US Pharm. 2016;41(3):HS18-HS.
ABSTRACT: Hyperphosphatemia is an abnormally high level of serum phosphate that contributes to chronic kidney disease (CKD). The management of hyperphosphatemia has included dietary phosphate restriction and use of phosphate binders. The first phosphate binders were aluminum- and magnesium-based antacids. Adverse effects and toxicity limited the use of these agents, and therapy evolved with calcium carbonate, calcium acetate, sevelamer, and lanthanum carbonate. Recently, two iron-based phosphate binders have been approved. Hyperphosphatemia in CKD stages 1 to 3 can typically be controlled with dietary changes. Calcium-based products are often started in stage 4 secondary to efficacy, safety, and cost. In CKD stage 5, hypercalcemia can increase the risk of cardiovascular disease. In this situation, sevelamer and lanthanum have demonstrated a cardiovascular mortality benefit. Sucroferric oxyhydroxide and ferric citrate are calcium-free and may offer benefits in those with a high pill burden and in patients with concurrent anemia, respectively.
Phosphorus is an electrolyte found primarily in the bones (80%-85%) and in the intracellular fluid.1 It is a major anion and is used as the source for the synthesis of adenine triphosphate (ATP) and phospholipids. Serum phosphorus concentration is normally 2.7 to 4.5 mg/dL (0.87-1.45 mmol/L). Hyperphosphatemia is defined by a serum phosphorus concentration of >4.5 mg/dL (1.45 mmol/L). Causes of hyperphosphatemia include impaired phosphorus excretion (renal failure or hypoparathyroidism), redistribution of phosphorus to the extracellular fluid (acid-base imbalance, rhabdomyolysis, muscle necrosis, or tumor lysis during chemotherapy), and increased phosphate intake. Medications causing hyperphosphatemia include phosphorus-containing laxatives, oral phosphorus supplements, vitamin D supplements, and the bisphosphonates.1,2
It is essential for the pharmacist to recognize that in the setting of advanced chronic kidney disease (CKD), dialysis does not remove all phosphorus as it does other electrolytes, and many patients will require a phosphate binder. Fifty percent of mortality in patients with CKD is related to cardiovascular complications, with the highest risk being in the presence of hyperphosphatemia, hypercalcemia, and hyperparathyroidism.3 In general, the goal is to achieve a phosphorus concentration of 2.7 to 4.6 mg/dL in patients not receiving dialysis. The target phosphorus concentration for dialysis patients is 3.5 to 5.5 mg/dL.3
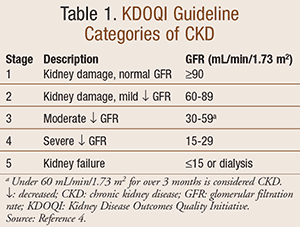
The Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines categorize CKD by stages (TABLE 1).4 These guidelines recommend that for high phosphorus uncontrolled by dietary measures, calcium-based phosphate binders are a reasonable choice for CKD stages 3 and 4. Stage 5 patients may use either calcium or non-calcium-based binders, and if a dialysis patient remains hyperphosphatemic (>5.5 mg/dL) it is reasonable to use a combination of both.4
Traditional Phosphate Binders
Dietary restriction of phosphate and protein is considered effective for most minor elevations of phosphorus. Phosphate binders such as aluminum-based antacids, magnesium-based antacids, calcium carbonate, calcium acetate, sevelamer, and lanthanum may be necessary for those patients whose phosphorus levels stay elevated despite dietary restrictions. TABLE 2 summarizes the place in therapy, dosing, adverse-effect profile, and patient considerations for these agents.
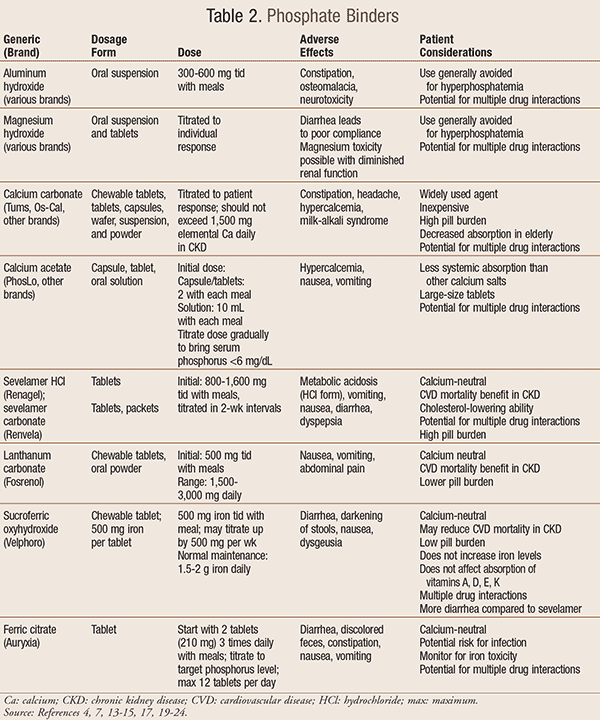
Aluminum Hydroxide: The antacid aluminum hydroxide (various formulations) is a phosphate binder used to treat hyperphosphatemia. This product is no longer considered a first-line agent, as long-term use is associated with constipation, aluminum toxicity, osteomalacia, and encephalopathy.5 Aluminum antacids may also decrease the absorption of many other medications such as fluoroquinolones, tetracyclines, and thyroid hormones. Additionally, studies have noted that this product is not as efficacious as calcium-based phosphate binders.6 For these reasons, aluminum hydroxide has been largely abandoned in current clinical practice.
Magnesium Hydroxide: The antacid/laxative magnesium hydroxide (various formulations) is available as tablets or oral suspension. Magnesium hydroxide has similar phosphate-lowering capacity compared to calcium-based agents and is infrequently used as add-on therapy.2 The most common adverse effect experienced by patients taking magnesium-based phosphate binders is diarrhea. Magnesium levels of patients on dialysis are typically higher than of those with normal renal function; use of magnesium salts may place a patient at risk for hypermagnesemia and respiratory arrest.
Calcium Carbonate: Calcium carbonate (Os-Cal, Tums, various other brands), commonly used as a calcium supplement or as an antacid, has phosphate-binding properties. It has been used for decades in patients with high serum phosphate who are undergoing dialysis and is one of the most commonly used phosphate binders in practice.2 The usefulness of calcium carbonate as a phosphate binder is limited by its insolubility at high gastric pH, which is common in those with renal disease.7 The greatest safety concern is hypercalcemia, which has the potential to cause arterial calcification and has been associated with cardiac death. Overall cost of this medication is low, so it is an attractive first-line agent if hypercalcemia is not a concern.8
Calcium Acetate: Approved in 1990, calcium acetate (PhosLo, various other brands) is considered a first-line therapy for lowering phosphate in CKD stage 4. An advantage for calcium acetate is less systemic absorption compared to other calcium salts, but it still can cause hypercalcemia. Calcium salts are associated with multiple drug interactions. Calcium acetate is fairly well tolerated but can be associated with hypercalcemia, nausea, and vomiting.2
Calcium-based phosphate binders are the mainstay of phosphate-lowering therapy in CKD stage 4.3,4 In stage 5, there is a greater increase in phosphate, and concomitant use of calcium-based phosphate binders leads to an increase in serum calcium and phosphate. When the serum calcium multiplied by the serum phosphorus is above 55, arterial calcification becomes a concern.3,4
Sevelamer Hydrochloride and Carbonate: Sevelamer hydrochloride (Renagel) was originally FDA-approved in 1998 for the treatment of hyperphosphatemia in hemodialysis patients and was approved in 2007 for patients on peritoneal dialysis.9 Sevelamer carbonate (Renvela) received an indication for hemodialysis in 2007. Sevelamer is an insoluble polymer that is not absorbed from the gastrointestinal (GI) tract and is considered as effective as calcium acetate or calcium carbonate in phosphorus-lowering ability.10 Sevelamer has been shown to decrease cardiovascular mortality in CKD patients.11 It can decrease absorption of various medications such as vitamins D, E, K, folic acid, levothyroxine, mycophenolate, tacrolimus, and quinolone antibiotics. Common adverse effects include vomiting, nausea, diarrhea, and dyspepsia. Sevelamer hydrochloride has the potential to reduce serum bicarbonate, which has led to some cases of metabolic acidosis in dialysis patients; this effect has not been reported in the carbonate form.11
Lanthanum Carbonate: Lanthanum carbonate (Fosrenol) is a trivalent cation rare-earth element that binds phosphate. It is insoluble in water and minimally absorbed from the GI tract. Progressive accumulation with continued use has been demonstrated in animals, and has been detected in human bone.12 Lanthanum is as effective as calcium carbonate, but with a much lower incidence of hypercalcemia. Upward dose titration may be required to keep the phosphate level <6 mg/dL. Like sevelamer, lanthanum is often associated with lowering mortality from cardiovascular problems; however, the mechanism for lanthanum is less clear.2
Newer Phosphate Binders
Two iron-based phosphate binders have been FDA-approved since 2013.13,14 These include sucroferric oxyhydroxide and ferric citrate. In some patients, the use of calcium-based phosphate binders can cause hypercalcemia; thus, calcium-free preparations are desirable. In addition, many patients with CKD are iron-deficient. Use of ferric citrate may mitigate the need for IV iron and erythropoietin-stimulating agents, which should be used with caution if used concomitantly. IV iron is associated with an increased risk of infection. This has not been reported in iron-based phosphate binders, but it remains a consideration.13
Sucroferric Oxyhydroxide: The first iron-based phosphate binder, sucroferric oxyhydroxide (Velphoro), was approved in 2013.15 This product is indicated for the treatment of hyperphosphatemia in patients with CKD on dialysis. Sucroferric oxyhydroxide is an iron(III) oxyhydroxide molecule bound to a carbohydrate molecule, with iron constituting approximately 20% of the molecular weight. Sucroferric oxyhydroxide uses a ligand exchange reaction with hydroxyl molecules to bind phosphorus in the GI tract. The active form of the drug is insoluble and cannot be metabolized or absorbed. Sucroferric oxyhydroxide was found to be noninferior to sevelamer carbonate in reducing serum phosphate in an open-label clinical trial of 1,059 patients.14 The sugar portion is also absorbable, with one tablet producing 1.4 grams of carbohydrates, which may be of concern for the diabetic patient.15 The initial dose is 500 mg three times daily with meals and can be titrated at weekly intervals by 500 mg/day until serum phosphorus levels are 5.5 mg/dL. The tablets should not be swallowed but can be chewed or crushed.15
The most common adverse effects of sucroferric oxyhydroxide in clinical trials were diarrhea, discolored feces (black), nausea, and abnormal taste.14-16 Sucroferric oxyhydroxide may affect absorption of some medications; alendronate and doxycycline should be separated by at least one hour and concurrent use of levothyroxine and vitamin D should be avoided entirely. Sucroferric oxyhydroxide has not been studied in patients who had conditions where iron accumulation is common or who had GI disorders. Caution should be used in patients who have peritonitis during peritoneal dialysis or hemochromatosis and immediately after GI surgery. Sucroferric oxyhydroxide has been studied in clinical trials for up to 52 weeks.16
Ferric Citrate: Ferric citrate (Auryxia) is an oral iron-based phosphate binder that was approved by the FDA in 2014 for management of hyperphosphatemia in patients with CKD on dialysis.17 Upon ingestion, this product dissociates into its ferric iron and citrate components. Iron in the ferric form binds phosphate in the GI tract and forms a precipitate of ferric phosphate, which is then excreted as fecal matter. Ferric citrate demonstrated similar phosphoric changes when compared to sevelamer and/or calcium citrate.18 Each 1-g tablet of ferric citrate contains 210 mg of ferric ion. Package labeling indicates a starting dose of 2 tablets orally 3 times per day with meals, adjusting the dose by 1 to 2 tablets as needed to maintain serum phosphorus levels at target, with the maximum being 12 tablets daily. This iron-containing product is contraindicated in iron-overload syndromes such as hemochromatosis. Patients should be monitored regularly for iron overload. Ferric citrate has the potential to decrease the absorption of doxycycline. Common adverse effects from clinical trials include diarrhea, discolored feces (dark), constipation, nausea, and vomiting.14 This product has been studied in clinical trials up to 52 weeks.17
Role of the Pharmacist
The pharmacist should be able to recognize when oral phosphate binders are needed and be familiar with the risks and benefits of available treatments. Additionally, pharmacists should be able to recommend patient-specific phosphate binders based upon electrolyte, anemic, and diabetic status, as well as infection risk. Finally, the pharmacist is in a key position to help patients optimize therapy with an understanding of drug interactions, adverse effects, medication costs, and overall pill burden.
Conclusion
Oral phosphate binders are widely used in clinical practice for patients with end-stage renal disease. It should be recognized that all phosphate binders have equal ability to maintain serum phosphorus in the targeted level provided the patient is compliant and tolerant of the treatment regimen. Calcium-based phosphate binders are often used for CKD stages 3 to 5; they are inexpensive, but have a potential to cause hypercalcemia. Recent advancements have been made in phosphate-binder treatment. Sevelamer and lanthanum can be used in the setting of hypercalcemia, and they offer a cardiovascular mortality benefit. The recently approved iron-based products may have a role in certain patients, but the long-term safety risk of these products has not been established. It is important for the pharmacist, as an essential member of the healthcare team, to be familiar with these new treatments in order to optimize therapy in the setting of hyperphosphatemia.
REFERENCES
1. Malesker MA, Morrow LE. Fluids and electrolytes. In: Chisholm-Burns MA, Wells BG, Schwinghammer TL, et al, eds. Pharmacotherapy Principles and Practice. 3rd ed. New York, NY: McGraw Hill; 2013.
2. Tonelli M, Pannu N, Manns B. Oral phosphate binders in patients with kidney failure. N Engl J Med. 2010;362(14):1312-1324.
3. Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Work Group. KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int. 2009;76(suppl 113):S1-S130.
4. National Kidney Foundation. K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis. 2003;42(4 suppl 3):S1-S201.
5. Schucker JJ, Ward KE. Hyperphosphatemia and phosphate binders. Am J Health Syst Pharm. 2005;62:2355-2361.
6. Salusky IB, Foley J, Nelson P, Goodman WG. Aluminum accumulation during treatment with aluminum hydroxide and dialysis in children and young adults with chronic renal disease. N Engl J Med. 1991;324:527-531.
7. Phosphate binders. Med Lett Drugs Ther. 2006;48(1228):15-16.
8. Lexi-Drugs. Lexi-Comp Online. Hudson, Ohio: Lexi-Comp, Inc. http://online.lexi.com. Accessed February 9, 2016.
9. Gold Standard, Inc. Sevelamer. Clinical Pharmacology [online database]. www.clinicalpharmacology.com. Accessed February 9, 2016.
10. Qunibi W, Hootkins R, McDowell L, et al. Treatment of hyperphosphatemia in hemodialysis patients: the Calcium Acetate Renagel Evaluation (CARE Study). Kidney Int. 2004;65(5):1914-1926.
11. Rastogi A. Sevelamer revisited: pleiotropic effects on endothelial and cardiovascular risk factors in chronic kidney disease and end-stage renal disease. Ther Adv Cardiovasc Dis. 2013;7(6):322-342.
12. Swainston Harrison T, Scott L. Lanthanum carbonate. Drugs. 2004;64(9):985-996.
13. Goldsmith D, Covic A. Oral phosphate binders in CKD—is calcium the (only) answer? Clin Nephrol. 2014;81(6):389-395.
14. Floege J, Covic AC, Ketteler M, et al. A phase III study of the efficacy and safety of a novel iron-based phosphate binder in dialysis patients. Kidney Int. 2014;86(3):638-647.
15. Velphoro (sucroferric oxyhydroxide) package insert. Waltham, MA: Fresenius Medical Care North America; September 2014.
16. Floege J, Covic A, Ketteler M, et al. Long-term effects of the iron-based phosphate binder, sucroferric oxyhydroxide, in dialysis patients. Nephrol Dial Transplant. 2015;30(6):1037-1046.
17. Auryxia (ferric citrate) package insert. New York, NY: Keryx Biopharmaceuticals, Inc; July 2015.
18. Van Buren PN, Lewis JB, Dwyer JP, et al. The phosphate binder ferric citrate and mineral metabolism and inflammatory markers in maintenance dialysis patients: results from prespecified analyses of a randomized clinical trial. Am J Kidney Dis. 2015;66(3):478-488.
19. Phoslyra (calcium acetate oral solution) package insert. Waltham, MA: Fresenius Medical Care North America; April 2011.
20. PhosLo (calcium acetate) gelcaps package insert. Waltham, MA: Fresenius Medical Care; October 2006.
21. Calcium carbonate package insert. Columbus, OH: Roxane Laboratories; 2008.
22. Renagel (sevelamer hydrochloride) package insert. Cambridge, MA: Genzyme Corporation; 2015.
23. Renvela (sevelamer carbonate) package insert. Cambridge, MA: Genzyme Corporation; 2011.
24. Fosrenol (lanthanum carbonate) package insert. Wayne, PA: Shire US Inc; September 2014.
To comment on this article, contact rdavidson@uspharmacist.com.






