammatory bowel disease, gastroesophageal reflux disease, ulcers, viral gastroenteritis, chronic fatigue syndrome, allergies, parasitic infection, gallbladder disease, colitis, cystic fibrosis, psychological dysfunction, and lactose intolerance.5

Lactose intolerance is a common misdiagnosis in CD patients because the mucosal injury renders them unable to digest lactose-containing products. Malabsorption secondary to mucosal injury also explains nutritional deficiencies of the fat-soluble vitamins A, D, E, and K, as well as the B vitamins, thereby diminishing the absorption of iron, calcium, and folic acid.1 As a result, patients may develop iron-deficiency anemia refractory to oral iron supplementation, and potentially osteoporosis and osteopenia due to bone loss secondary to decreased calcium and vitamin D absorption.1 Reproductive abnormalities, such as delayed puberty, secondary amenorrhea, infertility, or subfertility, may be explained by the combination of nutritional deficiencies and the damaging effects of systemwide chronic inflammation.11 Other common manifestations, such as dermatitis herpetiformis (DH; a papulovesicular rash), result from the immunologic responses to gluten ingestion.1
While all persons with CD are intolerant to gluten, not all individuals who are intolerant to gluten have CD. Recent studies have confirmed the existence of nonceliac gluten sensitivity (NCGS), a hypersensitivity or form of gluten intolerance wherein individuals experience a complex of symptoms similar to that of CD. Unlike CD, NCGS is not hereditary and is not associated with malabsorption, nutritional deficiency, or an increased risk of autoimmune disorders or intestinal malignancies.3,7 Furthermore, immunologic mechanisms or serologic markers for NCGS have not yet been identified.3 Diagnosis of NCGS is made by excluding both CD and an IgE-mediated allergy to wheat, and is otherwise based on the manifestation of symptoms associated with gluten consumption.
According to the American College of Gastroenterology’s (ACG) clinical guideline on diagnosis and management of celiac disease, CD should be tested for if the patient has signs and symptoms of malabsorption (e.g., chronic diarrhea with weight loss, steatorrhea, postprandial abdominal pain, bloating) or laboratory evidence of malabsorption, particularly in the case of a first-degree family member with a confirmed diagnosis of CD, a personal history of an autoimmune disease, or an IgA deficiency.7 CD should also be suspected in patients with biopsy-proven DH, iron-deficiency anemia refractory to oral supplementation, or hypertransaminasemia with no other etiology.7
In all cases, the adoption of a lifelong GFD has been shown to relieve symptoms, and in CD patients it has been shown to normalize serologic markers of CD and restore intestinal villi otherwise degenerated by inflammatory reactions.3 Nonadherence to the GFD can result in serious complications associated with malabsorption, including fractures secondary to low bone mineral density, and in some cases intestinal malignancies such as intestinal T-cell lymphomas, small-bowel adenocarcinoma, esophageal cancer, and B- and T-cell non-Hodgkin lymphomas.7
Factors That May Impair Drug Absorption
Gluten is a common ingredient in many commercial food products. Less commonly known, however, is that gluten may be used in the manufacture of such unsuspected products as medications, supplements, and vitamins. Although product selection can be a nuisance, it is extremely important for persons with CD to review the nutrition labels of all foods and beverages, as well as the package inserts (PI) of medications and supplements, prior to ingestion.12
Gluten’s role in small-intestinal inflammation and damage over time results in decreased absorption of common dietary nutrients and therefore likely causes malabsorption of oral medications, as well.13 GI complications that can also hinder nutrient and drug absorption include impaired gastric-emptying rate, abnormal permeability, pH changes, decreased epithelial surface area, and reduced metabolizing enzymes.13 Oral administration of most medications relies on absorption through the small intestine via passive diffusion, in which drug molecule characteristics play a large role.13 GI-tract abnormalities may shift this diffusion process into systemic circulation, ultimately resulting in increased or decreased absorption of drug molecules. Since drug molecules have varying and unique chemical properties, it is difficult to determine the exact mechanism of absorption for all drugs in patients with CD. Based on their molecular properties, drugs currently under investigation for their absorption characteristics in gluten sensitivity include acetaminophen, aspirin, indomethacin, levothyroxine, prednisolone, propranolol, and certain antibiotics.13 Routine monitoring of serum drug levels for medications with narrow therapeutic indexes may be warranted in CD.
Legislation
Increased awareness of CD in recent years not only has identified more cases of CD, but also has sparked legislation on gluten-free labeling. In May 2013, H.R. 2003, the Gluten in Medicine Disclosure Act of 2013, was introduced and referred to the Committee on Energy and Commerce to amend the Federal Food, Drug, and Cosmetic Act. Enactment of this bill would have amended the law to require the labeling of drugs intended for human use to contain a statement identifying the source of any ingredient made of or derived from a grain or starch-containing ingredient.14 This bill did not move forward, however. Neither did a similar bill that preceded it in April 2012, H.R. 4972, Gluten in Medicine Identification Act of 2012.15
Previously, the Food Allergen Labeling and Consumer Protection Act (FALCPA) of 2004, which identified eight food groups as major allergens, was passed by Congress.15 FALCPA required food manufacturers to include the word “Contains” followed by the name of the food source from which the major allergen (e.g., wheat) was derived or to give the common name of the major allergen in the list of ingredients, including the name of the food source.16
Although the FALCPA regulations safeguarded consumers seeking to identify wheat-containing products, they did not apply to rye and barley, which also contain gluten. FALCPA did, however, require the FDA to issue a proposed rule to define and permit voluntary use of the term “gluten-free” on food labels. In August 2013, the FDA issued a final rule under FALCPA on the use of the term “gluten-free” in food labeling for manufacturers choosing to use the term. Manufacturers who labeled FDA-regulated foods and dietary supplements as “gluten-free,” “no gluten,” “free of gluten,” or “without gluten” were given until August 5, 2014, to comply with the new rules and ensure that the product contained no more than 20 parts per million of gluten-containing ingredients or derivatives (i.e., 20 mg gluten/kg of food). Failure to meet the requirements for the claim would be considered misbranding.17
At present, no legislation is in place to facilitate the identification of gluten-containing ingredients in prescription and nonprescription medications.
Management
Currently, no medications are available to manage symptoms or prevent small-intestinal damage associated with gluten ingestion in CD.7 Management of this chronic condition relies solely on the permanent avoidance of gluten-containing foods and products, which may necessitate significant changes in lifestyle, since gluten is a ubiquitous component of food, beverages, medications, and supplements.5,7
Avoiding Gluten-Containing Foods and Beverages: A working knowledge of common foods that contain gluten and how to inspect food labels to determine the presence of gluten-containing additives (e.g., emulsifiers and stabilizers) is essential for preventing symptoms and secondary complications of CD. Wheat, barley, and rye contain gluten and should be avoided. Buckwheat, corn, flax, potato, quinoa, rice, soybean, and tapioca flours are considered gluten-free and safe to consume. Consumption of pure oats in limited quantities is considered safe for most people with CD.5,18 However, oats may be contaminated with gluten during the manufacturing process; therefore, they should be carefully introduced with ongoing monitoring of symptoms.7 The ACG strongly recommends that CD patients be referred to a registered dietitian for nutritional counseling.7 In addition to food products, individuals with CD should avoid alcoholic beverages containing gluten (i.e., most beers, ales, lagers, and malt vinegars, although some varieties are specifically marketed as gluten-free and are safe for consumption).5 Distilled alcohol drinks and wine are safe to ingest.5
Avoiding Gluten-Containing Medications, OTC Products, Supplements, and Vitamins: As previously mentioned, manufacturers of prescription or OTC medications are not required to disclose on the package labeling whether the product contains gluten. Consequently, it is important to investigate each orally ingested medication, supplement, and vitamin for the presence of any ingredients or excipients that may contain gluten. This often can be accomplished by evaluating the ingredient list, contacting the manufacturer, or utilizing a variety of other resources. Gluten can potentially be introduced and contaminate otherwise gluten-free products during the manufacturing process, although the likelihood is low.6 Key points to consider are that even if a brand product is confirmed to be gluten-free, it cannot be assumed that the generic version is also gluten-free, and that if a product has a new formulation, appearance, or manufacturer, it is prudent to reassess it and confirm that it remains gluten-free.6
When evaluating the gluten content of prescription and OTC products, it should be remembered that gluten can be masked in an excipient. Starches used as excipients in pharmaceutical products are often derived from rice, potato, or tapioca, which are gluten-free. However, if the source of the starch is not explicitly stated, the excipients may contain gluten. Sources of excipients that contain gluten include barley, farina, kamut, rye, spelt, triticale, and wheat.19,20 See TABLE 3 for a list of excipients that may contain gluten and should be investigated to determine the source of the starch.6

Excipients that are considered safe for consumption in individuals with gluten intolerance include sweeteners such as glucose, fructose, and corn syrups, as well as fillers, thickening agents, and polymers such as gums and cellulose derivatives.5,6 Sugar alcohols such as mannitol and sorbitol, which commonly are used as sweeteners and bulking agents, are gluten-free, but if ingested in large quantities can cause gluten-like symptoms, such as diarrhea.21 Barley-based brown rice syrup, wheat-based dextrin, and maltodextrin should be avoided.5
Additional Considerations: Other manifestations of CD include nutritional deficiencies and bone loss secondary to malabsorption. It is recommended that CD patients be assessed for nutritional deficiencies (vitamins A, D, E, and B12, carotene, copper, iron, folic acid, magnesium, selenium, and zinc) and receive supplementation, if needed.7 Patients should be screened for osteopenia or osteoporosis and should be managed accordingly, if needed, to ensure that the recommended dietary allowance for calcium is being met.7
Prophylactic administration of the pneumococcal vaccine may be reasonable in this population, given that one-third of adults with CD have inadequate splenic function.22 At this writing, the CDC has not provided any formal recommendations specifically for the use of the pneumococcal vaccine in CD.
Resources
Given the lack of validated resources to verify the gluten content of prescription and nonprescription medications, it is best to check with the manufacturer, especially since there may be conflicting information in what appears in published materials and what is provided by the manufacturer.18 Utilizing PIs, contacting pharmaceutical manufacturers, and visiting websites can assist pharmacists in determining whether a medication potentially contains gluten.
For prescription medications, the PI should include a detailed listing of excipients; however, if this information is not readily available, the FDA provides drug labeling information for prescription and some OTC medications at DailyMed (dailymed.nlm.nih.gov). For nonprescription products, there often is nothing in the PI regarding gluten content, necessitating a call to the manufacturer for verification.
Manufacturer contact information may be located by checking the product or its packaging, conducting an Internet search using the manufacturer’s name, or accessing drug-information resources (online databases) such as Clinical Pharmacology, Facts & Comparisons, and Martindale. When requesting information from a manufacturer, it is helpful to provide the lot number. Exploratory research by Mangione and colleagues concluded that information concerning the gluten content of nonprescription products is often readily available and easy to access if the manufacturer is contacted.18
Additionally, there are some websites that may assist pharmacists in identifying gluten-free products. GlutenFreeDrugs.com, which is maintained by a clinical pharmacist, contains a detailed chart listing selected brand and generic medications that are gluten-free, as well as those free of lactose or soy. (Note that this is not an all-encompassing list of products.) At Celiac.org, the Celiac Disease Foundation offers a variety of resources and provides information on the treatment of CD, tips on living gluten-free, and support-group contact information.
Conclusion
Pharmacists play a pivotal role in educating patients about gluten-containing foods, medications, and supplements in order to help them adhere to a GFD and in ensuring that patients receive additional follow-up care, if needed. CD patients may not have gluten documented as an allergy in their medical chart, and if it is listed, it may not integrate with computerized interaction alerts. Therefore, pharmacists can identify products that may contain gluten by reviewing the contents of each product, contacting the manufacturer, and utilizing Internet resources.
REFERENCES
1. Pelkowski TD, Viera AJ. Celiac disease: diagnosis and management. Am Fam Physician. 2014;89:99-105.
2. Crowe SE. In the clinic. Celiac disease. Ann Intern Med. 2011;145:ITC5-1–ITC5-15.
3. Tonutti E, Bizzaro N. Diagnosis and classification of celiac disease and gluten sensitivity. Autoimmun Rev. 2014;13:472-476.
4. Mooney P, Hadjivassiliou M. Coeliac disease. BMJ. 2014;348:g1561.
5. Mangione RA, Patel PN. Caring for patients with celiac disease: the role of the pharmacist. J Am Pharm Assoc (2003). 2008;48:e125-e135.
6. Plogsted S. Medications and celiac disease—tips from a pharmacist. Pract Gasteroenterol. 2007;31:58-64.
7. Rubio-Tapia A, Hill ID, Kelly CP, et al. ACG clinical guidelines: diagnosis and management of celiac disease. Am J Gastroenterol. 2013;108:656-676.
8. Gujral N, Freeman HJ, Thomson AB. Celiac disease: prevalence, diagnosis, pathogenesis and treatment. World J Gastroenterol. 2012;18:6036-6059.
9. Henriksson C, Boström AM, Wiklund IE. What effect does breastfeeding have on coeliac disease? A systematic review update. Evid Based Med. 2013;18:98-103.
10. Adams S. Celiac disease and gluten-free diet. www.celiac.com. Accessed July 30, 2014.
11. Soni S, Badawy SZ. Celiac disease and its effect on human reproduction: a review. J Reprod Med. 2010;55:3-8.
12. King AR. Gluten content of the top 200 medications: follow-up to the influence of gluten on a patient’s medication choices. Hosp Pharm. 2013;48:736-743.
13. Tran TH, Smith C, Mangione RA. Drug absorption in celiac disease. Am J Health Syst Pharm. 2013;70:2199-2206.
14. GovTrack.us. Text of the Gluten in Medicine Disclosure Act of 2013. https://www.govtrack.us/congress/bills/113/hr2003/text. Accessed November 14, 2014.
15. FDA. Food Allergen Labeling and Consumer Protection Act of 2004 (Public Law 108-282, Title II). www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/Allergens/ucm106187.htm. Accessed November 14, 2014.
16. FDA. Gluten-free labeling of foods. www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/Allergens/ucm362510.htm. Accessed November 14, 2014.
17. GovTrack.us. Text of the Gluten in Medicine Identification Act of 2012. https://www.govtrack.us/congress/bills/112/hr4972/text. Accessed November 14, 2014.
18. Mangione RA, Patel PN, Shin E, Fiebert J. Determining the gluten content of nonprescription drugs: information for patients with celiac disease. J Am Pharm Assoc. 2011;51:734-737.
19. Crowe JP, Falini NP. Gluten in pharmaceutical products. Am J Health Syst Pharm. 2001;58:396-401.
20. Cacace JL. Formulating for the gluten-sensitive individual. Int J Pharm Compd. 2005;9:357-358.
21. Gluten Intolerance Group. Medications & celiac disease. https://www.gluten.net/wp-content/uploads/2013/12/EDU_MdctnsCD_12.11.2013.pdf. Accessed July 17, 2014.
22. Di Sabatino A, Brunetti L, Carnevale Maffè G, et al. Is it worth investigating splenic function in patients with celiac disease? World J Gastroenterol. 2013;19:2313-2318.
To comment on this article, contact rdavidson@uspharmacist.com.





