US Pharm.
2008;33(3):28-47.
Vaccines are medical miracles,
saving more lives and preventing more deaths than any other medical advance in
the last century. Vaccines stimulate protective immune responses against acute
and chronic infectious diseases, as well as some infectious diseases that
cause cancer.1-4 Benefits of vaccines include protection from
symptomatic illness, improved quality of life and productivity, and prevention
of death.5 When the U.S. Department of Health & Human Services
(HSS) developed Healthy People 2010, a comprehensive, nationwide
health-promotion and disease-prevention campaign, one of their main areas of
focus was improving immunization rates in the United States.6-8
The pharmacist's role in
immunization has progressed considerably in the last decade.6
Currently, 46 states allow pharmacists to administer immunizations under a
collaborative-practice agreement with physicians.9 The goal of this
article is to educate and update practitioners on immunization statistics;
vaccine-preventable diseases; current immunization schedules; administration
essentials; available vaccines; vaccine adverse event reporting and patient
information; vaccine controversies; and the unique and empowering role
pharmacists can play in improving the U.S. public health.
Immunization Statistics
Despite widespread education, more
than 100,000 people are affected by vaccine-preventable diseases in the U.S.
each year. Twenty percent of children and approximately 50% of adolescents
have not received at least one recommended immunization.10,11 It
has been estimated that, in every birth cohort, vaccination with seven of the
12 routinely administered childhood vaccines prevents 33,000 deaths, prevents
14 million cases of disease, saves $10 billion in direct costs, and saves
society an additional $33 billion in costs that include disability and lost
productivity.1
Immunization Schedules
National
recommendations currently provide guidance for use of vaccines to prevent or
eliminate 17 diseases, namely, diphtheria; pertussis; tetanus; poliomyelitis;
measles; mumps; rubella; influenza; invasive Haemophilus influenzae
type b (Hib); hepatitis A; hepatitis B; rotavirus; varicella; herpes zoster
(shingles); and disease caused by many of the most important types of
Streptococcus pneumoniae, Neisseria meningitides, and human
papillomavirus (HPV).1 These recommendations are made by the
Advisory Committee on Immunization Practices (ACIP). The ACIP states that any
person or institution that provides vaccination services should adopt these
standards to improve vaccination delivery and protect infants, children,
adolescents, and adults from vaccine-preventable disease.5
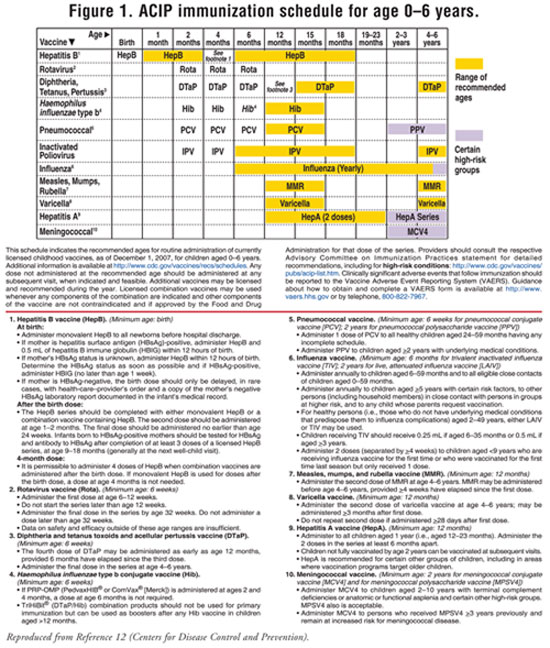
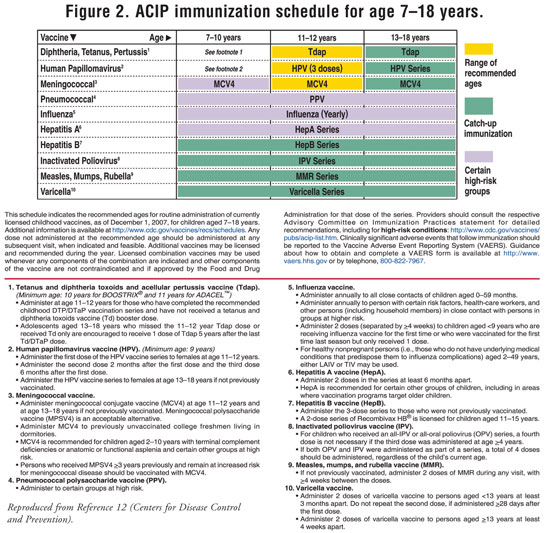
The current ACIP immunization
schedules for age 0 to 6 years and age 7 to 18 years appear in FIGURES 1
and 2, respectively; for the catch-up immunization schedule for age 4
months to 18 years, visit www.cdc.gov/mmwr/PDF/wk/mm5701-Immunization.pdf.
12
Vaccine-Preventable
Diseases
Diphtheria,
Tetanus, Pertussis13-17:
Diphtheria is an acute toxin-mediated infectious disease caused by
toxin-producing strains of Corynebacterium diphtheriae. This bacterium
lives in the nose and throat of an infected person and is easily spread by
coughing and sneezing. The toxin can cause severe or fatal illness
characterized by membranous inflammation of the upper respiratory tract and
damage to the myocardium and nervous system. Tetanus is caused by a potent
endotoxin released by Clostridium tetani, a bacterium commonly
found in soil, dust, and manure, and occurs when the bacterium comes in
contact with an open wound. It manifests primarily as neuromuscular
dysfunction, rigidity, and painful spasms of skeletal muscle. Once an
individual is infected, it can take three to 21 days for symptoms to appear.
Tetanus is not contagious, and only about 50 cases are reported in the U.S.
each year; however, one of every 10 cases is fatal. Pertussis (whooping cough)
is a respiratory-tract disease caused by Bordetella pertussis. Children
with pertussis develop thick, sticky mucus in the windpipe, which causes
coughing spells. Pertussis is highly contagious and can cause serious and
often fatal disease, particularly in infants under 6 months old. The roles of
the different components produced by B pertussis in the disease process
are not fully understood.
Polio18:
Poliovirus is an enterovirus belonging to the picornavirus family. Poliovirus
infections are generally asymptomatic or cause minimal symptoms, but about 2%
of infected individuals have central nervous system involvement and develop
paralytic disease. Polio has been controlled in the U.S. with the use of
vaccines, but worldwide eradication has not occurred. As a result, the ACIP
continues to recommend vaccination against poliovirus as a standard part of
the childhood immunization schedule.
Hepatitis A17,19,20
: Hepatitis A, a
member of the picornavirus family, is the most frequently reported type of
hepatitis in the U.S., causing an estimated 125,000 to 200,000 cases yearly.
About a third of these cases occur in children younger than 15 years. The
virus, found mainly in feces, is spread through personal contact or by
consuming contaminated food or water. The course of hepatitis A infection is
extremely variable, ranging from asymptomatic infection to fulminant hepatitis
and death. Hepatitis A does not cause long-term illness or liver damage,Ü but
about 100 people each year die from liver failure caused by severe hepatitis
A.
Hepatitis B21,22
: Hepatitis B is one
of several hepatitis viruses that cause a systemic infection, with the major
pathology in the liver. Currently there is no specific treatment for this
disease. The Centers for Disease Control and Prevention (CDC) estimates that
the U.S. has more than 125 million chronic carriers of hepatitis B. For the
entire U.S. population, the overall risk of contracting the virus is less than
20%. Serious complications and sequelae of hepatitis B infection include
massive hepatic necrosis, cirrhosis of the liver, and chronic active
hepatitis; it also is associated with an increased risk of hepatocellular
carcinoma. Modes of transmission include sexual contact with an infected
individual, percutaneous or mucosal exposure to infectious blood, and
perinatal exposure to an infected mother.
Hib23-26:
Prior to the introduction of Hib conjugate vaccines, this bacterial infection
was the most frequent cause of bacterial meningitis and a leading cause of
serious, systemic bacterial disease in young children worldwide. Invasive
disease usually occurs in children younger than 5 years of age and has a
mortality rate of 5%. Up to 35% of survivors develop neurologic sequelae
including seizures, deafness, and mental retardation.
HPV27:
HPV causes squamous cell cervical cancer, its associated precursor lesions,
cervical adenocarcinoma, 35% to 50% of vulvar and vaginal cancers, and genital
warts. The CDC estimates that 20 million U.S. women and men had HPV in 2005.
Many people with HPV may not show signs or symptoms. HPV is transmitted
through sexual activity involving genital contact with an infected person.
Traditionally, cervical cancer screening focused on routine screening and
early intervention, with a reduced cancer rate of 75%. Vaccination protects
against certain types of HPV, but it may not protect everyone and does not
prevent all types of cervical cancer; therefore regular cervical cancer
screening is recommended.
Influenza28-31
: Influenza is a
significant cause of death; along with pneumonia, it is the seventh leading
cause of death across generations. Rates of infection are highest among
children, and the virus causes more pediatric hospitalizations than any other
vaccine-preventable disease. Influenza vaccination is the primary method of
preventing influenza and its severe complications. As of February 27, 2008,
the ACIP recommends that all children age 6 months to 18 years receive an
annual flu vaccination.
Measles, Mumps, Rubella
17,32-35:
Measles, mumps, and rubella are viral childhood diseases that may be
associated with serious complications and/or death. Measles, which is highly
contagious, is characterized by a rash over the entire body and can cause
pneumonia and encephalitis. Children who are exposed and are not immune
probably will get the disease. Mumps is characterized by swelling of the
cheeks and jaw with resulting inflammation of the salivary glands; it can lead
to aseptic meningitis, deafness, and orchitis. Rubella is generally a mild
disease, but the greatest danger is to unborn babies. If a woman gets rubella
in early pregnancy, there is an 80% chance that the baby will be born deaf or
blind, with a damaged heart or small brain, or mentally retarded. These three
diseases are transmitted through coughing, sneezing, or breathing. Since
vaccination of these diseases began, five decades ago, the number of cases has
declined more than 99%.
Meningococcal Disease
36,37: N
meningitidis causes both endemic and epidemic meningococcal disease,
principally meningitis, severe swelling of the brain and spinal cord, and
meningococcemia. Because childhood immunization programs have led to decreased
rates of H influenzae and S pneumoniae infection, N meningitidis
has become the leading cause of bacterial meningitis in children and young
adults in the U.S. The incidence of disease is rare; only 1,400 to 2,800 cases
are reported in the U.S. However, the fatality rate remains at around 11.5%,
even with the administration of appropriate antibiotics.
Pneumococcal Disease
17,38,39:
Disease caused by S pneumoniae is a significant source of morbidity and
mortality in all ages worldwide. In children older than 1 month, S
pneumoniae is the leading cause of invasive disease. Children at highest
risk are between the ages of 1 month and 2 years. Pneumococcal meningitis in
children has been associated with an 8% mortality rate and may result in
neurologic sequelae and hearing loss in survivors.
Rotavirus40:
A leading cause of severe acute gastroenteritis in infants and young
children, rotavirus infects more than 95% of children by 5 years of age. While
many children may have few or no symptoms, rotavirus can cause fever and
episodes of vomiting and diarrhea lasting up to nine days that may lead to
dehydration and hospitalization. Only certain disinfectants can kill
rotavirus. The most severe cases occur in children aged 6 to 24 months.
Varicella41,42
: Varicella
(chickenpox), which is caused by the varicella-zoster virus, is a highly
contagious disease affecting both children and adolescents. The disease
generally manifests as 300 to 500 maculopapular and/or vesicular lesions and
is accompanied by a fever exceeding 100F. Between 1980 and 1994, approximately
3.5 million cases were reported each year, and the peak age for disease
occurrence was years 5 to 9. Although varicella is usually a benign,
self-limiting disease, it can be associated with serious complications (e.g.,
bacterial superinfection, pneumonia, encephalitis, Reye's syndrome) and death.
Vaccine Essentials
The ACIP has
developed general recommendations for vaccine administration (for the complete
document, see www.cdc.gov/vaccines).5 The immunization schedule is
based on many factors, including the nature of the vaccine and the age and
immune status of the recipient. Vaccines are recommended for members of the
youngest age group at risk for the disease for whom efficacy and safety data
have been reported.5 The following sections summarize specific
information that is important for pharmacists administering vaccinations.
Pharmacists administering
vaccines should adhere as closely as possible to the recommended immunization
schedules. In some circumstances, patients may need to receive a vaccination
earlier than required, such as when they are behind schedule or planning to
travel. Except for oral typhoid vaccine, an interruption in the vaccination
schedule does not require restarting of the entire vaccine series or the
addition of an extra dose. Pharmacists should accept only written, dated
records as evidence of vaccination (with the exception of influenza and
pneumococcal vaccines). If the patient's records cannot be located, the
patient should be considered susceptible and should be started on the
age-appropriate vaccination schedule.5
Simultaneous
Administration: The
ACIP encourages the simultaneous administration of vaccines. The practice of
administering all eligible vaccines during the same visit increases the
chances of a patient's being fully vaccinated by the appropriate age.5,43
The use of combination vaccines also may reduce the number of injections a
patient receives and increases the likelihood that the immunization series is
completed. Licensed combination vaccines can be used whenever any component of
the combination is indicated and the other components are not contraindicated.
When vaccines are given simultaneously, they should never be mixed in the same
vial or syringe; they should be administered at separate sites with different
syringes. The only exception to simultaneous administration is when two
injectable or nasally administered live vaccines are to be administered; they
should be spaced out by more than four weeks. Oral live vaccines can be
administered with other live or inactivated vaccines.5
Interchangeability of
Vaccines: For several
vaccine-preventable diseases, multiple brands are available from different
manufacturers. Due to differences in antigen content, amount, and method of
formulation, the ACIP recommends that the same brand be used throughout the
immunization series whenever feasible. If the same brand is not available or
the brand previously used is unknown, however, do not defer vaccination.
5,44,45
Contraindications and
Precautions: A
vaccine should never be administered if a contraindication is present. The
only universal contraindication for vaccines is a history of severe allergic
reaction after a previous dose of a vaccine or vaccine constituent. Live
vaccines generally should not be used in severely immunocompromised patients;
live-attenuated vaccines should not be used in pregnant women.5 See
Reference 15 for specific contraindications for vaccines with a pertussis
component.
A precaution might compromise
a vaccine's ability to produce immunity. Vaccination generally should be
deferred when a precaution exists; however, vaccination may be indicated if
the benefit of protection outweighs the risk of adverse reaction. Precautions
for all vaccines include moderate or severe acute illness and unstabilized
evolving neurologic conditions in children. All vaccines can be administered
during minor acute illness, and studies indicate that failure to vaccinate
during a minor illness can impede overall vaccination efforts.5,46
Patients with clotting-factor disorders are at risk for hematoma formation
after intramuscular (IM) injection. The ACIP has issued a caution for this
population: If, in the opinion of a physician familiar with the patient's
bleeding risk, the vaccine can be administered IM with reasonable safety, IM
administration can be used.5 See Reference 15 for specific
precautions for vaccines with a pertussis component.
Injection Route and Site:
All currently recommended pediatric and adolescent vaccines are administered
IM. This is due mainly to the presence of an adjuvant in the vaccine. If given
subcutaneously or intradermally, local irritation, induration, skin
discoloration, inflammation, and granuloma formation could occur. Also,
deviation from the recommended IM route could result in decreased vaccine
efficacy.5,47,48 IM injections are administered at a 90 angle to
the skin, preferably into the anterolateral aspect of the thigh or the deltoid
muscle of the upper arm, depending on the patient's age.5 See the
package insert for specific administration information.
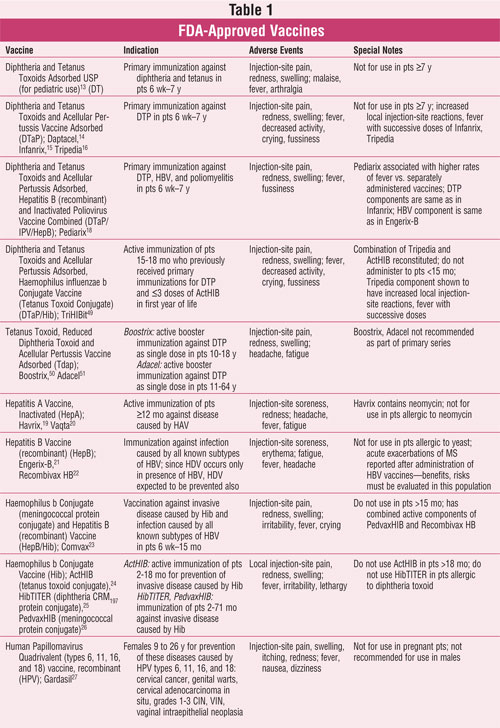
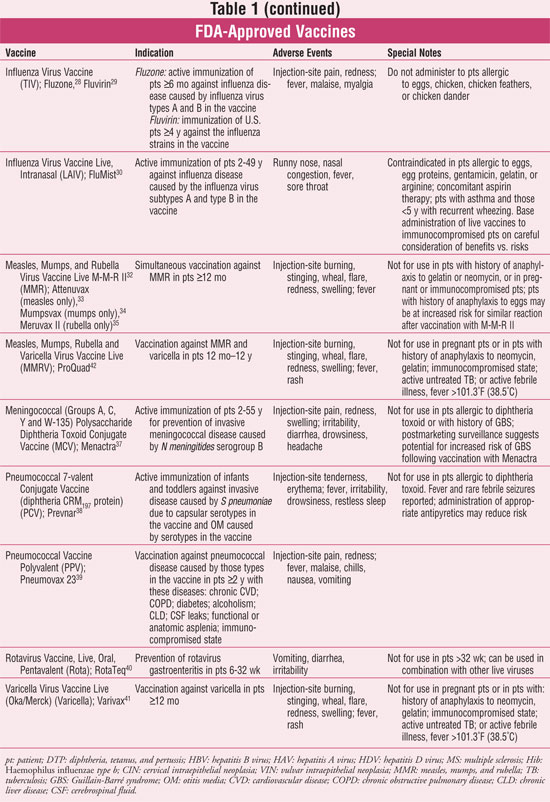
FDA-Approved Vaccines
In recent years, vaccines have been
combined to reduce the number of injections given to infants and adolescents.
The availability of multiple brands for the same vaccine-preventable disease
and the development of combination products for different indications can be
confusing for providers administering vaccines. See TABLE 1 for a list
of each vaccine with indications, adverse events, and additional important
information. The common abbreviation for the vaccine (e.g., DT), the vaccine's
makeup, and brand name(s) are included due to interchangeability in various
immunization references.
Vaccine Adverse Event Reporting System
The National Vaccine Injury
Compensation Program, established by the National Childhood Vaccine Injury Act
(NCVIA) of 1986, requires that the health care provider record the
manufacturer's name and the lot number of the vaccine administered in the
recipient's permanent medical record, along with the date of administration
and the name, address, and title of the provider who administered the vaccine.
The NCVIA also requires the health care provider to report the occurrence of
certain adverse events to the HSS Vaccine Adverse Event Reporting System
(VAERS).5 See http://vaers.hhs.gov/pdf/ReportableEventsTable.pdf
for a table of specific reportable events. VAERS forms are available at
www.vaers.hhs.gov/vaers.htm.52
Vaccine Controversies
There has been much controversy
about whether vaccines and autism are linked. This purported link dates to a
1998 study published in The Lancet that described 12 children with
inflammatory bowel conditions and regressive developmental disorders, mostly
autism.53,54 In eight of these cases, the child's parent(s) or
physician believed that the measles-mumps-rubella (MMR) vaccine may have
contributed to onset of the disorder.54 Independent researchers
have been unable to replicate this syndrome, and no correlation has been found
with onset of regression.53,55,56 After reviewing data on MMR
vaccine and risk of autism-spectrum disorders, the Immunization Safety Review
Committee (ISRC) of the Institute of Medicine (IOM) concluded that the data
favor rejection of a causal relationship.53,57
It has been speculated that
thimerosal, a mercury-containing preservative long used in vaccines, is linked
to the increased rates of autism. However, it was found that while autism's
incidence and prevalence in Sweden and Denmark grew rapidly in the 1990s,
average thimerosal exposure had begun to decrease in the late 1980s and was
virtually eliminated in those countries by the early 1990s.53,58
Another study has suggested that the associations found in some studies may
have been chance findings.59 The IOM ISRC determined that evidence
is sufficient to reject a causal association.57,59 The U.S. Public
Health Service and the American Academy of Pediatrics has called for the
removal of thimerosal from infant vaccines as a precautionary measure.58
To date, many vaccines no longer contain thimerosal and the remaining vaccines
that do are using other preservatives or are being switched over to
preservative-free formulations.59
Pharmacist's Role
The American Public
Health Association recently issued a policy statement calling on pharmacists
to increase their involvement in public health.60 The expanded role
of pharmacists in administering vaccines is an excellent beginning. In 2007,
the State of New Mexico began allowing pharmacists to prescribe and administer
vaccines after taking a 12-hour continuing-education program. This innovative
program has resulted in substantial gains in increased immunization rates,
especially in rural, underserved areas.61 Due to easy access,
advanced clinical training, and pre-existing relationships with many patients,
pharmacists are uniquely positioned to become significantly involved in
immunization education and administration and in improving public health in
the U.S.
REFERENCES
1. Roush SW, Murphy
TV. Historical comparisons of morbidity and mortality for vaccine-preventable
diseases in the United States. JAMA. 2007;298:2155-2163.
2. Mast EE, Weinbaum
CM, Fiore AE, et al. A comprehensive immunization strategy to eliminate
transmission of hepatitis B virus infection in the United States.
Recommendations of the Advisory Committee on Immunization Practices (ACIP)
part II: immunization of adults. MMWR Recomm Rep. 2006;55:1-33.
3. Mast EE, Margolis
HS, Fiore AE, et al. A comprehensive immunization strategy to eliminate
transmission of hepatitis B virus infection in the United States.
Recommendations of the Advisory Committee on Immunization Practices (ACIP)
part I: immunization of infants, children, and adolescents. MMWR
Recomm Rep. 2006;54:1-31.
4. Markowitz LE, Dunne
EF, Saraiya M, et al. Quadrivalent human papillomavirus vaccine.
Recommendations of the Advisory Committee on Immunization Practices (ACIP).
MMWR Recomm Rep. 2007;56:1-24.
5. Kroger AT, Atkinson
WL, Markuse EK, Pickering LK. General recommendations on immunization.
Recommendations of the Advisory Committee on Immunization Practices (ACIP).
MMWR Recomm Rep. 2006;55:1-48.
6. Sokos DR.
Pharmacists' role in increasing pneumococcal and influenza vaccination. Am
J Health Syst Pharm. 2005;62:367-377.
7. US Department of
Health & Human Services. Healthy People 2010.
www.healthypeople.gov/publications/cornerstone.pdf. Accessed Feb 20, 2008.
8. US Department of
Health & Human Services. Healthy People 2010. 14: Immunization and infectious
diseases. www.healthy people.gov/document/html/Volume1/14Immunization.htm.
Accessed Feb 20, 2008.
9. American Pharmacists
Association. www.aphanet.org. Accessed Feb 20, 2008.
10. Cannon HE. Pharmacy
management of vaccines. J Manag Care Pharm. 2007;13(suppl 7 B):S7-S11.
11. Poland GA, Shefer
AM, McCauley M, et al. Standards for adult immunization practices. Am J
Prev Med. 2003;25:144-150.
12. CDC. Recommended
immunization schedules for persons aged 0ñ18 years--United States, 2008.
MMWR. 2008;57:Q1-Q4.
13. Diphtheria and
Tetanus Toxoids Adsorbed USP (for pediatric use) package insert. Swiftwater,
PA: Aventis Pasteur; Oct 2001.
14. Daptacel
(diphtheria and tetanus toxoids and acellular pertussis vaccine adsorbed)
package insert. Swiftwater, PA: Sanofi Pasteur; Mar 2003.
15. Infanrix
(diphtheria and tetanus toxoids and acellular pertussis vaccine adsorbed)
package insert. Research Triangle Park, NC GlaxoSmithKline Beecham; Aug 2007.
16. Tripedia
(diphtheria and tetanus toxoids and acellular pertussis vaccine adsorbed)
package insert. Swiftwater, PA: Aventis Pasteur; Dec 2003.
17. CDC. Parents' guide
to childhood immunizations.
www.cdc.gov/vaccinespubs/parents-guide/downloads/2008-parents-guide.pdf.
Accessed Mar 4, 2008.
18. Pediarix
(diphtheria and tetanus toxoids and acellular pertussis vaccine adsorbed,
hepatitis B [recombinant] and inactivated poliovirus vaccine combined) package
insert. Research Triangle Park, NC: GlaxoSmithKline; Jun 2007.
19. Havrix (hepatitis A
vaccine, inactivated) package insert. Research Triangle Park, NC:
GlaxoSmithKline; Dec 2006.
20. Vaqta (hepatitis A
vaccine, inactivated) package insert. Whitehouse Station, NJ: Merck & Co; Dec
2007.
21. Engerix-B
(hepatitis B vaccine [recombinant]) package insert. Research Triangle Park,
NC: GlaxoSmithKline; Dec 2006.
22. Recombivax HB
(hepatitis B vaccine [recombinant]) package insert. Whitehouse Station, NJ:
Merck & Co; Dec 2007.
23. Comvax (haemophilus
b conjugate [meningococcal protein conjugate] and hepatitis B [recombinant]
vaccine) package insert. Whitehouse Station, NJ: Merck & Co; Aug 2004.
24. ActHIB (haemophilus
b conjugate vaccine [tetanus toxoid conjugate] package insert. Swiftwater, PA:
Sanofi Pasteur; Dec 2005.
25. HibTITER
(haemophilus b conjugate vaccine [diphtheria CRM197 protein
conjugate] package insert. Philadelphia, PA: Wyeth Pharmaceuticals; Jan 2007.
26. Liquid PedvaxHIB
(haemophilus b conjugate vaccine [meningococcal protein conjugate]) package
insert. West Point, PA: Merck & Co; Jan 2001.
27. Gardasil (human
papillomavirus quadrivalent [types 6, 11, 16, and 18) vaccine, recombinant)
package insert. Whitehouse Station, NJ: Merck & Co; Dec 2007.
28. Fluzone (influenza
virus vaccine) package insert. Swiftwater, PA: Sanofi Pasteur; Jun 2007.
29. Fluvirin (influenza
virus vaccine) package insert. Emeryville, CA: Novartis Vaccines and
Diagnostics; Apr 2007.
30. FluMist (influenza
virus vaccine live, intranasal) package insert. Gaithersburg, MD: MedImmune
Vaccines; Sept 2007.
31. CDC. Press release.
www.cdc.gov/od/oc/media/pressrel/2008/r080227.htm. Accessed Mar 4, 2008.
32. M-M-R II (measles,
mumps, and rubella virus vaccine live) package insert. Whitehouse Station, NJ:
Merck & Co; Dec 2007.
33. Attenuvax (measles
virus vaccine live) package insert. Whitehouse Station, NJ: Merck & Co; Sept
2002.
34. Mumpsvax (mumps
virus vaccine live) package insert. Whitehouse Station, NJ: Merck & Co; Sept
2002.
35. Meruvax II (rubella
virus vaccine live) package insert. Whitehouse Station, NJ: Merck & Co; Feb
2006.
36. Menomune
(meningococcal polysaccharide vaccine, groups A, C, Y and W-135 combined)
package insert. Swiftwater, PA: Sanofi Pasteur; Dec 2005.
37. Menactra
(meningococcal [groups A, C, Y and W-135 polysaccharide diphtheria toxoid
conjugate vaccine) package insert. Swiftwater, PA: Sanofi Pasteur; Oct 2007.
38. Prevnar
(pneumococcal 7-valent conjugate vaccine [diphtheria CRM197
protein]) package insert. Philadelphia, PA: Wyeth Pharmaceuticals; Dec 2007.
39. Pneumovax 23
(pneumococcal vaccine polyvalent) package insert. Whitehouse Station, NJ:
Merck & Co; Sept 2007.
40. RotaTeq (rotavirus
vaccine, live, oral, pentavalent) package insert. Whitehouse Station, NJ:
Merck & Co; Sept 2007.
41. Varivax (varicella
virus vaccine live [Oka/Merck]) package insert. Whitehouse Station, NJ: Merck
& Co; Feb 2007.
42. ProQuad (measles,
mumps, rubella and varicella virus vaccine live) package insert. Whitehouse
Station, NJ: Merck & Co; Jan 2008.
43. National Vaccine
Advisory Committee. Standards for child and adolescent immunization practices.
Pediatrics. 2003;112:958-963.
44. CDC. Pertussis
vaccination: use of acellular pertussis vaccines among infants and young
children. Recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 1997;46:1-25.
45. CDC. Use of
diphtheria toxoid-tetanus toxoid-acellular pertussis vaccine as a five-dose
series. Supplemental recommendations of the Advisory Committee on Immunization
Practices (ACIP). MMWR Recomm Rep. 2000;49:1-8.
46. Szilagyi PG,
Rodewald LE. Missed opportunities for immunizations: a review of the evidence.
J Public Health Manag Pract. 1996;2:18-25.
47. Shaw FE, Guess HA,
Roets JM, et al. Effect of anatomic injection site, age and smoking on the
immune response to hepatitis B vaccination. Vaccine. 1989;7:425-430.
48. Zuckerman JN. The
importance of injecting vaccines into muscle. Different patients need
different needle sizes. BMJ. 2000;321:1237-1238.
49. TriHIBIT
(haemophilus b conjugate vaccine [tetanus toxoid conjugate]) package insert.
Swiftwater, PA: Sanofi Pasteur; Dec 2005.
50. Boostrix (tetanus
toxoid, reduced diphtheria toxoid and acellular pertussis vaccine adsorbed)
package insert. Research Triangle Park, NC: GlaxoSmithKline; Jun 2007.
51. Adacel (tetanus
toxoid, reduced diphtheria toxoid and acellular pertussis vaccine adsorbed)
package insert. Swiftwater, PA: Sanofi Pasteur; Jan 2006.
52. Vaccine Adverse
Event Reporting System. Reportable events table.
http://vaers.hhs.gov/pdf/ReportableEventsTable.pdf. Accessed Feb 25, 2008.
53. DeStefano F.
Vaccines and autism: evidence does not support a causal association. Clin
Pharmacol Ther. 2007;82:756-759.
54. Wakefield AJ, Murch
SH, Anthony A, et al. Ileal-lymphoid-nodular hyperplasia, non-specific
colitis, and pervasive developmental disorder in children. Lancet.
1998;351:637-641.
55. Fombonne E,
Chakrabarti S. No evidence for a new variant of measles-mumps-rubella-induced
autism. Pediatrics. 2001;108:E58.
56. Richler J, Luyster
R, Risi S, et al. Is there a ëregressive phenotype' of autism spectrum
disorder associated with the measles-mumps-rubella vaccine? A CPEA study. J
Autism Dev Disord. 2006;36:299-316.
57. Immunization Safety
Review Committee. Immunization Safety Review. Vaccines and Autism.
Board of Health Promotion and Disease Prevention, Institute of Medicine.
Washington, DC: National Academy Press; 2004.
58. Stehr-Green P, Tull
P, Stellfeld M, et al. Autism and thimerosal-containing vaccines: lack of
consistent evidence for an association. Am J Prev Med. 2003;25:101-106.
59. FDA. Thimerosal in
vaccines. www.fda.gov/cber/vaccine/thimerosal.htm.
60. Vincent WR, Smith
KM, Steinke D. Opportunities for pharmacists in public health. Am J Health
Syst Pharm. 2007;64:2002-2007.
61. Brake AD, Vonderau
BL. Pharmacists as immunization providersñthe impact on New Mexico school
employees and their dependents. J Manag Care Pharm. Mar 2008. In press.
To comment on this article, contact
editor@uspharmacist.com.





