US Pharm. 2009;34(4):HS8-HS15.
Methicillin-resistant Staphylococcus aureus (MRSA) skin and soft tissue infections (SSTIs) are a growing issue worldwide with significant economic impact. As a pharmacist, one must be familiar with the risk factors for infection as well as the treatment options that are available today. The pharmacist is often the first-line health care professional that people seek, and he or she should be able to make an educated professional referral based upon the discussion with the patient. Patients need to be educated regarding the risk factors of a staphylococcal infection in order to seek medical attention when necessary.
Etiology
According to the CDC, Staphylococcus aureus is a species of bacteria commonly carried on the skin or in the nose of healthy people. Approximately 25% to 30% of the population is colonized (i.e., bacteria are present but not causing an infection) in the nose with S aureus bacteria.1 MRSA is a type of S aureus that is resistant to beta-lactam antibiotics such as methicillin, oxacillin, penicillin, and amoxicillin. MRSA is significantly more likely to be resistant to erythromycin, ciprofloxacin, clindamycin, gentamicin, and trimethoprim-sulfamethoxazole (TMP-SMZ) than methicillin-susceptible S aureus (MSSA) isolates.2 Only about 1% of the population is colonized with MRSA.1 MRSA infections that are acquired by persons who have not been recently hospitalized (within the past year) or had a medical procedure (i.e., dialysis, surgery, catheters) are known as community-associated MRSA (CA-MRSA) infections. MRSA and other S aureus infections in the community are usually manifested as skin infections, such as pimples and boils, and occur in otherwise healthy people.1 The anatomical site of infection does not seem to affect the clinical outcome of therapy.3
In a study of all MRSA isolates in San Francisco, CA-MRSA isolates were less likely to be resistant to antibiotics than were hospital-associated MRSA (HA-MRSA) isolates (P <.0001).2 This is logical, as CA-MRSA has a shorter history than nosocomial or HA-MRSA. In that same study, a large proportion of CA-MRSA strains were originally hospital clones that over time moved into the community. Other strains were strictly community based.2
Impact of MRSA
The reach of MRSA extends worldwide, from the United States to Asia and from the Netherlands to Australia. It has been reported in at least 34 countries.4 In our own backyard, cases have been reported in at least 31 states (TABLE 1).4 The SENTRY Antimicrobial Surveillance Program (1997-1999) reported that 39.2% of all SSTIs were due to S aureus. The SENTRY centers were located in the U.S., Canada, Latin America, Europe, and the Western Pacific Region. S aureus was the most common cause of infection. In the U.S., 41.6% of all infections were to due to S aureus, of which 30% were resistant to methicillin. During the three years of the SENTRY program, the resistance rates of both nosocomial and community-onset S aureus increased. MRSA strains that were resistant to methicillin were more likely to be resistant to other antimicrobial agents.5
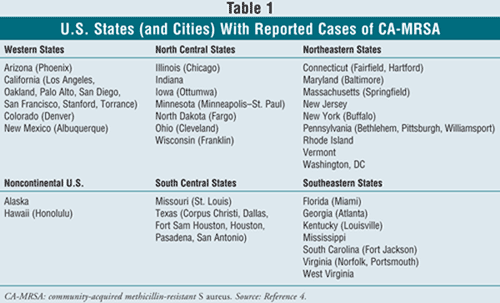
The Institute of Medicine estimates the annual cost of infections caused by antibiotic-resistant bacteria in the U.S. to be $4 to $5 billion.6 In a study of the economic impact on the Canadian health care system, cost data combined with epidemiologic data suggested that the direct health care cost associated with MRSA was $54 million to $110 million annually. The total average cost per patient infected with MRSA was $12, 216. This included hospitalization (81%), barrier precautions (13%), antimicrobial therapy (4%), and laboratory investigations (2%).7 In U.S. studies, total reported cost per MRSA patient has ranged between $16,575 and $39,400.7 The cost of MRSA SSTIs has been reported as $23,616.8
MRSA and Athletes
CA-MRSA can spread more easily among athletes.9 This subset of patients is at a higher risk for infection for multiple reasons. MRSA is such a widespread problem that the CDC has devoted many resources to the education of athletes and athletic departments, as evidenced by the volume of resources and information available on its Web site. Athletes spread MRSA to each other more readily due to many factors, including9:
• Repeated skin-to-skin contact
• Contact with shared items or surfaces (e.g., bars of soap, towels, used bandages, weight-training equipment surfaces)
• Breaks in the skin, such as cuts and abrasions
• Difficulty staying clean.
In a study of the St. Louis Rams during the 2003 football season, players reported that skin abrasions were more frequent and severe when competition took place on artificial turf instead of natural grass.10 In areas where wound care and physical therapy took place, trainers did not have regular access to hand hygiene, including alcohol-based hand-sanitizer products. As the CDC reported, towels were shared on the field during practice and games with as many as three players using the same towel. Another important item to note, although not statistically significant, is that use of antimicrobial agents during the previous year was associated with MRSA infection. During that year, each team player received an average of 2.6 antimicrobial-drug prescriptions. This is more than 10 times the rate of the general population for that age and sex. A nasal-swab survey of St. Louis Rams players and staff indicated that 40% of players and 46% of staff were colonized with MSSA but not MRSA.10
Prevention
Patients, caregivers, and the general public should be educated on ways to decrease the spread of MRSA. Some easily implemented methods that should be shared with patients include regular hand washing and proper hygiene. These methods and others are described in TABLE 2.11 On September 8, 2008, the CDC launched the National MRSA Education Initiative. The goal of this initiative is to help Americans better recognize and prevent MRSA skin infections. Through this program, health care professionals have access to consumer and professional educational materials including posters, brochures, podcasts, pictures, e-cards, and more (call 1-800-CDC-INFO or visit www.cdc.gov/MRSA for further details).12

The Environmental Protection Agency (EPA) publishes a list, known as List H, which contains registered products effective against MRSA.13 List H contains hospital cleaners as well as readily available consumer cleaners that can be recommended to patients. This would be an excellent place to start when creating a prevention plan.
An innovative study is examining whether or not a tea tree oil body scrub is effective against MRSA in gravely ill hospitalized adults. This trial was started in 2007 and will be completed in 2010.14
Treatment Options
Before antimicrobial therapy is discussed, it is important to note that incision and drainage of fluctuant lesions is shown to have benefit in studies and is recommended by current guidelines.9,10,15 In 2005, the Infectious Diseases Society of America (IDSA) developed and issued guidelines that practitioners should follow for the diagnosis and management of SSTIs.15 Oral treatment options for MRSA SSTIs, as stated by the guidelines, are clindamycin, doxycycline, minocycline, and TMP-SMZ. IV options include daptomycin, linezolid, and vancomycin, the latter being the parenteral drug of choice (TABLE 3). More data from controlled clinical trials are needed to establish optimal regimens for the treatment of MRSA SSTIs.16
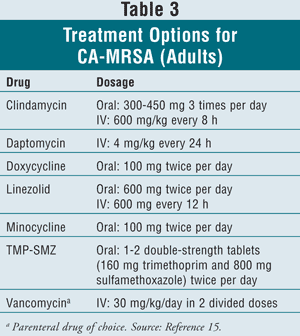
Any patient with potential for MRSA, when sent home with a treatment regimen, should be reevaluated in 24 to 48 hours to ensure clinical response. If the infection is progressing, it could be due to a resistant strain or a deeper underlying infection. Please refer to the full IDSA guidelines for complete management options.15
In a study conducted in the second half of 2006, 332 emergency room (ER) physicians were surveyed regarding treatment of MRSA SSTIs.17 If MRSA was suspected in a patient who was well enough to be discharged home from the ER, these physicians would prescribe, alone or in combination, the following drugs: vancomycin (18%), TMP-SMZ (55%), clindamycin (22%), rifampin (14%), and cephalexin (14%). Patients who required admission were given vancomycin IV (65%), TMP-SMZ (15%), and linezolid (4%).
Other treatment options not mentioned in the guidelines include injectable daptomycin and tigecycline. Cyclic lipopeptide and glycylcycline, respectively, are FDA approved for complicated skin and soft tissue structure infections, which can include MRSA.18,19
Because many systemic agents that are used for MRSA SSTIs achieve poor concentrations on the skin surface, topical agents are often used. These agents are used to address surface colonization as well as active infection.20 Agents used include mupirocin, chlorhexidine, and triple antibiotic ointment containing neomycin, polymyxin B, and bacitracin.
It is important to treat MRSA SSTIs immediately. Lack of antibiotic therapy within the first 48 hours after incision or positive swab is independently associated with treatment failure.3
Complementary and Alternative Medicine
Patients may try to treat SSTIs with complementary and alternative medicine (CAM). It is important for a clinician to understand what the literature states about such methods in order to help educate patients. One such example is the topical application of components of eucalyptus oil that have been reported to show clinical activity in the setting of MRSA skin infections.20
A study conducted on three types of honey produced in Northern Ireland and one honey produced in France demonstrated that, in vitro, the samples had an antimicrobial activity against CA-MRSA. Each honey was able to reduce the cultural count of all CA-MRSA to none detectable within 24 hours. In addition, inoculated honey remained positive for CA-MRSA until 72 hours postinoculation, at which point no culturable organisms could be detected.21
In an ethnobotanical survey carried out in the Akwapim-North district of the Republic of Ghana, 25 plant species, used in traditional medicine to treat skin disease and/or antimicrobial (viral, bacterial, or protozoan) infections were identified.22 While many of the species showed antimicrobial activity, Alchornea cordifolia inhibited the growth of all 15 MRSA isolates similarly to or better than the other plant species against corresponding bacteria. MRSA was also less inhibited by the extracts of the other species tested.
While the topic of CAM is exciting and the research promising, it is important to educate patients that much more research is needed and to continue with their prescribed therapy.
Future Therapies
The future is very promising for both systemic and topical antibiotic therapies effective against MRSA SSTIs. Telavancin is showing promising results in clinical trials and is under regulatory review by the FDA.23 Dalbavancin is a parenterally administered semisynthetic lipoglycopeptide that may be FDA approved soon.24 Ceftobiprole, a cephalosporin available in Canada, is currently in development for the U.S. and showing unique characteristics against MRSA.25 With current strategies for the prevention and management of MRSA SSTIs, in addition to novel agents on the way, MRSA will hopefully become a bug of the past.
REFERENCES
2. Charlebois ED, Perdreau-Remington F, Kreiswirth B, et al. Origins of community strains of methicillin-resistant Staphylococcus aureus. Clin Infect Dis. 2004;39:47-54.
3. Elston D. Topical antibiotics in dermatology: emerging patterns of resistance. Dermatol Clin. 2009;27:25-31.
4. Cohen PR. Cutaneous community-acquired methicillin-resistant Staphylococcus aureus infection: a personal perspective of a worldwide epidemic. Expert Rev Dermatol. 2006;1:631-637.
5. Diekema DJ, Pfaller MA, Schmitz FJ, et al. Survey of infections due to Staphylococcus species: frequency of occurrence and antimicrobial susceptibility of isolates collected in the United States, Canada, Latin America, Europe, and the Western Pacific Region for the SENTRY Antimicrobial Surveillance Program, 1997-1999. Clin Infect Dis. 2001;32(suppl 2):S114-S132.
6. McGowan JE Jr. Economic impact of antimicrobial resistance. Emerg Infect Dis. 2001;7:286-292.
7. Goetghebeur M, Landry PA, Han D, Vicente C. Methicillin-resistant Staphylococcus aureus: a public health issue with economic consequences. Can J Infect Dis Med Microbiol. 2006;18:27-34.
8. Shah NP, Reddy P, Paladino JA, et al. Direct medical costs associated with using vancomycin in methicillin-resistant Staphylococcus aureus infections: an economic model. Curr Med Res Opinion. 2004;20:779-790.
9. About methicillin-resistant Staphylococcus aureus (MRSA) among athletes. Updated November 2008. www.cdc.gov/ncidod/dhqp/ar_MRSA_AthletesFAQ.html. Accessed January 14, 2009.
10. Kazakova SV, Hageman JC, Matava M, et al. A clone of methicillin-resistant Staphylococcus aureus among professional football players. N Engl J Med. 2005;352:468-475.
11. Gorwitz RJ, Jernigan DB, Powers JH, and Participants in the CDC Convened Experts’ Meeting on Management of MRSA in the Community. Strategies for clinical management of MRSA in the community: summary of an experts’ meeting convened by the CDC. March 2006. www.cdc.gov/ncidod/dhqp/pdf/
12. CDC steps up efforts to fight drug-resistant germ. Press release. September 8, 2008. www.cdc.gov/media/pressrel/
13. U.S. EPA Office of Pesticide Programs. List H: EPA’s registered products effective against methicillin resistant Staphylococcus aureus (MRSA) and vancomycin resistant Enterococcus faecalis or faecium (VRE). June 30, 2008. www.epa.gov/oppad001/list_h_
14. Thompson G, Blackwood B, McMullan R, et al. A randomized controlled trial of tea tree oil (5%) body wash versus standard body wash to prevent colonization with methicillin-resistant Staphylococcus aureus (MRSA) in critically ill adults: research protocol. BMC Infect Dis. 2008;8:161.
15. Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft-tissue infections. Clin Infect Dis. 2005;41:1373-1406.
16. Gorwitz RJ, Jernigan DB, Powers JH, et al. Strategies for clinical management of MRSA in the community: summary of an experts’ meeting convened by the Centers for Disease Control and Prevention. March 2006. www.cdc.gov/ncidod/dhqp/ar_
17. LoVecchio F, Perera N, Casanova L, et al. Board-certified emergency physicians’ treatment of skin and soft tissue infections in the community-acquired methicillin-resistant Staphylococcus aureus era. Am J Emerg Med. 2009;27:68-70.
18. Ruhe JJ, Smith N, Bradsher RW, Menon A. Community-onset methicillin-resistant Staphylococcus aureus skin and soft-tissue infections: impact of antimicrobial therapy on outcome. Clinl Infect Dis. 2007;44:777-784.
19. Cubicin (daptomycin for injection) package insert. Lexington, MA: Cubist Pharmaceuticals, Inc.; August 2008.
20. Dunbar LM, Tang DM, Manausa RM. A review of telavancin in the treatment of complicated skin and skin structure infections (cSSSI). Ther Clin Risk Manag. 2008:4:235-244.
21. Maeda Y, Loughrey A, Earle JA, et al. Antibacterial activity of honey against community-associated methicillin-resistant Staphylococcus aureus (CA-MRSA). Complement Ther Clin Pract. 2008;14:77-82.
22. Pesewu GA, Cutler RR, Humber DP. Antibacterial activity of plants used in traditional medicines of Ghana with particular reference to MRSA. J Ethnopharmacol. 2008;116:102-111.
23. Townsend ML, Pound MW, Drew RH. Tigecycline in the treatment of complicated intra-abdominal and complicated skin and skin structure infections. Ther Clin Risk Manag. 2007;3:1059-1070.
24. Bennett JW, Lewis JS, Ellis MW. Dalbavancin in the treatment of complicated skin and soft-tissue infections: a review. Ther Clin Risk Manag. 2008:4:31-40.
25. Anderson SD, Gums JG. Ceftobiprole: an extended-spectrum anti–methicillin-resistant Staphylococcus aureus cephalosporin. Ann Pharmacother. 2008;42:806-816.
To comment on this article, contact rdavidson@jobson.com.





